American Mineralogist: Journal of Earth and Planetary Science:
All our papers are special, so each month, the American Mineralogist editors will share highlights on each. We hope this information is enjoyable and useful.
The links below will take you to American Mineralogist on GSW (https://pubs.geoscienceworld.org/ammin) -- if your access is via your institution,Z you should seamlessly be able to access everything. If you are an MSA member, then first go to http://www.msapubs.org to authenticate in (pro tip use a different tab) then you should be able to click the links and see the full article. If you want paper-per-view or other options available from GSW, click the one of the full-text choices on that site and read the options carefully. After a while, Crossref may direct links to the Am Min site at Degruyters. Use the methods above if you wish to view on our society page or GSW.
For past volumes 98-103 (2013-2018) please click here.
American Mineralogist Volume 108
March 2024
The composition of mackinawite
https://doi.org/10.2138/am-2023-8943A critical statistical meta-analysis by Rickard shows that the composition of the tetragonal iron sulfide, mackinawite, is stoichiometric Fe1.0S. In the 60 years since its discovery, various formulations of mackinawite composition, such as Fe1+xS, have been listed in mineralogical databases and used by researchers. These formulations are wrong. Most mackinawites are simple FeS, but distinct cobaltian, nickelian, and (possibly) cupriferous varieties exist where minor elements substitute for Fe in the mackinawite structure. The result is fundamental to understanding the role of mackinawite in geologic, environmental, biologic, and industrial processes.
Halogen fractionation during vapor-brine phase separation revealed by in situ Cl, Br, and I analysis of scapolite from the Yixingzhai gold deposit, North China Craton
https://doi.org/10.2138/am-2022-8826Gao et al. examine scapolite from the Yixingzhai deposit, which is formed from oxidized and high salinity magmatic-hydrothermal fluids under high temperature and low-pressure conditions. The extremely high Br/Cl and I/Cl values of the scapolite mainly result from halogen fractionation during vapor-brine phase separation where Br and I preferentially partition into the brine phase compared to Cl the separation.
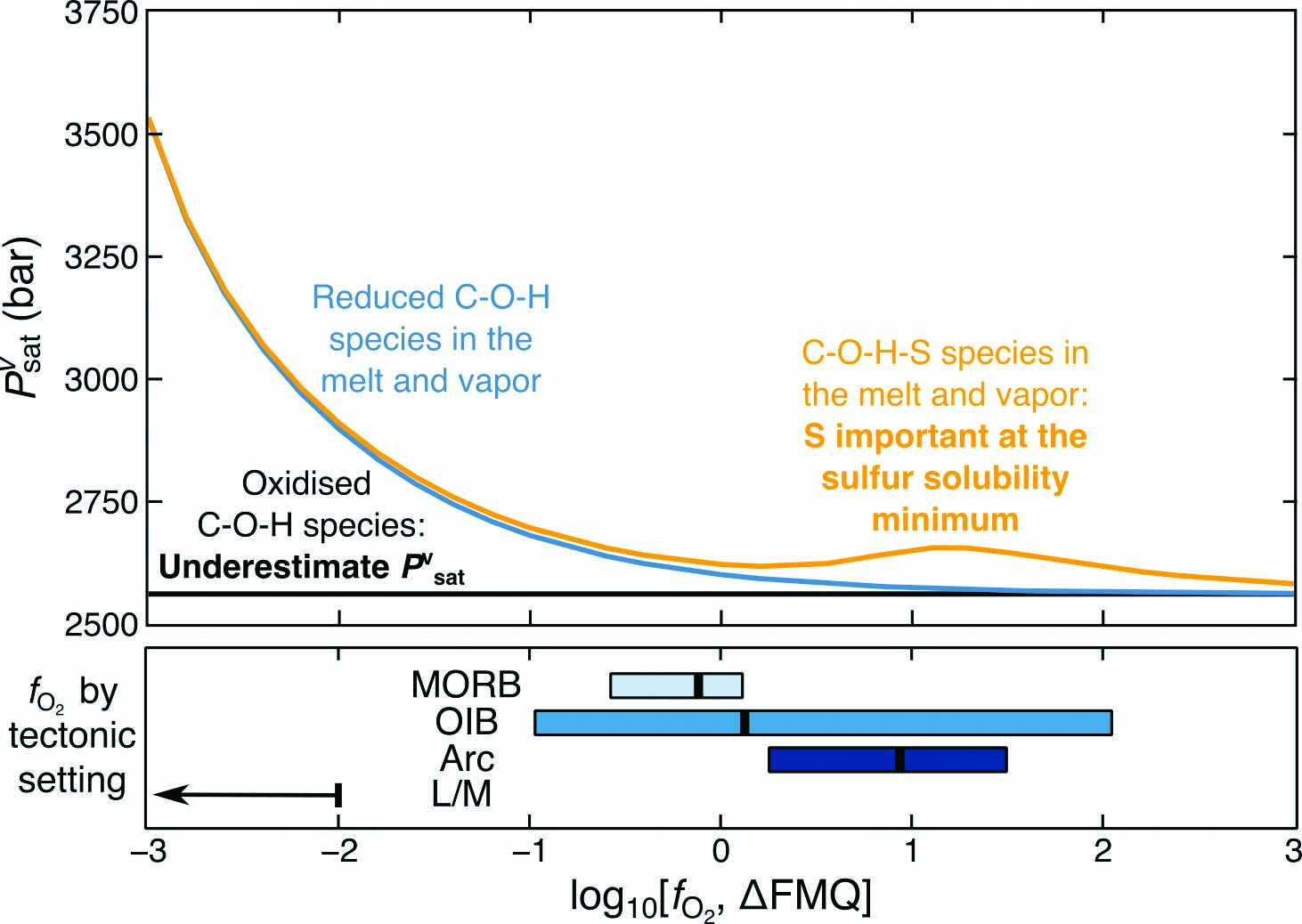
The effects of oxygen fugacity and sulfur on the pressure of vapor-saturation of magma
https://doi.org/10.2138/am-2022-8739The volatile content of the melt can be used as a barometer if the melt is vapor-saturated (pressure of vapor-saturation: Pvsat). Most Pvsat calculations assume the melt and vapor contain only oxidized C-O-H species. Hughes et al. show that the exclusion of reduced C-O-H species results in large Pvsat underestimations at reduced conditions while excluding S causes underestimates of Pvsat at intermediate redox conditions. Hence, magma depths are deeper than previously calculated at these conditions.
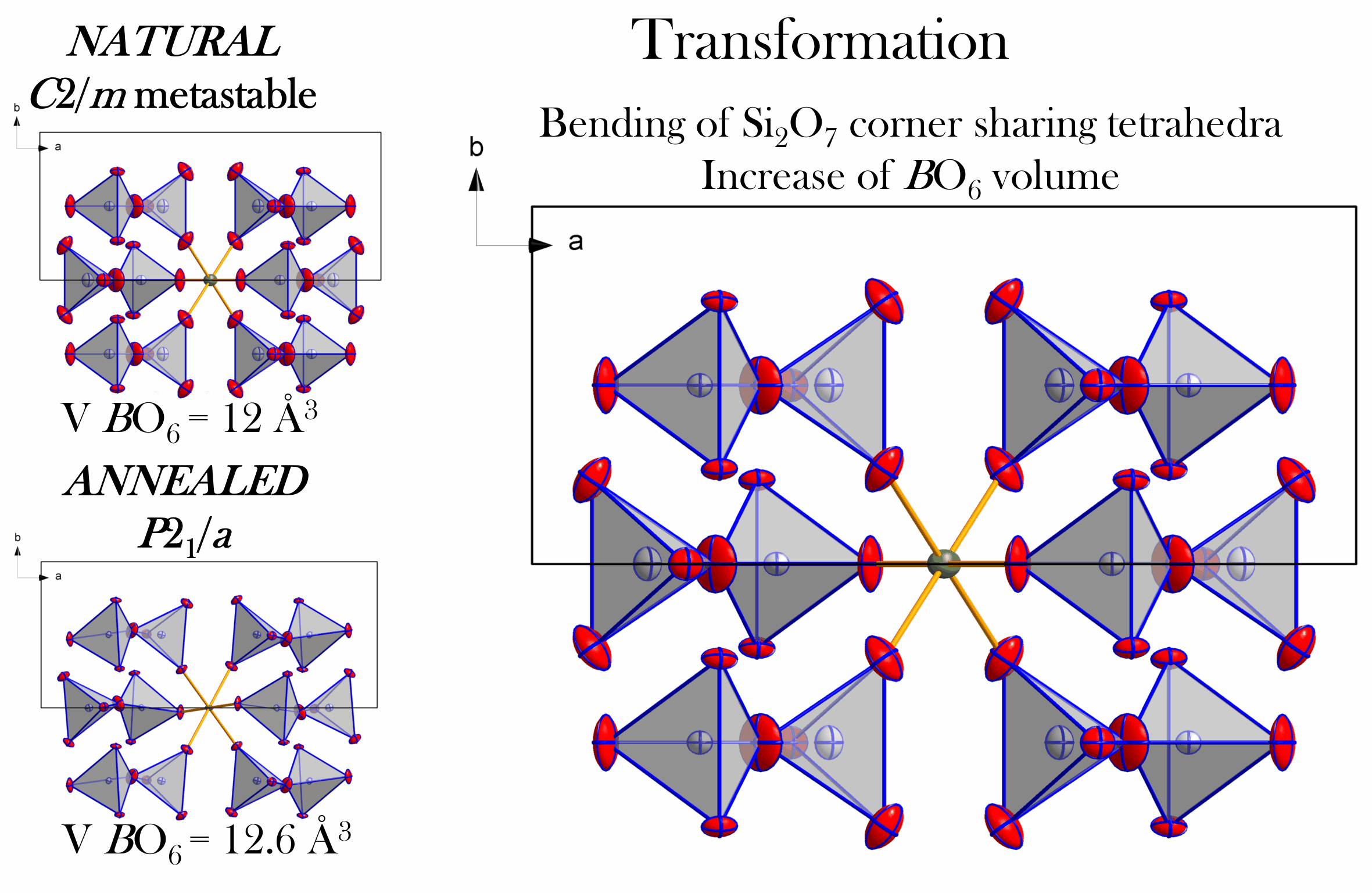
A revisit to phase transition behavior of K-feldspar at high-pressure and high-temperature: Implications on metastable K-feldspar in cold subduction
https://doi.org/10.2138/am-2022-8877To better understand the phase relations of K-feldspar in cold subduction, He and Li carried out a series Raman measurements and quenching experiments to systematically study the effect of pressure and temperature on the phase behavior of K-feldspar and produced a more comprehensive P-T phase diagram of K-feldspar than previously known. K-feldspar undergoes a pressure-induced transition from C-1 to P-1 (metastable K-feldspar) symmetry at ~10.2 GPa. Metastable K-feldspar is an intermediate phase from K-feldspar to K-holl-I in extremely cold subduction slabs (<2 °C/km) or the center of some old and cold subducting slabs. Metastable K-feldspar is stable even at 11.4 GPa and 400°C in enriched hydroxyl group (OH) environments, and shows greater high P-T stability than K-feldspar. This study could help us to explore the possible formation paths and conditions of K-holl-I, the existing depth of K-feldspar stability, and have furthered the understanding of continental subduction.
Equation of state and structural evolution of manganese dolomite (kutnohorite) under high pressures
https://doi.org/10.2138/am-2023-8949Xu et al. studied manganese dolomite at various high pressures. They report: (1) X-ray diffraction patterns of single-crystal manganese dolomite at pressures up to 51.2 GPa; (2) The equation of state of manganese dolomite from 1 bar to ~13.3 GPa; and (3) The split and disappearance of diffraction spots up to 51 GPa indicating the highly distorted rhombohedral structure of manganese dolomite.
A possible origin of the lunar spinel-bearing lithologies as told by the meteorite NWA 13191
https://doi.org/10.2138/am-2022-8728Pink spinel anorthosite (PSA) and pink spinel troctolite (PST) are two lunar lithologies known to contain Mg-rich spinel. They are widely distributed on the Moon's near and far sides. The study of their petrogenesis is important to understand the evolution of the Lunar crust. Xie et al. report their study of NWA 13191, a recently approved lunar meteorite that contains a large amount of spinel-bearing clasts, and discuss its origin. The characteristics of its petrology and mineralogy indicate that the Mg-Al-rich melt may be produced by the impact melting of Mg-rich anorthosite precursors. The spinel is a metastable crystallization product along with plagioclase and vitric melt near the Moon's surface. This realization provides observational evidence for previous simulated crystallization experiments and theoretical speculations.
Vergasovaite to cupromolybdite topotactic transformation with crystal shape preservation
https://doi.org/10.2138/am-2022-8753Whereas SC-SC transformations have been studied in detail to date, well-documented topotactic transitions in pure inorganic compounds and minerals with crystal shape preservation are as yet very rare. The thermal behavior of vergasovaite, Cu3O(SO4)(MoO4), and its synthetic analog was studied by Nazarchuk et al. by high-temperature single-crystal X-ray diffraction. The mineral is stable until 950; at 975 K, the topotactic transformation of vergasovaite into cupromolybdite, Cu3O(MoO4)2 occurs.
Thermal conductivity of aluminous garnets in Earth's deep interior
https://doi.org/10.2138/am-2023-8953Hung et al. precisely determined the thermal conductivity of aluminous garnets under high pressure-temperature conditions, which significantly advances our understanding of the thermal properties of garnets and their impacts on the complex thermochemical evolution in Earth's deep interior. Their results also offer an important basis for future novel design and fabrication of laser optics and electronic devices based on a variety of synthetic non-silicate garnets.
Interaction of seawater with (ultra)mafic alkaline rocks -- Alternative process for the formation of aegirine
https://doi.org/10.2138/am-2023-8928Rapprich et al. present a new model for the origin of aegirine in (ultra)mafic and relatively Na-poor alkaline rocks from Central Europe, which did not experience significant magmatic differentiation. Their data suggest that the late- to post-emplacement alteration of a mafic alkaline rock due to its contact with seawater leads to the degradation of diopside, resulting in the growth of secondary aegirine. The observed transformation of diopside into aegirine does not lead to a significant enrichment of the rock in Na (sourced from the seawater), as the process is blocked by a lack of available ferric iron. This effect may explain the limited Na-enrichment of submarine alkaline rocks, which is negligible compared to large-scale sodic-metasomatism of MORB tholeiites acquired during spilitization.
Experimental study on the kinetics of magnesiohornblende dehydration and its implications
https://doi.org/10.2138/am-2022-8692This experimental study by Han et al. provides a further understanding of seismic and high-electrical-conductivity anomalies in the subduction zone. This research will be of interest to colleagues who study the physical and chemical properties of hydrous minerals in subduction zones.
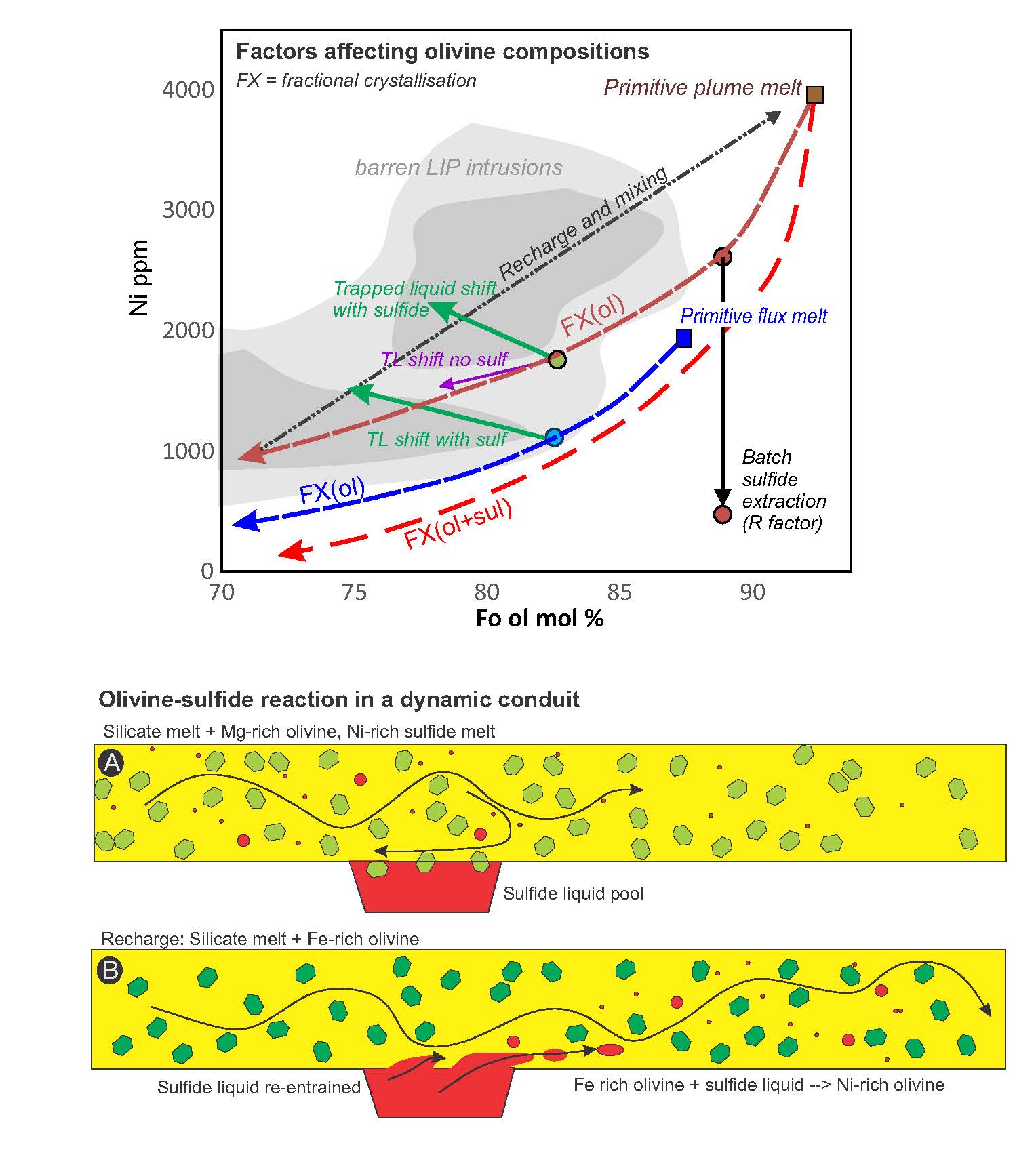
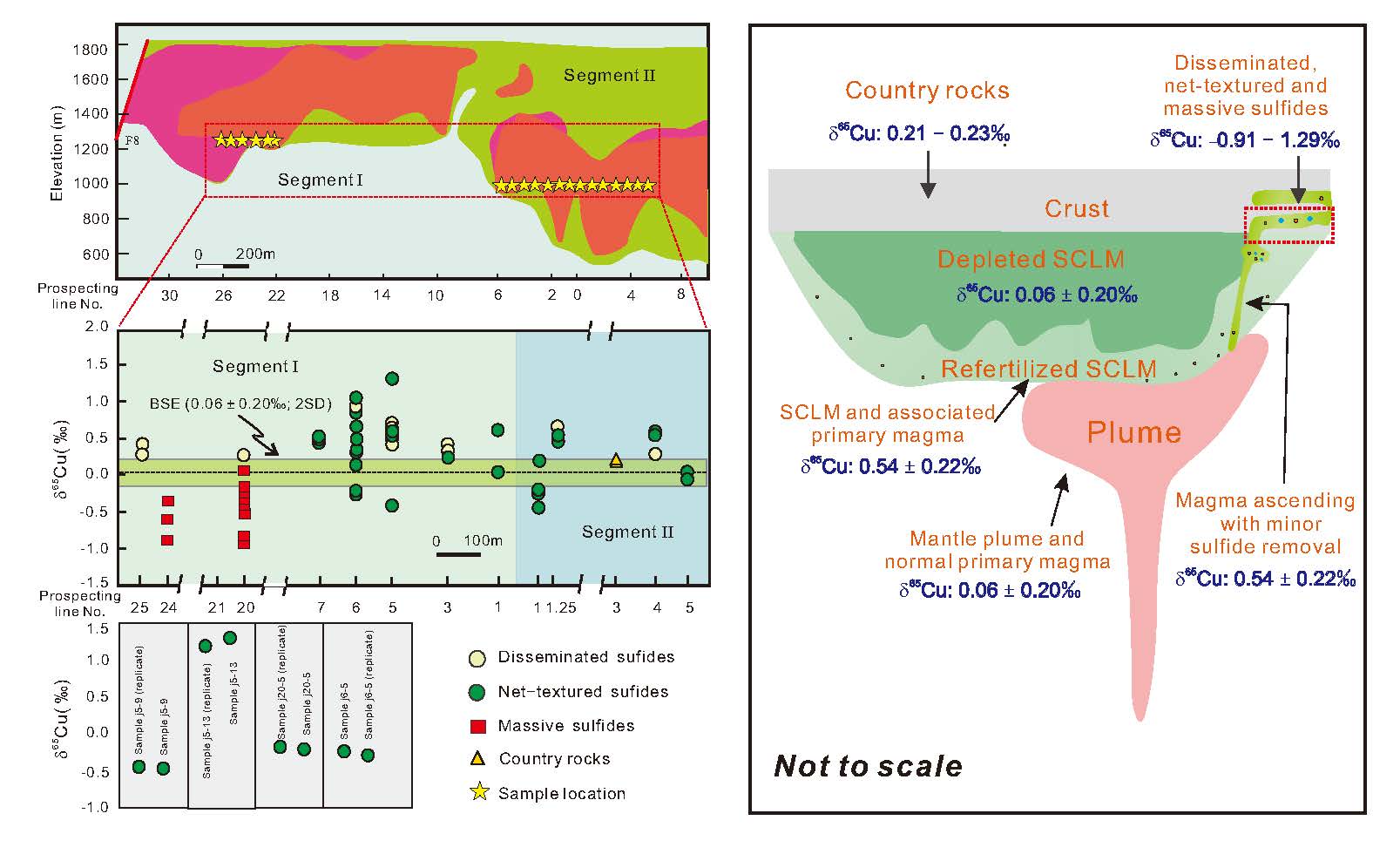
Predicting olivine formation environments using machine learning and implications for magmatic sulfide prospecting
https://doi.org/10.2138/am-2022-8893The natural formation from various mantle melts at diverse environments makes olivine one of the most informative messengers from Earth's mantle and a recorder of the fate of magma related to important mineral resources. Here, Xue et al. train a machine-learning-based model using a global olivine dataset to discriminate between olivine-forming environments and evaluate the ore deposit potential of magmatic sulfide mineralization. Thisr study documents that this model is excellent in discriminating between olivine origins and provides a reliable prospecting indicator for global Ni-Cu-PGE exploration.
Reaction between volatile-bearing eclogite and harzburgite as a function of degree of interaction: experimental constraints at 4 GPa
https://doi.org/10.2138/am-2022-8799The mantle is known to be heterogeneous, mainly composed of peridotite and eclogite. Eclogite-derived hydrous melts may interact with harzburgite in subduction zones and in the sub-continental lithospheric mantle. Elazar and Kessel studied the interaction of volatile-bearing eclogite-derived melt and harzburgite as a function of the degree of interaction. They experimentally simulated such interaction by using a capsule containing an eclogite layer juxtaposed next to a harzburgite layer in the presence of H2O and CO2 at 4 GPa and 1200 °C. A diamond trap was placed between the two layers to trap the fluid or melt, allowing direct determination of its composition. The multi-anvil apparatus was rotated at different frequencies to examine the effect of the increasing degree of interaction on the melt composition as well as the mineral compositions. The interaction of eclogite-derived hydrous melt with harzburgite results in a Opx + garnet reaction layer at the interface between the two lithologies. The interaction of the melt with the harzburgite refertilizes it, forming a lherzolite rock containing olivine, Opx, Cpx, and garnet. Melt is formed during this interaction. Both the eclogitic and the peridotitic garnet compositions approach each other and become intermediate between the composition of the garnet in the eclogite+H2O+CO2 system and the garnet in the harzburgite+H2O+CO2 system at these conditions. The Mg# of the peridotitic olivine and Opx is reduced. The alkalinity of the melt increases with increasing degrees of interaction. With increasing interaction, the mode of the melt fraction, peridotitic Opx, Cpx and garnet increase on the expense of peridotitic olivine and the eclogitic garnet. These experiments demonstrate the influence of the degree of interaction on the range of melt compositions found in volcanic arcs as well as the degree of metasomatism in the mantle found in the sub-continental lithospheric mantle.
Thermal behavior of borax Na2B4O5(OH)4·8H2O
https://doi.org/10.2138/am-2023-8970Borate minerals display extensive structural complexity resulting from the great diversity of their fundamental building blocks (FBBs), which are similar to that of silicates. The FBB in borax is an isolated B4O5(OH)4 cluster consisting of two BO2(OH) triangles and two BO3(OH) tetrahedra linked by common oxygen atoms to form a double ring with two common tetrahedra. Nishiyasu and Kyono reveal that a large electron cloud shared on the two borate tetrahedra leads to an extremely rigid B4O5(OH)4 cluster. Thus, once the B4O5(OH)4 cluster is formed, it is very stable as long as it is not exposed to strong acidic environments. In conclusion, the existence of borate minerals containing the B4O5(OH)4 cluster should provide evidence of the existence of moderately acidic or alkaline water where the borate minerals were grown via the incorporation of B4O5(OH)4 clusters. These types of clusters, consisting of BO3 triangles and BO4 tetrahedra, in borate minerals can potentially be used as a paleoenvironmental indicator.
Multiple magmatic processes revealed by distinct clinopyroxene populations in the magma plumbing system: A case study from a Miocene volcano in West Qinling, Central China
https://doi.org/10.2138/am-2022-8744Clinopyroxene (Cpx) with various origins (phenocryst, antecryst, xenocryst, etc.) and textures (e.g., zoning texture and spongy texture) in a basaltic plumbing system could provide information about the deep storage, mixing, migration, and physicochemical conditions of magmas. Zhang et al. carried out detailed petrographic, mineralogical, and whole-rock geochemical analyses to unravel the origins of various Cpx crystals and the reaction between Cpx xenocrysts/antecrysts and melts/fluids in the lithosphere. The detailed investigation of compositional and textural features of Cpx antecrysts/xenocrysts in this study suggests that the interactions between various interconnected magma reservoirs are widespread in the magmatic plumbing system. Their study emphasizes the importance of Cpx-melt/fluid reactions that can significantly modify the whole-rock compositions and lead to the formation of spongy textures without the need for fractures or cracks in minerals.
Genetic implications, composition, and structure of trioctahedral micas in xenoliths related to Plinian eruptions from the Somma-Vesuvius volcano (Italy)
https://doi.org/10.2138/am-2022-8782Balassone et al. present a detailed, systematic mineralogical and petrographic study of mica-bearing xenoliths (mainly skarns and composite skarns-marbles, with minor cumulates and a syenite) from the Somma-Vesuvius volcano (Roman Magmatic Province, southern Italy), that are related to the Pompeii (AD 79) and Avellino (3945 ± 10 cal yr BP) eruptions. The crystal-chemical and structural features of the Pompeii and Avellino micas are characterized, extending the known range of the other Vesuvian micas from literature. Genetic inferences are also given and provide valuable information for reconstructing the environmental conditions in similar geological systems. For example, the Ti-depletion and wide degree of dehydrogenation of phlogopites from skarns and composite skarns-marbles suggest that the studied samples originated under variable pressure conditions.
Magmatic and hydrothermal controls on diverse Nb mineralization associated with carbonatite-alkaline complexes in the southern Qinling orogenic belt, Central China
https://doi.org/10.2138/am-2022-8651Few carbonatites and/or alkaline rocks contain significant amounts of Nb minerals for economic exploitation; pyrochlore-group minerals generally are the dominant primary Nb-rich minerals in these rocks. In some cases, the occurrence of diverse Nb mineralization, such as columbite and Nb-rutile, may impede beneficiation and metallurgy. Ma et al. provide strong evidence that late-stage external or authigenic hydrothermal fluids could not remobilize Nb on a large scale as is observed for REE. This could result in the decoupling of REE and Nb during magmatic evolution or hydrothermal overprinting, suggesting that hydrothermal veins are potential targets for REE exploration whereas the complex itself could be the target for Nb exploration. In particular, this indicates that more attention should be paid to the country rocks, especially those rich in U or reduced, that may contribute to U enrichment and columbitization during magma ascent and hydrothermal overprinting, respectively, which may result in the diversity of Nb mineralization.
Potassium isotope fractionation during silicate-carbonatite melt immiscibility and phlogopite fractional crystallization
https://doi.org/10.2138/am-2022-8898Potassium (K) isotopes have been precisely measured since 2016 and explored in geochemistry and cosmochemistry. The available K isotope data reveal large δ41K variations in igneous rocks, which were mostly attributed to control of source composition. Magma differentiation is considered to have an insignificant effect on K isotope fractionation, mainly because of lack of studies on more-evolved magmatic rocks. Su et al. measured K isotopic compositions of a magmatic suit of alkaline silicate-carbonatite affinity, which were formed from melt immiscibility and subsequent phlogopite fractionation. Significant K isotope fractionation (1‰) induced by melt immiscibility and phlogopite fractionation span the δ41K range of most igneous rocks. This reveals an alternate mechanism of K isotope fractionation, which is important to constrain K recycling and K isotope variation in geological processes. This paper highlights (1) K isotope fractionation during silicate-carbonatite melt immiscibility; (2) K isotope fractionation induced by phlogopite fractional crystallization; and (3) Distinct mechanisms of K recycling and K isotope variation in various tectonic settings.
Yuchuanite-(Y), Y2(CO3)·H2O, a new hydrous yttrium carbonate mineral from the Yushui Cu deposit, South China
https://doi.org/10.2138/am-2023-8971Yao et al. report a new hydrous yttrium carbonate mineral, yuchuanite-(Y), Y2(CO3)·H2O. Yuchuanite-(Y) has been approved by the International Mineralogical Association Commission on New Minerals, Nomenclature and Classification (IMA2022-120). Moreover, this new mineral has important implications for heavy rare earth mineralization in Yushui and other sediment-hosted Cu deposits.
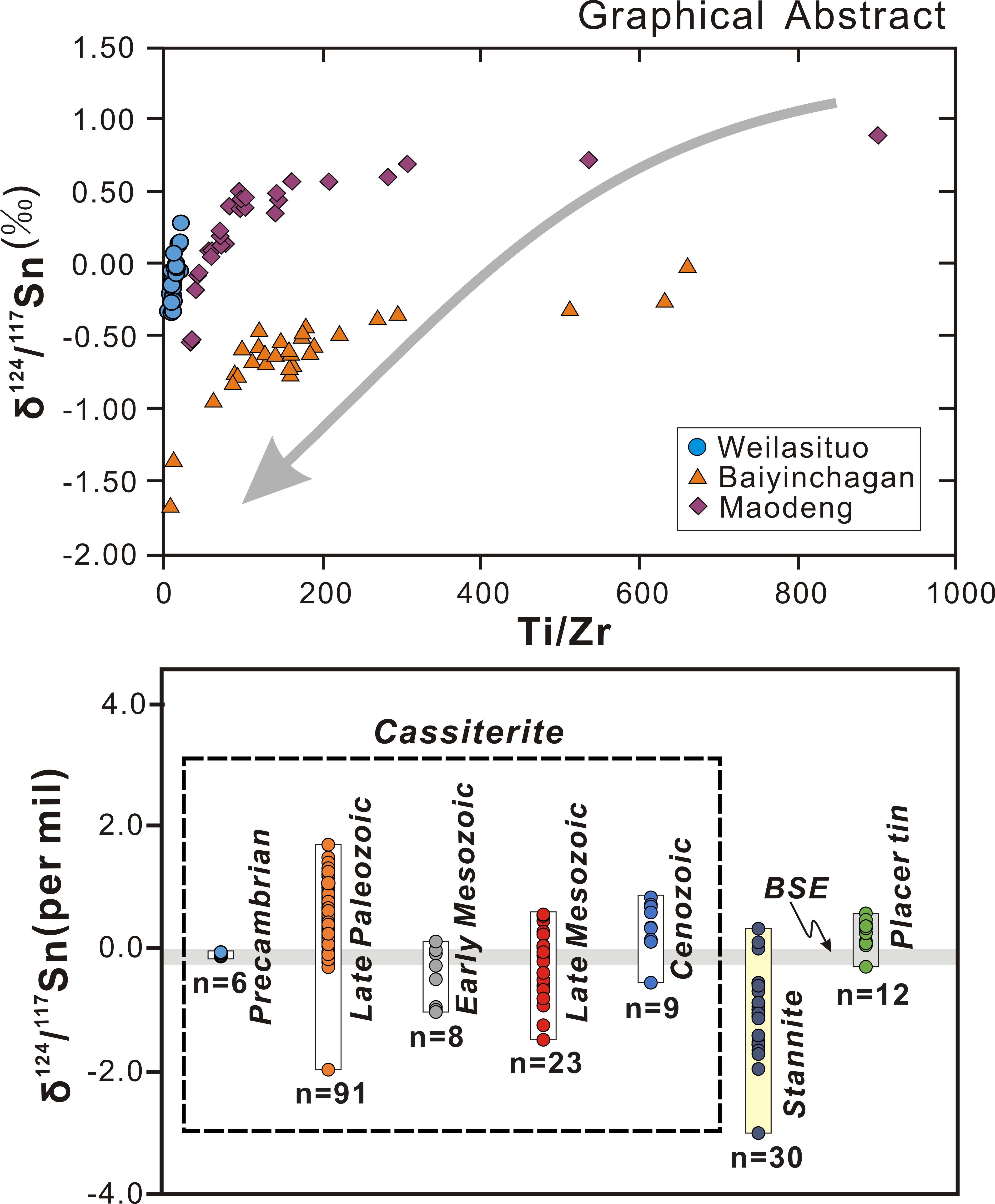
Nature and timing of Sn mineralization in southern Hunan, South China: Constraints from LA-ICP-MS cassiterite U-Pb geochronology and trace element composition
https://doi.org/10.2138/am-2022-8823Ren et al. present three points of interest related to Sn mineralization the rocks they studied: (1) The chronology of cassiterite defines three stages of magmatic mineralization in tin deposits in southern Hunan; (2) Cassiterite compositions are a physicochemical pathfinder of magmatic-hydrothermal processes; and (3) Multiple types of cassiterite need to be studied to establish an idealized tin mineralization model.
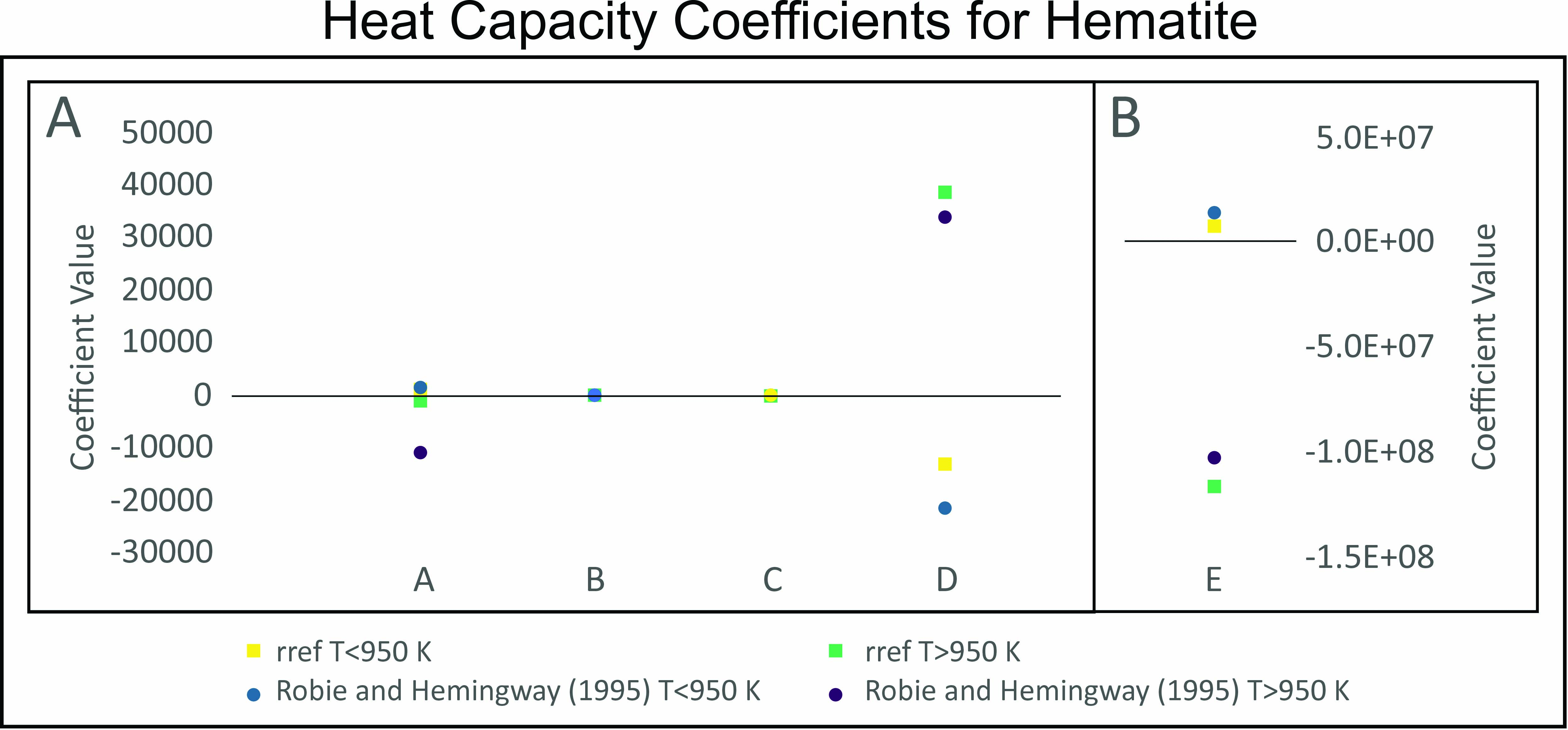
A simple method for obtaining heat capacity coefficients of minerals
https://doi.org/10.2138/am-2023-9109Bowman et al. developed a simple method for obtaining the heat capacity coefficients of minerals. Determination of heat capacity coefficients through this direct calculation yields comparable results to those obtained from conventional methods. Accurate heat capacity measurements can be utilized to calculate geothermal gradients in active basins, geothermometry, and study geodynamic transformations in tectonically active regions.
Molybdenum isotopic fractionation in the Panzhihua mafic layered intrusion in the Emeishan large igneous province, southwest China
https://doi.org/10.2138/am-2023-9154The large Mo isotopic fractionations between different geological reservoirs make this isotopic system a potential useful tool for constraining the origins of magmatism. However, the effect of magmatic differentiation on Mo isotopes is still controversial. Zhang et al. focused on the effect of the fractional crystallization of Fe-Ti oxides on Mo isotope fractionation. They obtained Mo isotope data for the Panzhihua gabbroic intrusion and mineral separates, which exhibit large Mo isotopic fractionations with δ98/95Mo values as follows: magnetite < clinopyroxene < ilmenite < plagioclase. Iron-Ti oxides have Mo contents that are one order of magnitude higher than those of clinopyroxene and plagioclase. Rayleigh fractionation modeling shows that the removal of magnetite and ilmenite results in significant Mo isotopic fractionation in the residual magma. Due to the low Mo contents of clinopyroxene and plagioclase, Mo isotopes are not significantly fractionated by the removal of these minerals. Their study highlights that fractionation of Fe-Ti oxides can cause considerable Mo isotopic fractionation; consequently, caution is needed when using Mo isotopes to infer magma origins.
February 2024
Crystal chemistry and thermodynamic properties of zircon structure-type materials
https://doi.org/10.2138/am-2022-8632Strzelecki et al. evaluated recent advances in the crystal chemistry and thermodynamic properties of zircon structure-type materials, identifying several knowledge gaps. The review will help researchers develop a fundamental understanding of zircon-type minerals and promote using zircon-type materials for such applications as actinide waste forms and environmental barrier coatings in aeronautical engineering.
Thermal and combined high-temperature and high-pressure behavior of a natural intermediate scapolite
https://doi.org/10.2138/am-2023-8962Scapolites, which are intermediate between the marialite and meionite end members, usually show a primitive symmetry. Lotti et al. reported an intermediate scapolite with an unusual I4/m symmetry, and they investigated it at high-T and combined high-T and high-P with in situ diffraction techniques. Elastic behavior and structural deformation mechanisms have been described, and a phase transition observed. Implications on the potential metastable preservation of I4/m scapolites are also discussed.
Crystal structure, hydrogen bonding, and high-pressure behavior of the hydroxide perovskite MgSi(OH)6: A phase relevant to deep subduction of hydrated oceanic crust.
https://doi.org/10.2138/am-2022-8889Welch et al. reported for the first time the full crystal structure, including hydrogen bonding, of the high-pressure synthetic hydroxide perovskite MgSi(OH)6. This phase is considered to likely play a significant role in the hosting and transfer of H2O in the Earth's deep mantle at cold subduction zones. Experiments to 8 GPa described here provide insights into the structural behavior of MgSi(OH)6 and provide an essential basis for future experimental and computational studies of its stability.
Equilibrium Sn isotope fractionation between aqueous Sn and Sn-bearing minerals: Constrained by first-principles calculations
https://doi.org/10.2138/am-2022-8804Sun et al. performed first-principles calculations on the equilibrium Sn isotopic fractionation between aqueous Sn species and minerals. Major outcomes are: (1) The Sn isotope geothermometers of cassiterite-stannite and malayaite-stannite have been established; (2) The controlling factors of Sn isotope fractionation between different aqueous Sn complexes are revealed; (3) The precipitation of aqueous Sn complexes into various tin minerals (malayaite, cassiterite and stannite) lead to the fractionation of Sn isotope in different degrees and directions. The calculated results are essential for further application of Sn isotope in various Sn-involved geological processes.
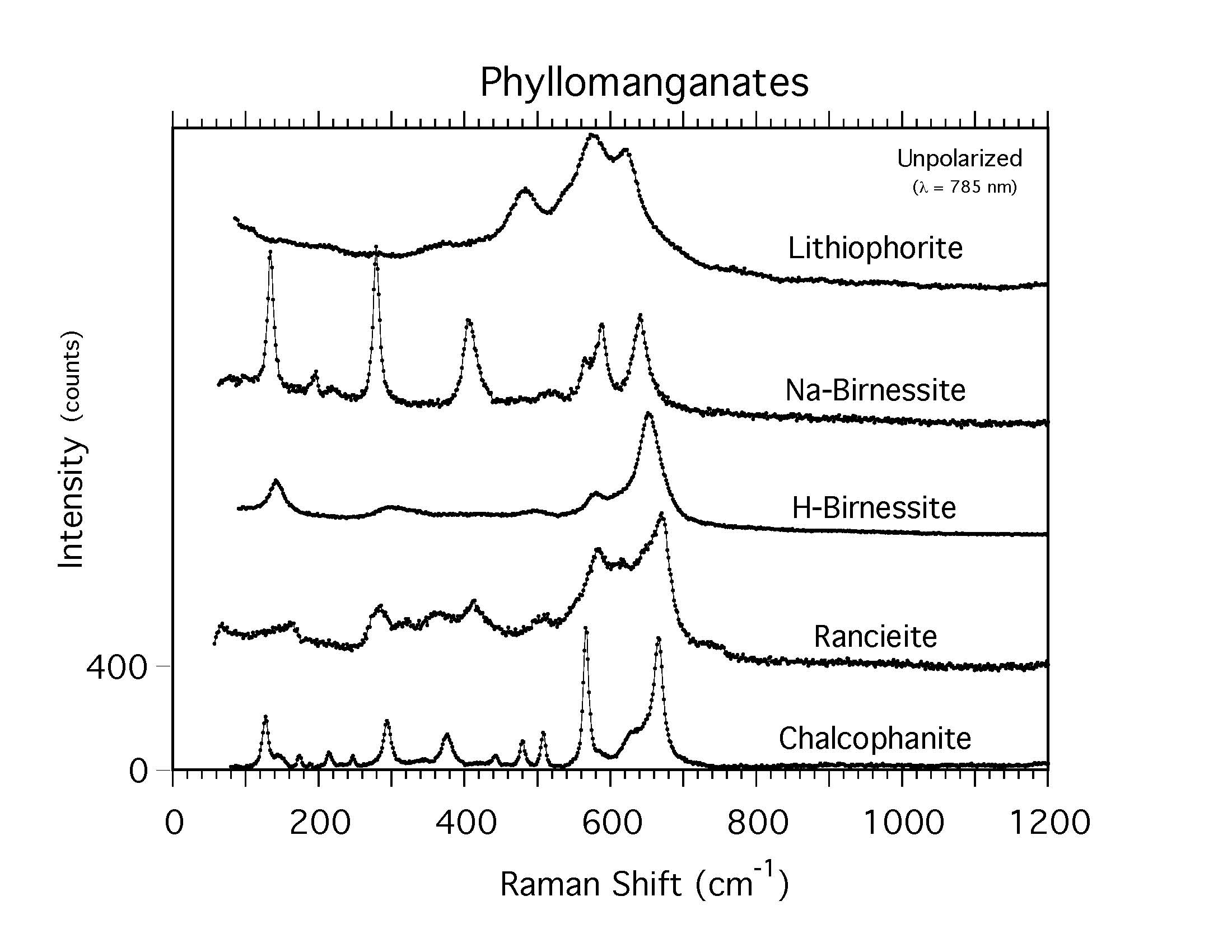
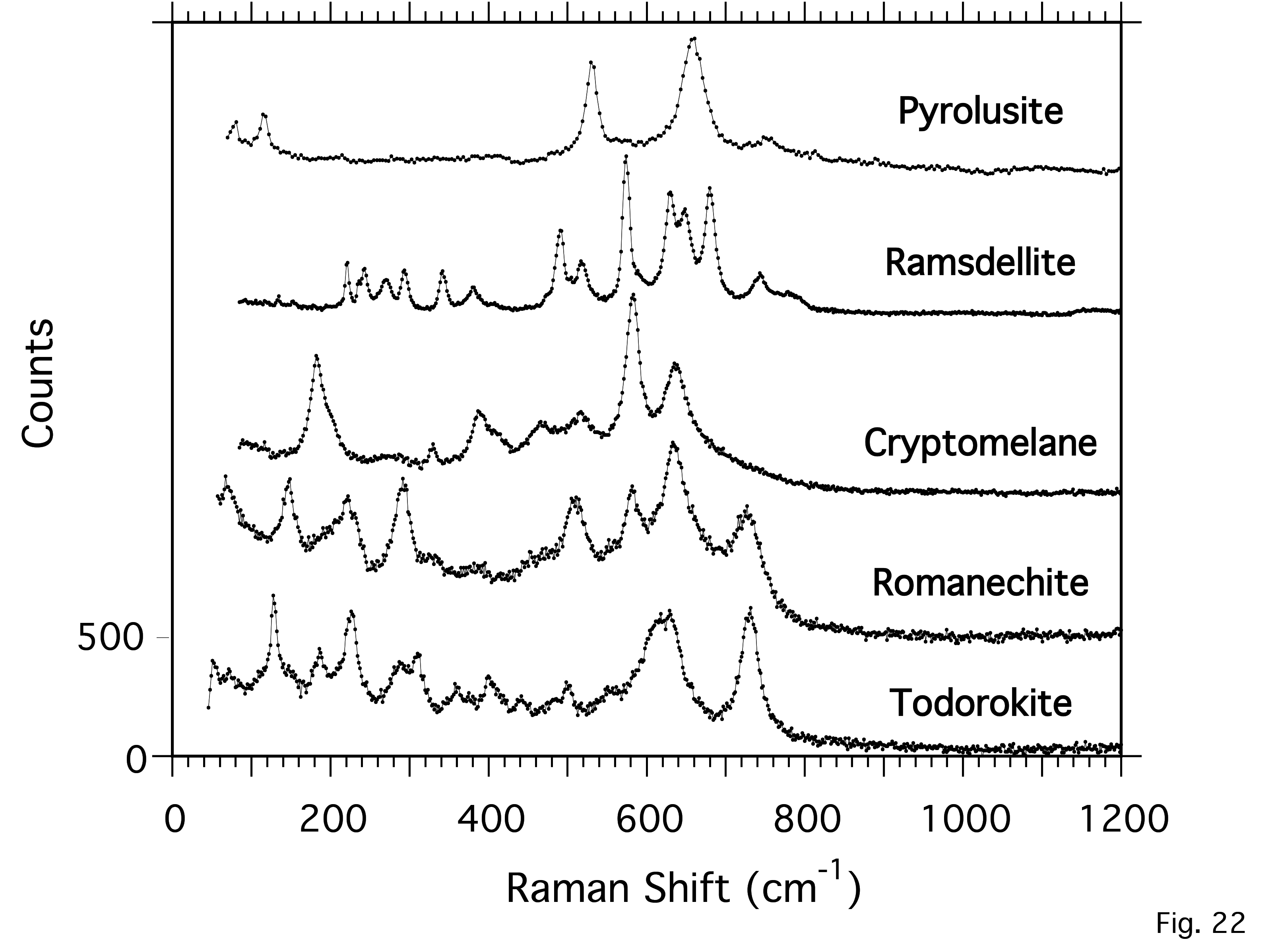
Raman Spectroscopic Investigation of Selected Natural Uranyl Sulfate Minerals
https://doi.org/10.2138/am-2023-8932Spano et al. reported Raman spectra of 18 uranyl sulfate minerals. To better understand underlying structural and chemical features that give rise to spectroscopic observables, they related differences in structural topology, charge balancing cations, and locality of origin to features observed in the Raman spectra of selected natural uranyl sulfates.
Modified magnetite and hydrothermal apatite in banded iron-formations and its implications for high-grade Fe mineralization during retrogressive metamorphism
https://doi.org/10.2138/am-2022-8525Magnetite (an abundant and widespread oxide mineral) and apatite (a common tracer mineral) in banded iron-formations (BIFs) are ideal minerals to study the hydrothermal and metamorphic processes. Shi et al. investigated the geochemical composition of modified magnetite and hydrothermal apatite, and in situ U-Pb geochronology on apatite from the Huogezhuang BIF-hosted Fe deposit, northeastern China. The chemical compositions recorded in modified magnetite and hydrothermal apatite from BIFs provide essential insights into the supernormal enrichment of iron during retrogressive metamorphism of BIFs.
Apatite trace element composition as an indicator of ore deposit types: a machine learning approach
https://doi.org/10.2138/am-2022-8805Qiu et al. used machine learning to link the concentration variability of trace elements in apatite to the five most important types of ore deposits. However, machine-learning approaches are often considered a black box in that they may be too complex to be easily interpretable by humans. Their application of the machine-learning model, however, revealed that the trace elements thorium, uranium, europium, and neodymium in apatite are the most distinctive elements for the discrimination of different types of ore deposits and, thus, the results are easily applicable.
Identifying serpentine minerals by their chemical compositions with machine learning
https://doi.org/10.2138/am-2022-8688Ji et al. introduce machine-learning algorithms called XGBoost and k-means to classify and cluster serpentine minerals based on the analysis of chemical composition datasets. As a complement to traditional geochemical methods, the machine-learning models are more effective in determining serpentine minerals. They also use a k-means model to show that the tectonic environment in which serpentine minerals form correlates with their chemical composition. Their models can be used to constrain the mass transfer and surrounding environments during the subduction of hydrated oceanic crust.
Crystal habit (tracht) of groundmass pyroxene crystals recorded magma ascent paths during the 2011 Shinmoedake eruption
https://doi.org/10.2138/am-2022-8765Okumura et al. analyzed the shape variations due to combination of crystallographic faces (i.e., tracht) of groundmass pyroxene crystals in pumices from the 2011 eruption of Shinmoedake volcano, Japan. They also acquired tracht-specific crystal size distributions (CSDs) by scanning electron microscopy to obtain a more comprehensive view of the crystallization kinetics. They discuss the mechanism of the pyroxene tracht change and conclude that the tracht change resulted from a large degree of effective undercooling due to rapid decompression in the shallow conduit. Syneruptive magma ascent paths affect eruptive style, and the crystallization kinetics of groundmass crystals as small as nanolites provide clues to the conduit mechanisms controlling transitions in eruptive style. The findings reveal magma dynamics from a different perspective than conventional textures such as crystal number density and crystallinity.
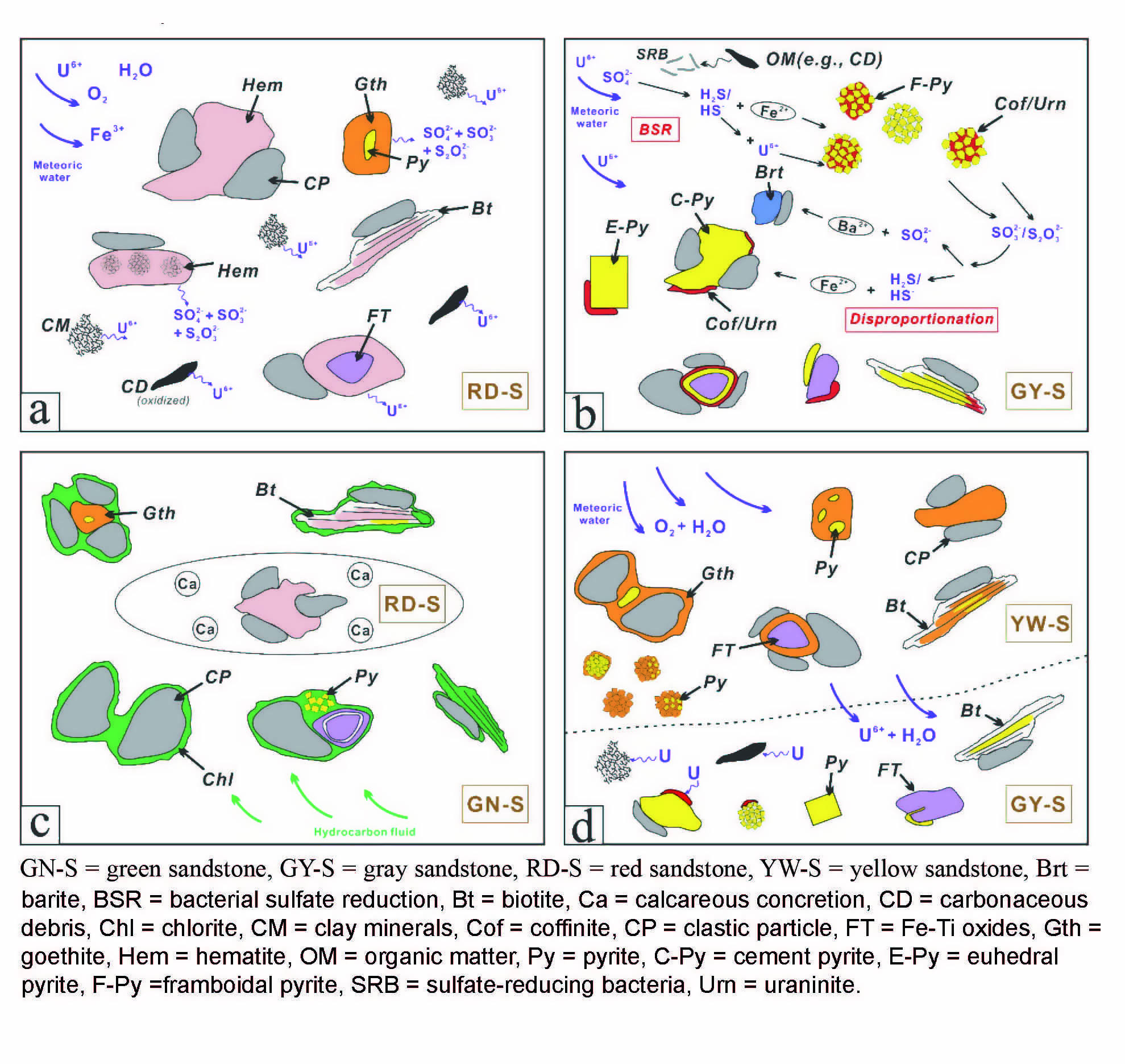
Reconstructing diagenetic mineral reactions from silicified horizons of the Paleoproterozoic Biwabik Iron Formation, Minnesota
https://doi.org/10.2138/am-2022-8776Duncanson et al. present petrographic observations, SEM, EMPA, and Raman spectroscopy from iron mineral phases preserved within silica-cemented horizons of the ~1.9 Ga Biwabik Iron Formation (Minnesota, USA) to constrain texturally early iron formation mineralogy from this crucial post-GOE (Great Oxidation Event) interval. Based on textural relationships, the iron silicate greenalite is identified as the earliest-forming iron silicate mineral preserved within silica-cemented horizons. The Mg- and Al-rich iron silicates chamosite and stilpnomelane are preserved proximal to fine-grained, non-silicified horizons, suggesting local geochemical exchange during early diagenesis. The presence of well-preserved, early-forming silicates containing predominantly ferrous iron may indicate reducing conditions at the sediment-water interface during deposition of the Biwabik Iron Formation. Future studies using iron silicate mineralogy as seawater geochemistry proxies should consider preservation by silica cementation, in addition to the effects of local geochemical exchange during diagenesis.
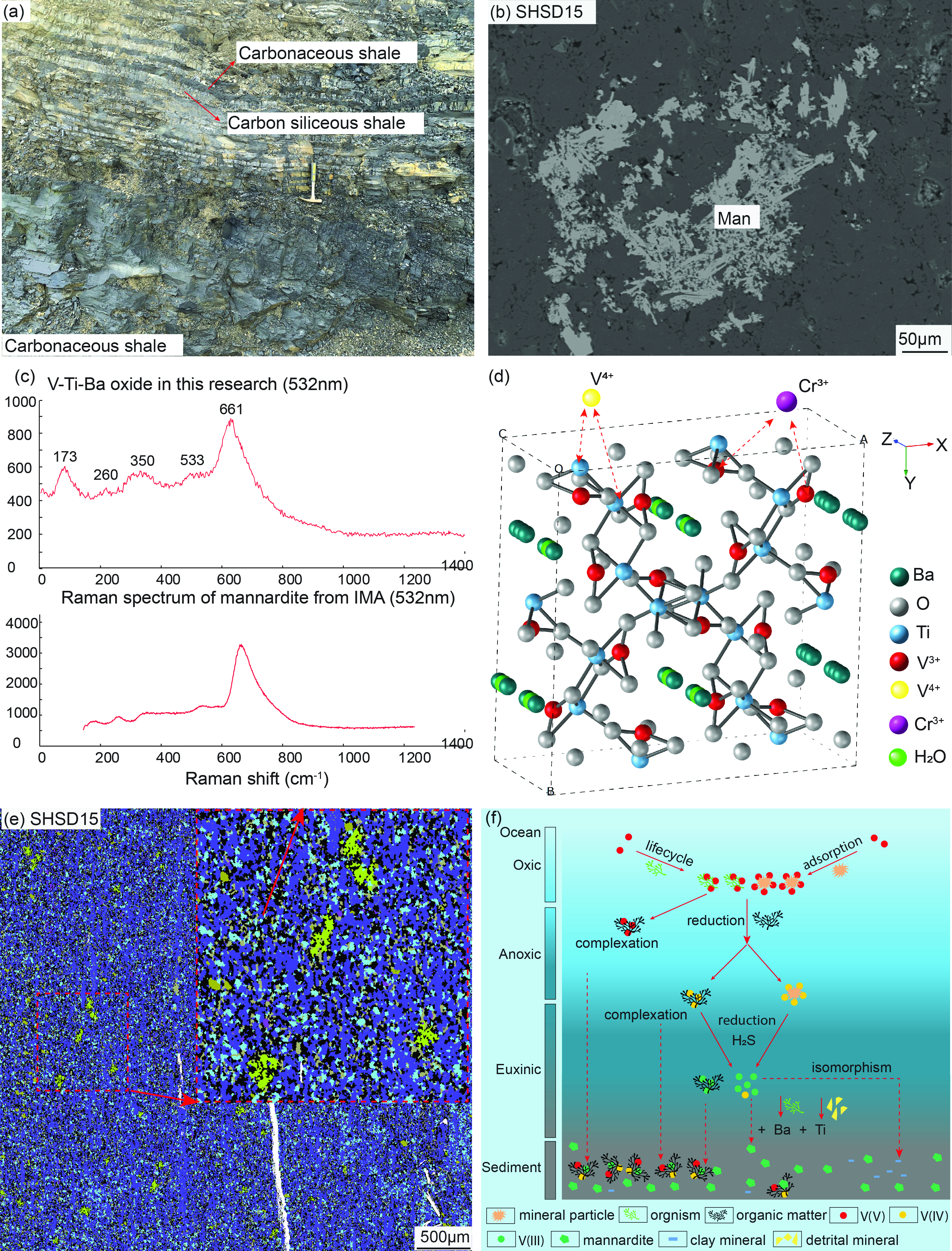
Mannardite as the main vanadium-hosting mineral in black shale-hosted vanadium deposits, South China
https://doi.org/10.2138/am-2022-8903Yang et al. identified mannardite in the black shale-hosted vanadium deposits, which is the main vanadium-hosting mineral and provides 12.32% - 44.06% vanadium. The pentavalent vanadium species in seawater accumulated through organism activities may be reduced into quadrivalent vanadium by organic matter and then into trivalent vanadium species by H2S. Mannardite may be precipitated under the reductive condition with sufficient trivalent vanadium species, titanium (Ti) and biogenic barium (Ba). The results put forward new cognition on the occurrence states of vanadium, which shows mineralogical significance on understanding the metallogenic mechanism as well as beneficiation of vanadium in the black shale. Based on the proportion of vanadium from the mannardite (average of 24.95%), mannardite in the black shale-hosted vanadium deposits preserves a total of 19.6% vanadium in the world.
Molybdenite-bearing vugs in microgranite in the preissac pluton, Quebec, Canada: Relicts of aqueous fluid pockets?
https://doi.org/10.2138/am-2022-8770Mulja and Williams-Jones demonstrated that (1) there exists rare occurrence of molybdenite-bearing miarolitic cavities in highly evolved fine-grained monzogranite dikes with REE of 17.2, EuCN of 0.27 and Eu/Eu* of 0.24; (2) field, mineral-chemical, whole-rock composition, and stable isotope (O and S) data indicate a magmatic-hydrothermal origin for the cavities, molybdenite and other associated phases; (3) the cavities represent relicts of vapors (gas-fluid mixtures) that exsolved from an ascending felsic magma through fractures where it quenched as fine-grained monzogranite dikes; and (4) molybdenum that partitioned into the vapor phase precipitated as molybdenite in the cavities, where, based on volumetric relationships and thermodynamic data, it attained a maximum concentration of 7800 ppm.
The equilibrium boundary of the reaction Mg3Al2Si3O12 + 3CO2 = Al2SiO5 + 2SiO2 + 3MgCO3 at 3-6 GPa
https://doi.org/10.2138/am-2022-8696The reaction between garnet and CO2 fluid is of interest because it constrains the stability of CO2 fluid in eclogites, whose minerals were found in the CO2-bearing diamonds. Vinogradova et al. determined the equilibrium boundary for the reaction Mg3Al2Si3O12 (Prp) + 3CO2 (F) = 3MgCO3 (Mgs) + Al2SiO5 (Ky) + 2SiO2 (Coe/Qz) over the pressure interval 3-6 GPa using a multianvil press. The established boundary crosses the graphite-to-diamond transition curve at 4.4 GPa and 1060 °C. Thus, the assemblage garnet + CO2 fluid is stable in the diamond stability field under P-T conditions of the continental geotherm with a heat flow of 40 mW/m2.
Comment on Lee et al. (2022) “Reexamination of the structure of opal-A: A combined study of synchrotron X-ray diffraction and pair distribution function analysis” —Concerning opal
https://doi.org/10.2138/am-2022-8863Reply: On "Reexamination of the structure of opal-A: A combined study of synchrotron X-ray diffraction and pair distribution function analysis"—Reply to de Jong
https://doi.org/10.2138/am-2023-9216January 2024
Highlights and Breakthroughs: The search for a universal law of crystal growth: The law of proportionate effect?
https://doi.org/10.2138/am-2023-9182Crystal growth according to the law of proportionate effect
https://doi.org/10.2138/am-2022-8851Almost all solids are composed of crystals. Therefore, an investigation of the material world should include an understanding of how crystals grow. This paper by Eberl summarizes a theory of crystal growth based on the processes necessary to produce the shapes of crystal size distributions (CSDs). The most common shape for CSDs is lognormal, a shape that is produced mathematically by the Law of Proportionate Effect. Application of this law leads to the conclusions that crystal growth is a stochastic process that proceeds by the incorporation of crystal fragments (rather than single atoms) during growth cycles. The shapes of CSDs are set during and immediately after nucleation, and reflect the degree of supersaturation present in solution. Then these shapes are preserved by proportionate growth as crystals grow larger. Experimental evidence indicates that advection rather than diffusion is the most important process that supplies nutrients necessary for growth. The results of this theory can be applied to the determination of geologic history from mineralogy, and for the control of CSD shapes during industrial crystallization.
Melt-mediated re-equilibration of zircon produced during meltdown of the Chernobyl reactor
https://doi.org/10.2138/am-2022-8824In this contribution, Fougerouse et al. determine a mechanism by which the composition of zircon crystals can be changed by interacting with a melt. The composition of zircon is without a doubt the most commonly used tool in the Earth sciences because zircons record the age when rocks formed and by which geological process. However, they show that zircon can be modified under conditions that were previously not considered.
High pressure behavior and structural transition of beryl-type johnkoivulaite, Cs(Be2B)Mg2Si6O18
https://doi.org/10.2138/am-2022-8908This contribution by Gatta et al. investigated the high-pressure behavior of the beryl-group mineral johnkoivulaite. This mineral experiences a P6/mcc to P-3c1 (second-order) phase transition on isothermal compression at the critical pressure of 4.13 GPa. The high-P polymorph shows a superstructure with a′ = a√3, c′ = c, according to P-3c1, and is almost twice as compressible as its low-P form. This behavior is unique for any beryl-derived structure and can be attributed to the high degree of freedom for atomic displacements in the superstructure. The reduced symmetry can also be understood as the effect of the driving mechanism of the transformation. The extra-framework Cs channel components counteract any lateral deformation of the channels parallel to [0001] within the microporous framework and, similar to pezzottaite, are responsible for maintaining the trigonal/hexagonal lattice metrics.
Subsolidus breakdown of armalcolite: Constraints on thermal effects during shock lithification of lunar regolith
https://doi.org/10.2138/am-2022-8790Lunar regolith breccias are precious samples that can be studied in the laboratory to unravel the formation and evolution history of the Moon. Properly obtaining scientific information from regolith breccias relies on the quantitative understanding of their thermal history. Strong shock lithification on the surface of the Moon is an important process that could have significant thermal effects. However, potential thermal effects of this process on lithic and mineral fragments have not yet been quantitatively constrained. Du et al. constrain the thermal effect during strong shock lithification by using the subsolidus breakdown reaction of armalcolite, which is an important temperature indicator. Their study indicates that the temperature rise in lithic clasts from strongly shock-lithified regolith breccias is heterogeneous, but could be high as at least 600 °C at the margin of lithic clasts close to the melt that formed during strong shock lithification. Such a temperature rise is also high enough to disturb the U-Pb systematics of apatite and paleomagnetic records of some magnetic minerals.
Melting and melt segregation processes controlling granitic melt composition
https://doi.org/10.2138/am-2022-8594Rb, Sr, and Ba abundances of granites result from the melting reactions and feldspar fractionation during melt segregation in the source. Yu et al. find that the REE and Nd isotope signatures of granite are determined by complex accessory mineral reactions in the source. Thus, the granite composition is controlled by both melting and melt segregation processes in source.
Magmatic degassing controlled the metal budget of the Axi epithermal gold deposit
https://doi.org/10.2138/am-2022-8610This contribution by Li et al., provides evidence for a linkage between mafic recharge, andesite formation, and epithermal gold mineralization. The bulk metal budget of an epithermal system is controlled primarily by nature of the magma. Gold mineralization is associated with repeated mafic magma injections. Silver-Cu-Pb-Zn-Sb enrichment is related to quiescent degassing of the felsic magma. The enrichment of fluid-immobile elements is caused in part by fluid-rock interaction.
Formation of mixed-layer sulfide-hydroxide minerals from the Tochilinite-Valleriite Group during experimental serpentinization of olivine
https://doi.org/10.2138/am-2022-8625Tochilinite-valleriite group (TVG) minerals are sparse components of serpentinites, but have the potential to provide useful constraints on the environmental conditions present when they formed (fluid composition, sulfur source, temperature, etc.). McCollom et al. report formation of the TVG minerals valleriite and haapalaite during experimental simulation of serpentinization, and discuss possible implications for natural serpentinites.
Two discrete gold mineralization events recorded by hydrothermal xenotime and monazite, Xiaoqinling gold district, central China
https://doi.org/10.2138/am-2022-8635This study by Jian et al. implies that the large-scale Jurassic mineralization event in eastern China, known for its porphyry Mo deposits, has also caused widespread gold mineralization in the Qinling-Dabie Orogen. The fact that a few Jurassic gold mineralization ages have been reported so far is due to the lack of suitable geochronometers to record Jurassic hydrothermal processes, which have been overprinted by the Early Cretaceous gold mineralization event. This study also presents a rare example of xenotime compositional alterations and resetting of U-Pb ages induced by low to moderate salinity carbono-aqueous fluids at low temperatures. Since low-to-moderate salinity carbono-aqueous fluids are commonly involved in the formation of lode gold deposits, it's crucial to examine xenotime textures and recognize potential alteration textures before carrying out isotopic dating of xenotime collected from these deposits. Without prior chemical and textural characterization, attempts to date such composite crystals could yield mixed age information and meaningless ages.
Formation of amphibole lamellae in mantle pyroxene by fluid-mediated metasomatism: A focal plane array FTIR study from the Carpathian-Pannonian region
https://doi.org/10.2138/am-2022-8662Amphibole, the most abundant hydrous mineral in the upper mantle, is considered to form by melt-rock interaction. However, its genesis may be ambiguous when present as lamellae in anhydrous mantle minerals (pyroxenes). Liptai et al. demonstrate that hyperspectral imaging facilitates calculating the volume proportion of the lamellae and the amount of water needed for its formation. This can be used to determine whether lamellae exsolution is a realistic scenario or if an external water source is needed.
Origin of gem-quality turquoise associated with quartz-barite veins in western Hubei Province, China: constraints from mineralogical, fluid inclusion and C-O-H isotopic data
https://doi.org/10.2138/am-2022-8643It has long been thought that gemstone turquoise is formed by supergene processes via cool meteoric water leaching the wall rocks of common black shales or by the supergene oxidation of primary sulfides in porphyry deposits. In this study, Li et al. report the black-slate-hosted, gem-quality turquoise deposits found in Shanxi and Hubei provinces of China were not formed by supergene processes, instead, they may have been formed by non-magmatic, hydrothermal processes at temperatures up to 400 °C. The responsible hydrothermal fluids are characterized by the coexistence of two unmixed fluids, in which one resembles a high-to-moderate temperature and low salinity aqueous fluid produced by regional metamorphism, and the other is a CH4-N2 fluid formed by interaction with black slates. From the mineralogical point of view, the high-quality bluish-green gem turquoise tends to evolve to turquoise-planerite solid series as the fluid-rock interaction lessened and are characterized by textual and geochemical variations.
The 450 nm (2.8 eV) cathodoluminescence emission in quartz and its relation to structural defects and Ti contents
https://doi.org/10.2138/am-2022-8884The present study by Götze et al. was aimed to elucidate the origin of the common blue ~450 nm (2.8 eV) CL emission in quartz and to find possible quantitative relations to the structural incorporation of Ti. The investigation of quartz of different origin and containing different concentrations of Ti revealed that both structural defects not related to trace elements (self-trapped exciton, STE) and also incorporation of Ti4+ into the quartz structure can activate the blue CL emission at the same wavelength position. The common activation of the ~450 nm luminescence emission in quartz by an intrinsic defect (STE related to oxygen vacancy) and structurally incorporated Ti4+ as an additional activator prevents a serious quantitative evaluation of the CL signal. This finding is especially interesting with regard to correlations of trace-element contents and CL intensities, in particular with panchromatic SEM-CL investigations. Their results show that blue CL emission at ~450 nm can even appear in high-purity quartz with very low trace element contents.
Correlation between Hi-index and stacking order-disorder in kaolinite
https://doi.org/10.2138/am-2022-8830Rao et al. employed a combination of experiments, first-principles simulation, and structure refinement analysis to prove that the Hi is directly related to the stacking order-disorder in kaolinite. They inferred that the stacking order-disorder in kaolinite will directly affect its lattice energy via experiments and this viewpoint was confirmed by first-principles simulations. Based on this, the stacking order-disorder in kaolinite is classified. Finally, a new crystallinity index was established, and it can be used independently or as a supplement to Hi-index.
Structure and titanium distribution of feiite characterized using synchrotron single-crystal X-ray diffraction techniques
https://doi.org/10.2138/am-2022-8750In this study, Zurkowski et al. applied single-crystal X-ray diffraction techniques to assess the structure and titanium distribution in the recently discovered mineral, Feiite (Fe3TiO5). This Fe-, Ti-oxide is particularly interesting for its potential as an indicator of oxygen fugacity and high pressure-temperature processing of Martian crustal and mantle material. They report a convergent crystal-structure model for three grains of a feiite-Fe4O5 solid solution synthesized at 12 GPa and 1200 °C. Their structural refinements indicate a CaFe3O4 structure (Cmcm space group), and limited titanium solubility into the structure at these synthesis conditions. By comparing their measured average bond lengths to optimized Ti4+- O , Fe2+- O, and Fe3+- O bond lengths, they also estimate the ferric and ferrous iron contents of each site. The Ti and Fe3+ content constrained by their crystal-chemical analyses suggest that at least ~30% of the available iron must be ferric (i.e., Fe3+/Fetotal = 0.3) for the sample synthesized at 12 GPa and 1200 °C. Higher P-T conditions may be needed to from the endmember feiite (Fe2+3TiO5).
Enrichment of precious metals associated with chalcopyrite inclusions in sphalerite and pyrite
https://doi.org/10.2138/am-2022-8636Zhang et al. investigated the distribution and occurrence of precious metals in sphalerite and pyrite from the Maluntou epithermal gold deposit, southeastern China. The key findings include: (1) the disseminated gold and silver bearing nanoparticles are closely associated with micro/nano chalcopyrite inclusions in sphalerite; (2) two origins of chalcopyrite inclusions involve recrystallization-driven phase separation from parent chalcopyrite-sphalerite solid-solutions, and replacement of pre-existing pyrite by Cu-bearing fluids; (3) mixing and boiling during the evolution of ore-forming fluids are key processes for the abnormal enrichment of precious metals in sphalerite. Such information is critical for understanding possible mechanisms of precious metal enrichment of in sulfides and industry processing of refractory ores.
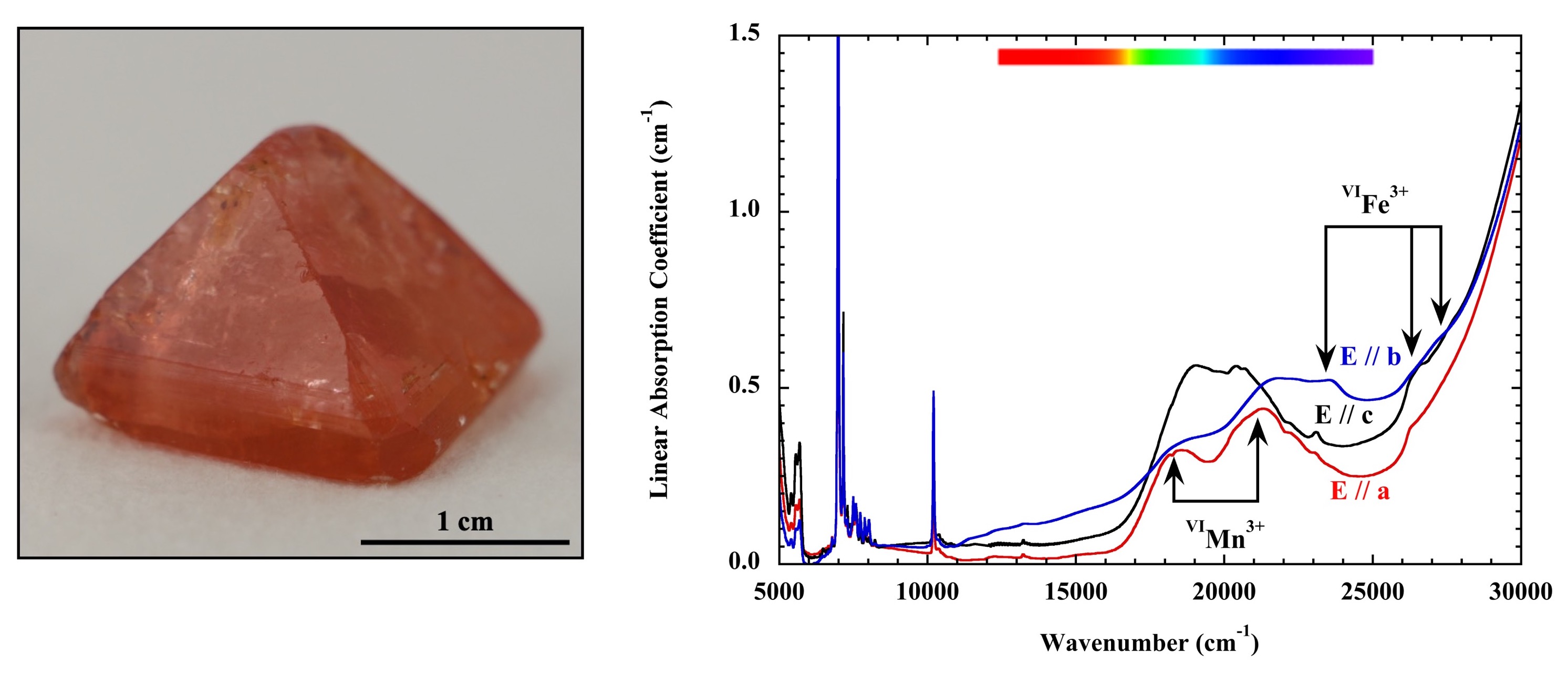
An UV/Vis/NIR optical absorption spectroscopic and color investigation of transition-metal-doped gahnite (ZnAl2O4 spinel) crystals grown by the flux method
https://doi.org/10.2138/am-2022-8708Synthetic flux-grown, end-member gahnite, ZnAl2O4, and a number of different colored crystals doped with transition metals including Ni, Cr, Co, Mn, and Fe were studied by Taran et al. with electron microprobe methods and UV/Vis/NIR single-crystal optical absorption spectroscopy. The authors measured and assigned the different electronic absorption features and analyzed the crystal colors using the experimental spectra and described them with the CIE 1931 color-space-chromaticity diagram.
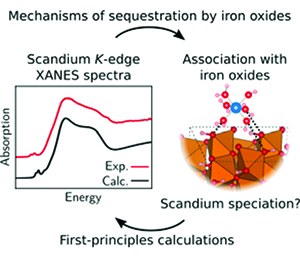
Understanding the unique geochemical behavior of Sc in the interaction with clay minerals
https://doi.org/10.2138/am-2023-8941Scandium can be incorporated into the lattice of clay minerals, whereas other rare earth elements (REEs) cannot. The microscopic structures, free energies, and hydrolysis of Sc3+ surface complexes determined in this study by Zhang et al. show that such unique behavior of Sc3+ originates from its high affinity to the vacancies on clay edges. The findings in this study highlight the importance of clay edge surfaces in REE enrichment and provide a physical basis for understanding the formation mechanism of Sc-rich clays. The structural and thermodynamic information presented in this study can be used to investigate the enrichment of REEs and develop efficient extraction techniques.
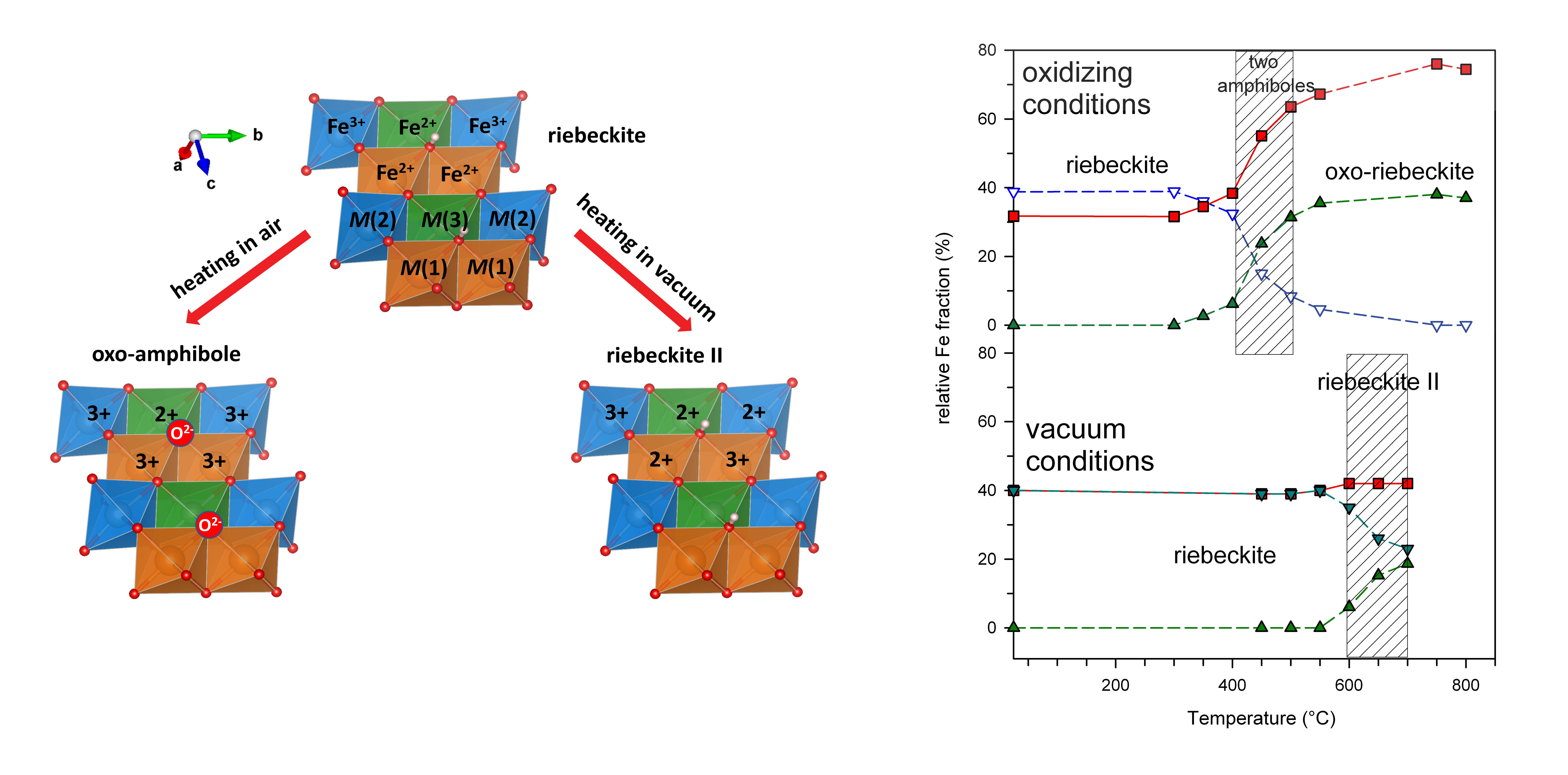
Scandian actinolite from Jordanów, Lower Silesia, Poland: Compositional evolution, crystal structure, and genetic implications
https://doi.org/10.2138/am-2022-8786The paper by Pieczka et al. on scandian actinolite from contact zones of a granitic pegmatite intruded into serpentinite in a quarry at Jordanów Ślaski in Lower Silesia, Poland, characterizes the second occurrence of the amphibole-supergroup mineral enriched in Sc. The occurrence is not connected with the presence of fluorine, considered as the strongest agent complexing Sc, because F is only detected in traces in titanite. It seems that in the case of Jordanów Ślaski, we may have a new type of geologic environment leading to the Sc enrichment, in addition to clinopyroxene-bearing, basic alkaline rocks, carbonatites, and laterite deposits. The authors discuss differences observed in the geologic environments mainly in relation to basic clinopyroxene-bearing rocks based on the avaiable geologic information related to rocks of the serpentinite Gogolow-Jordanów massif and Śleza ophiolite.
Characterizing a new type of nelsonite recognized in the Damiao anorthosite complex, North China Craton, with implications for the genesis of giant magmatic Fe-Ti oxide deposits
https://doi.org/10.2138/am-2022-8501Li et al. recognized a new type of nelsonite in the Proterozoic Damiao anorthosite complex, which is distinct from the two known types of nelsonite from the same complex. Through the comprehensive comparative study taking into consideration of all three types of nelsonites found in the Damiao anorthosite complex, they show that giant magmatic Fe-Ti oxide orebodies can form by the combined processes of liquid immiscibility and fractional crystallization.
Genesis of mesozoic high-Mg dioritic rocks from the eastern North China Craton: Implications for the evolution of continental lithosphere
https://doi.org/10.2138/am-2022-8755Jinling high-Mg diorites (HMDs), a Mesozoic intra-plate intrusion of the eastern North China Craton, were derived from partial melting of hydrous mantle peridotites, but are not associated with the interaction of slab-derived melt with mantle peridotite. This study by Guo et al. highlights that Pre-Cenozoic HMAs/HMDs, such as Archean TTG and sanukitoids, could be derived from partial melting of hydrous mantle peridotites in continental interiors, and thus might not always be related with the onset of plate subduction.
SEM and FIB-TEM analyses on nanoparticulate arsenian pyrite: implications for Au enrichment in the Carlin-type giant Lannigou gold deposit in SW China
https://doi.org/10.2138/am-2022-8550A significant difference in Au concentration between the ore fluid and the coexisting arsenian gold-bearing pyrite in Carlin-type gold deposits requires incorporation of Au into arsenian pyrite during mineralization. Based on previous NanoSIMS Au mapping, nano-phase pyrites in a Au-bearing pyrite rim were revealed by in-situ FIB-TEM and SEI analysis. Yan et al. proposed a model for attaching and aggregating growth process of arsenian pyrite rims and conclude that the numerous nano-particulate pyrites are a key constraint on effective Au chemisorption to form a giant Carlin-type gold deposit. In addition, the mechanisms of nano-particulate pyrites and nano-gold formation might provide new insights into mineralization process of other gold deposits and Au metallurgical recovery from refractory ores.
December 2023
Gas-mediated trace element incorporation into rhyolite-hosted topaz: A synchrotron microbeam XAS study
https://doi.org/10.2138/am-2022-8417Schaub et al. conducted synchrotron microbeam XAS analysis on picogram quantities of As, Mn, Ga, Ge, Fe, and Ti in topaz crystals from vesicular topaz rhyolite (Thomas Range, Utah) and obtained information on valence state, site assignments, and structural accommodation of these substituents. This information, coupled with trace-element abundances, provides new insights into the nature of the high-temperature magmatic gas involved in topaz formation.
Evidence for abundant organic matter in a Neoarchean banded iron formation
https://doi.org/10.2138/am-2023-8927Peng et al. investigated the mineral associations, major element concentrations, total carbon contents and carbon isotope compositions of a set of BIF samples with highly variable iron and TOC contents from the late Neoarchean Zhalanzhangzi BIF, China. The results show that graphite grains with crystallization temperatures comparable to that predicted for the regional metamorphic grade are widely distributed despite highly variable iron and total organic carbon contents. The graphite is interpreted to represent the metamorphosed product of syngenetic biomass based on its co-occurrence with apatite rosettes and negative bulk rock δ13C organic values. The close association of graphite with iron-bearing phases indicates that ferric (oxyhydr)oxides may have exerted a first-order control on the abundance of OM.
A dunite fragment in meteorite northwest Africa (NWA) 11421: A piece of the moon's mantle
https://doi.org/10.2138/am-2022-8911Treiman and Semprich describe a centimeter-sized fragment of dunite, the first recognized fragment of Moon mantle material, in the lunar highlands breccia meteorite Northwest Africa. From electron microscope data on dunite, they derive inferences about the bulk composition of the lunar mantle, processes related to the Lunar Magma Ocean and gravitational overturn of its cumulate rocks, and the present-day thermal structure of the Moon's mantle.
Electrical properties of iron sulfide-bearing dunite under pressure: Effect of temperature, composition, and annealing time
https://doi.org/10.2138/am-2023-9054Tauber et al. report electrical measurements of a mantle xenolith with varying amounts of metal sulfide compounds over a wide frequency range while under high pressure and temperature. Samples annealing at 1023 K is found to significantly affect the electrical response at temperatures up to ~1100 K. Iron-sulfur compounds strongly increase bulk conductivity at or above 6.5%. At this threshold, the molten iron-sulfur network is fragile. Samples with added Ni or Fe metal show lowered conductivities relative to the dunite host. This surprising behavior is likely caused by a lowered oxygen fugacity.
Hydrogen bond symmetrization and high-spin to low-spin transition of ε-FeOOH at the pressure of Earth's lower mantle
https://doi.org/10.2138/am-2022-8839Insixiengmay and Stixrude conducted density functional theory (DFT) calculations to study ε-FeOOH, the iron-bearing end-member of phase H, which is the dominant hydrous phase at lower mantle conditions. This phase exhibits rich behavior and serves as a model system for understanding two transitions that occur in a variety of Earth materials at high pressure: hydrogen bond symmetrization and a high-spin to low-spin transition. The authors examine the change in physical properties across each transition and explore implications for their possible effects on geophysical probes by looking at changes in seismic wave velocity and electrical conductivity.
CURIES: Compendium of uranium Raman and infrared experimental spectra
https://doi.org/10.2138/am-2022-8738No single database of Raman and IR spectra exists for uranium materials exclusively, and no large-scale summary of other important signatures originating from structural features beyond the axial U-O bond and other U-O moieties has been tabulated. Spano et al. provide new insights into Raman and infrared spectral features that are indicative of the underlying structural attributes from which they originate. The authors perform an extensive survey of all available experimental Raman and infrared spectra for supergene uranium minerals and employ multivariate statistical analyses to determine spectroscopic features that are common to groups of uranyl minerals based on secondary chemistry.
S2- and S3-radicals and the S42- polysulfide ion in lazurite, haüyne, and synthetic ultramarine blue revealed by resonance Raman spectroscopy
https://doi.org/10.2138/am-2022-8655Farsang et al. demonstrated the power of resonance Raman spectroscopy in search for sulfur species trapped in the cages of feldspathoids. The S42- polysulfide ion is reported in lazurite and haüyne for the first time. The results have implications in a broad range of fields, e.g., chemistry (identifying trace amounts of S-species), optoelectronics (entrapping S42- into sodalites/zeolites), and arts (identifying the provenance of natural ultramarines).
Effect of faceting on olivine wetting properties
https://doi.org/10.2138/am-2022-8808Huang et al. evaluated facet-bearing dihedral angles and their proportions in olivine–multicomponent aqueous fluid systems. The results show that: (1) faceting produces larger dihedral angles than those with curved interfaces in the olivine-fluid system; (2) grain boundary planes are frequently composed of low-index planes, and the facet-facet angles are explained by olivine crystallography; and (3) the facet proportion is consistent with electrical conductivity data; faceting effects could be magnified under sheared mantle.
The obscuring effect of magma recharge on the connection of volcanic-plutonic rocks
https://doi.org/10.2138/am-2022-8642The plutonic and volcanic realms are connected through the extraction of crystal-poor rhyolites from batholithic crystal mushes, whereas big data analyses of global volcanic and plutonic rocks reveal little evidence for significant segregation of liquids from plutons. Zhao et al. explore the possibility that magma recharge obscures the records of liquid-crystal segregation and remind us that big data analysis should be done with caution to avoid biased results.
In-situ study of microstructures induced by the olivine to wadsleyite transformation at conditions of the 410 km depth discontinuity
https://doi.org/10.2138/am-2022-8731Olivine transforms to wadsleyite at the conditions of 410 km depth in the Earth's mantle. How does this transformation affect microstructures and grain orientations? Ledoux et al. used in-situ experiments and followed the orientations of individual crystallites as the transformation proceeded. They do not observe relationships between the parent olivine and daughter wadsleyite phases and hence conclude that, in the Earth's mantle, microstructures should be reset below the 410 km depth discontinuity.
Effect of pre-existing crystals and melt homogeneity on the decompression-induced crystallization of hydrous rhyodacite magma
https://doi.org/10.2138/am-2022-8723Magma crystallization controls its rheology and volcanic eruption. Matsumoto et al. investigated the effect of pre-existing crystals and melt homogeneity on magma crystallization during decompression. The results indicate that the injection of high-temperature magma into the reservoir that changes pre-existing crystals and melt homogeneity may control crystal texture formed during magma ascent and volcanic eruption.
Origin of clinopyroxene-ilmenite symplectites in mafic granulites from eastern parts of the Chotanagpur granite gneissic complex, East Indian shield
https://doi.org/10.2138/am-2022-8715Chatterjee et al. report a rare occurrence of rod-like intergrowths of clinopyroxene-ilmenite that variably replace titanite in a suite of high-pressure mafic granulites from the Chotanagpur Granite Gneissic Complex, Eastern Indian Shield. The authors have modeled the reaction texture of clinopyroxene-ilmenite symplectite and deduced the physical condition of metamorphism. The clinopyroxene-ilmenite symplectites formed at the expense of garnet and titanite during steep near isothermal decompression (from ~13 kbar to ~7 kbar, at ~800°C), in the presence of fluid phase. It is also demonstrated that relative to Fe, Mg, and Ca, Ti was less mobile and that the mobility of Ti was restricted within the confines of the titanite being replaced by the clinopyroxene-ilmenite symplectite. Limited mobility of Ti even in the presence of presumed F-Cl bearing fluids or melt has important implications on the Ti mobility in the deep continental crust.
Single-crystal analysis of La-doped pyromorphite (Pb5(PO4)3Cl)
https://doi.org/10.2138/am-2022-8664Sordyl et al. synthesized two La-doped pyromorphite analogs (Pb5(PO4)3Cl) and two control samples (with the absence of La) from aqueous solutions at 25°C. Na+ and K+ were used as charge compensating ions to facilitate the incorporation of trivalent REE cations (La3+ + Na+ 2Pb2+ and La3+ + K+ 2Pb2+). The extent of La substitution, the position of La in the structure and the induced structural changes were determined using single-crystal X-ray diffraction. The results have applications in both fundamental understanding of apatite structure and environmental mineralogy (mineral-water interactions). Moreover, the results extend our knowledge of geofunctional materials that may be useful in future applications of Pb-apatite in REE recovery.
Crystal structure of calcium-ferrite type NaAlSiO4 up to 45 GPa
https://doi.org/10.2138/am-2022-8432Alkali-rich aluminous high-pressure phases including calcium-ferrite (CF) type NaAlSiO4 in MORB composition are thought to constitute ~20% by volume in the lower mantle. Qin et al. investigated the Fe-free and Fe-bearing CF phases using single-crystal synchrotron X-ray diffraction and Raman spectroscopy coupled with diamond anvil cells up to 45 GPa. The Fe-free and Fe-bearing CF samples display similar compressibility and roughly 2% more compressible than the CF-type MgAl2O4. Compared to jadeite, the ability of CF-type phases to contain incompatible elements such as Na up to lower mantle pressures requires the larger and less compressible NaO8 polyhedra. These data provide new knowledge on how large alkali metals can be hosted in aluminosilicate framework structures with stability well into the lowermost mantle.
Revision of the CaMgSi2O6–CO2 P-T phase diagram at 3-6 GPa
https://doi.org/10.2138/am-2022-8588Reactions involving CO2 and carbonates are the key factors determining the magma generation in the upper mantle and the deep carbon cycle. The CaO-MgO-SiO2-CO2 system is important for studying these reactions. Shatskiy et al. investigated the impact of carbonatization on the mafic suit solidus (the system diopside-CO2) in the range of 3–6 GPa and 850–1500 °C. They found that above 4.5 GPa, the liquidus reaction involving clinopyroxene and CO2 sweeps down through 350 °C via a pressure maximum near 5.3 GPa to meet the invariant point at 4.5 GPa. The shape of the solidus resembles that of lherzolite–CO2 but shifted by 2 GPa to higher pressures.
November 2023
Passive carbon sequestration associated with wollastonite mining, Adirondack Mountains, New York
https://doi.org/10.2138/am-2022-8622Carbon sequestration has the potential to help lower atmospheric CO2, and carbon mineralization (locking C into minerals) could be used to capture carbon in many industrial materials, such as concrete, slag, and mine wastes. In this contribution from Peck et al., wollastonite mines in the Adirondack Mountains of New York show evidence for passive capture of atmospheric CO2, where weathering has caused the formation of calcite + opal cements, sequestering carbon and cementing crushed ore. Carbon isotopes provide evidence for a microbial role in carbon sequestration, which might be important in future engineered carbon capture projects.
Geochemical variation in biotite from the Devonian South Mountain Batholith, Nova Scotia: Constraints on emplacement pressure, temperature, magma redox state and the development of a magmatic vapor phase (MVP)
https://doi.org/10.2138/am-2022-8691In this paper from Brenan et al., the composition of biotite from the South Mountain Batholith (SMB; Nova Scotia, Canada) is used to quantify intensive parameters during crystallization and to establish a baseline for variation to highlight anomalies associated with mineralization. MELTS thermodynamic modeling using biotite Fe/Fe+Mg-Ti relations implies reducing conditions during crystallization and suggests a role for graphitic sediments in SMB genesis. Biotite F-Cl-OH relations record a progressive decrease in Cl and OH, with increasing F, which can be modeled by SMB crystallization in the presence of a magmatic vapor phase (MVP). Results have implications for granophile element concentration processes in the SMB magmatic system, with the role of extensive crystallization as an important metal enrichment process. Biotite compositional variation in the context of quantitative modeling provides a powerful tool for unraveling the magmatic history and mineralization potential of felsic magmatic systems.
Nanostructural domains in martian apatites that record primary subsolidus exsolution of halogens: Insights into nakhlite petrogenesis
https://doi.org/10.2138/am-2022-8794Apatite has been used in many geologic and biologic disciplines. Over the last decade, in planetary sciences, apatite has been widely used to investigate the nature of volatiles (F, Cl, H, and their isotope systems) associated with magmas and fluids from which they crystallized. However, little is known about natural apatite microstructures, which are critical for the interpretation of these volatile measurements. In this paper, Martinez et al. investigate the nanostructures of apatites in two martian nakhlites that have not been studied previously by FIB-TEM techniques, and they utilize the acquired information to gain additional insights into martian volatiles and the existing petrogenetic models for the chassignites-nakhlites system. Their work reveals novel information about the nanoscale structure of martian apatite, the most important one being 5-10 nm sized domains characterized by differences in contrast and undulosity of the lattice fringes that indicate localized strain. The lattice fringes maintain continuity across different domains, indicating that the strain is elastic. They argue that this is the result of the segregation of the halogens F and Cl into different domains in the apatite structure and conclude that vacancies were key in stabilizing these ternary apatites that otherwise would have been immiscible. The domains are a primary feature that represent exsolution during subsolidus cooling (<800 °C). In addition, Nakhla intercumulus apatites show zonation of REEs, Si, and Fe at fine scales, which is consistent with crystallization from a melt that was enriched in Cl. Finally, the differences in Nakhla apatite compositions between and within melt pockets may result from degassing of the intercumulus melts, which influenced the F-Cl-OH contents in apatites.
Magnetism and equation of states of fcc FeHx at high pressure
https://doi.org/10.2138/am-2022-8452There is a core of iron alloy at the center of the Earth and Earth-like planets (e.g., Mercury and Ganymede). Gomi and Hirose performed theoretical calculations on iron alloyed with hydrogen. Their results suggest that the presence or absence of magnetism in the iron hydride affects the density and the speed of seismic wave propagation. Comparison between their prediction and previous observations of these quantities shows that the Earth’s core must contain elements other than hydrogen.
Hydrothermal alteration of magmatic titanite: Implications for REE remobilization and the formation of ion-adsorption HREE deposits, South China
https://doi.org/10.2138/am-2022-8644Ion-adsorption rare earth element (REE) deposits in South China are currently the main source of heavy rare earth elements (HREE). The newly discovered Gucheng deposit is one good example of HREE mineralization hosted in weathered coarse-grained biotite granites (CGBG). A systematic study of REE-rich magmatic minerals in CGBG provides insight into the remobilization and enrichment of HREE. The results from this work by Feng et al. show that magmatic titanite is an important HREE-rich mineral and multiple stages of replacement processes havetransformed magmatic titanite to more weatherable HREE-bearing minerals, resulting in enrichment of HREE, fractionation of LREE and HREE, and contributed to the ion-adsorption HREE mineralization.
Effects of crystal chemistry on adsorption, occurrence, and mobility of water in palygorskite tunnels
https://doi.org/10.2138/am-2022-8762Zhou et al. performed Grand canonical Monte Carlo and molecular dynamics simulations to investigate multiple water distribution models in the tunnels of palygorskite. The presence of structural counterions lowers the amount of zeolitic water (~2 water molecules per counterion). Zeolitic water in palygorskite tunnels has a 2~3-order lower mobility than bulk water, and counterions further decrease their mobility. The zeolitic water confined in the tunnel has a very low mobility, and the appearance of Na+ ions in the charged palygorskite further reduces the mobility of zeolitic water. Compared with other clay minerals, the much lower water mobility implies that palygorskite could be a more efficient in fixing foreign molecules or ions in environmental applications.
Temperature-induced densification in compressed basaltic glass revealed by in-situ ultrasonic measurements
https://doi.org/10.2138/am-2022-8694In this study, Xu et al. determined the acoustic velocity of a model basalt glass along different P-T paths, including cold compression, isothermal hot compression, and heating-cooling at high P. They found that temperature can facilitate the densification of the model basalt glass at high P, producing higher a acoustic velocity and a smoother pressure dependence of velocity than the cold-compressed glass. Their results clearly demonstrate that the glass acoustic properties are strongly dependent on its P-T path, and suggest that silicate glasses, especially cold-compressed ones, are not good analogs for studying the acoustic and elastic properties of their corresponding melts. This has significant implications for seismic detection of partial melts in the Earth's interior.
X-ray absorption records of Pd2+ on Ni site in pentlandite
https://doi.org/10.2138/am-2022-8704Norilsk sulfide ores (Russia) are one of the largest sources of palladium (Pd) on Earth. Palladium occurs in platinum group minerals (PGM) and in base metal sulfides (BMS), especially in pentlandite. But the exact form of Pd in pentlandite is still unknown. Brovchenko et al. present the results of the first X-ray absorption measurements of Pd in pentlandite. They detected divalent Pd in pentlandite and Ni substitution of Pd in the pentlandite structure. These results are essential to understand incorporation of noble metals in natural minerals with important implications for ore deposit formation.
Twinning in hydrous wadsleyite: symmetry relations, origin, and consequences
https://doi.org/10.2138/am-2022-8596Miyajima et al. report theirdiscovery of twinning in hydrous wadsleyite. The twins formed during crystal growth and may affect the physical properties of wadsleyite, as well as the phase transition to ringwoodite in the Earth's mantle transition zone. The twinning may reduce the mobility of dislocations in twinned wadsleyite grains and affect the deformation of wadsleyite-bearing rocks in the mantle transition zone. The twinning will also reduce the elastic anisotropy of wadsleyite and might enhance the kinetics of the phase transition from wadsleyite to ringwoodite.
An experimental crystallization of the Macusani obsidian in a thermal gradient with applications to lithium-rich granitic pegmatites
https://doi.org/10.2138/am-2022-8674London reports experiments with solid cores of the Macusani obsidian in a thermal gradient at 200 MPa pressure, which constitute the third experimental program with this glass, and the results are compared to the previous studies to elucidate the effects of the experimental methodology on the outcomes. This assessment is generally applicable to all experiments in which crystallization ensues from melt. The principal petrologic contributions of these experiments include a validation of the feldspar solvus algorithms to low, subsolidus temperatures, an experimentally determined solvus for the alkali feldspars attained through the crystallization of undercooled melt at low temperature, records of chemical gradients along and across the melt volumes, and documentation of the spatial zonation of crystalline assemblages and their textures. Oscillations in modal mineral proportions across the cores and along their lengths in the direction of the thermal gradients closely match similar patterns seen in natural pegmatites.
Amorphous Mn2SiO4: A potential manganese phase in the stagnant slab
https://doi.org/10.2138/am-2022-8606Tephroite (Mn2SiO4) recently was discovered in ophiolite-hosted diamonds from deep Earth. Through high-pressure and high-temperature experiments, it is found that tephroite undergoes a pressure-induced amorphous transformation and this transformation is independent of temperature in the temperature and pressure range of this study. Manganese significantly reduces the amorphization pressure of olivine. This work by Ye et al. indicates that tephroite may exist in the slab in an amorphized form during subduction into the deep Earth, which provides insight into oceanic crustal recycling.
The crystal structure of feitknechtite (β-MnOOH) and a new MnOOH polymorph
https://doi.org/10.2138/am-2022-8729Post et al. provide the complete structure description for β-MnOOH (feitknechtite). Additionally, they identified and report the structure for a second MnOOH phase, similar to β-MnOOH but with a different layer stacking, and as such seems to be a new MnOOH polymorph. Several researchers have described feitknechtite as a requisite intermediate phase during the synthesis of technologically important Mn (hydr)oxides. It is also assumed to play a critical role in abiotic and biotic processes that produce and alter Mn oxide phases in a variety of natural environments. The lack of knowledge of the atomic structure of feitknechtite has limited our understanding of its precise role and behavior in synthetic and natural redox reactions in manganiferous systems. This work provides the formal structure description required for model calculations that will provide a better understanding of this phase and make possible predictions about the behavior of β-MnOOH (feitknechtite) under a range of conditions. It also provides an essential starting point for analyzing powder diffraction data to determine and monitor unit-cell parameters and other structure details, and for quantitative phase analyses of Mn oxide samples that contain feitknechtite. Feitknechite has been part of the Mn oxide conversation for more than seven decades, and it is satisfying to finally know what it is.
Yakubovichite, CaNi2Fe3+(PO4)3, a new nickel phosphate mineral of non-meteoritic origin
https://doi.org/10.2138/am-2022-8800Yakubovichite, a new mineral containing up to 20 wt% NiO, represents a novel type of terrestrial phosphate mineral with extreme enrichment in Ni. The possible sources of Ni in the reported mineral assemblages are discussed by Britvin et al. The anomalous enrichment of pyrometamorphic assemblages in Ni may be related to metamorphic assimilation of Ni-rich minerals accumulated in the Cretaceous-Paleogene boundary layer, which was formed by the Chicxulub collision.
October 2023
Heavy halogen compositions of lamprophyres derived from metasomatized lithospheric mantle beneath eastern North China Craton
https://doi.org/10.2138/am-2022-8693Major findings by Zheng et al. include: (1) Lamprophyres derived from the metasomatized lithospheric mantle are enriched in water-bearing minerals and fluid-mobile large ion lithophile elements but strongly depleted in heavy halogens. (2) The mantle source of lamprophyres should be halogen-rich. (3) Volatile saturation and fluid exsolution lead to strong partitioning of the halogens into the exsolved fluid and, thus, noticeable halogen depletion. (4) Given the presence of Cl-poor amphibole, the halogen loss occurred before amphibole crystallization.
Compositional trends in Ba-, Ti-, and Cl-rich micas from metasomatized mantle rocks of the Gföhl Unit, Bohemian Massif, Austria
https://doi.org/10.2138/am-2022-8746This study by Zelinkova et al. describes exotic Ba, Cl-rich phases in mantle pyroxenites and defines principal substitution mechanisms in Ba-, Cl-, and Ti-bearing micas. Data show that XFe exerts an important control on mica composition. Mica with 10.98 wt% Cl represents the most Cl-rich mica ever described. Association with Cl-rich phosphates and carbonates indicates metasomatism of mantle with fluid/melt with high activity of Cl and CO2 and low H2O activity.
Experimental determination of quartz solubility in H2O-CaCl2 solutions at 600-900 °C and 0.6-1.4 GPa
https://doi.org/10.2138/am-2022-8387Makhluf et al. investigated the solubility of quartz in H2O-CaCl2 fluids at high pressure and temperature. They found that quartz grows less soluble as the salt content rises. The data were used to model the properties of CaCl2 brines, which can be important in a wide range of metasomatic environments.
The use of boron nitride to impose reduced redox conditions in experimental petrology
https://doi.org/10.2138/am-2022-8577Li et al. demonstrate that BN can impose QIF-like redox conditions when used as a capsule sleeve in experimental assembly. The underlying mechanism is proposed to be 2BN+3H2O=B2O3+N2+3H2. Therefore, the redox-imposing potential of BN is a function of H2O activity. Oxygen fugacity control is a difficult task. The improved understanding of such a process in this study may lead to better recognition and exploration of the redox-imposing potential of BN in experimental petrology.
Structures and transport properties of supercritical SiO2-H2O and NaAlSi3O8-H2O fluids
https://doi.org/10.2138/am-2022-8724Sun et al. demonstrate that the exponential decrease in the viscosity of silicate melt with increasing water content is due to a sharp decrease in the proportion of Q4 and increase in Si-O-H. They show that the supercritical fluids contain a large amount of partially polymerized or depolymerized silicate units bonded to hydrogen, which makes them rich in silicate while maintaining a low viscosity. This feature provides supercritical fluids the potential to transport elements that are hard to migrate in aqueous fluids or hydrous silicate melts, such as high field strength elements.
Hydrologic regulation of clay-mineral transformations in a redoximorphic soil of subtropical monsoonal China
https://doi.org/10.2138/am-2022-8706A red soil profile with evident redoximorphic features was investigated by Zhao et al. with a focus on clay-mineral transformations. The study shows that hydrologic conditions (i.e., acidic cation concentration in soil solutions, waterlogging, and redox oscillations) are tightly coupled with the transformation pathways of secondary minerals, especially the expandable 2:1 clay minerals (i.e., vermiculite and smectite groups). The results advance the understanding on the transformation processes between different clay minerals and their environmental controls in natural soil systems.
Witness to strain: Subdomain boundary length and the apparent subdomain boundary density in large strained olivine grains
https://doi.org/10.2138/am-2022-8441Li et al. used the electron backscatter diffraction technique to examine shock-induced subdomains in olivine single grains. The apparent subdomain density is quantified by Unit Segment Length, which is a Matlab-based code program measuring unit subdomain boundary length over the area of the measured grain. An increasing trend of USL is observed with an increase of shock degree. When comparing with non-shocked Hawaiian xenolith, they observe a significant USL difference, suggesting that shock metamorphism induces pervasive disturbance as the result of its destructive effects.
Libyan Desert Glass: New evidence for an extremely high-pressure-temperature impact event from nanostructural study
https://doi.org/10.2138/am-2022-8759Kovaleva et al. confirm the high-pressure, high-temperature origin of the Lybian Desert Glass, which has been under question for many years. This means that the impact crater responsible for this glass’s origin is yet to be discovered. They also demonstrate that transmission electron microscopy is a very powerful yet insufficiently used tool for unraveling deformation features in accessory minerals, which provides information about the pressure, temperature, and cooling histories of the deformation process.
Crystal versus melt compositional effects on the partitioning of the first-row transition and high field strength elements between clinopyroxene and silicic, alkaline, aluminous melts
https://doi.org/10.2138/am-2022-8586He et al. demonstrate that trace element partitioning in silicic melts differs from that in basaltic melts, and they present a scheme for evaluating the specific influence of different crystal and melt compositional variables on the partitioning of FRTE and HFSE. The results have important implications for the partitioning behavior of trace elements in evolved silicic systems, particularly for trace element systematics relevant to mantle partial melting or metasomatic processes operated by highly silicic melts.
Microbially induced clay weathering: Smectite-to-kaolinite transformation
https://doi.org/10.2138/am-2022-8442The occurrence of the microbe-induced clay transformation from 2:1 type smectite to 1:1 type kaolinite has lacked solid experimental evidence. Yang et al. provide a novel pathway to achieve microbial kaolinization of smectite by a silicophilic bacterium at a highly efficient transformation rate. Mineralogical change and kaolinization mechanism during the transformation process have been analyzed in detail, which attributes a previously unrevealed but efficient microbial pathway to the widespread clay weathering.
Hydrous wadsleyite crystal structure up to 32 GPa
https://doi.org/10.2138/am-2022-8380Wang et al. used new ultrafast diffraction setups in single-crystal X-ray diffraction to determine the full structure of hydrous wadsleyite up to 35 GPa, with a focus on the hydrogen bonds. By using two different synthetic samples of mantle composition (~Fo90) olivine with different amounts of water (~2 wt% and ~0.25 wt%), they systematically compared the influence of water on the wadsleyite structure at elevated pressures. Both Fo90 wadsleyite samples became monoclinic at pressures above 9 GPa, and there is evidence that hydrogen bond becomes incompressible in wadsleyite at >25 GPa.
Multiple fluid sources in skarn systems: Oxygen isotopic evidence from the Haobugao Zn-Fe-Sn deposit in the southern Great Xing’an Range, NE China
https://doi.org/10.2138/am-2022-8523Liu et al. investigated fluid sources and fluid evolution in skarn systems through a systematic study in petrology, composition, and in-situ oxygen isotope of Haobugao deposit. They found garnets at Haobugao deposited from aqueous fluids that were predominantly of magmatic and meteoric origin and mixtures thereof. Mixing of these components occurred at the start of the skarn stage. There are multiple stages of hydrothermal fluid at Haobugao, and episodic flux of magmatic fluid was recorded in the skarn minerals. The amount of meteoric water did not increase with decreasing depth, and the central exoskarn displays complex fluid mixing.
Crocobelonite, CaFe3+2(PO4)2O, a new oxyphosphate mineral, the product of pyrolytic oxidation of natural phosphides
https://doi.org/10.2138/am-2022-8757Britvin et al. report on a new phosphate mineral, which represents a specific, novel type of natural phosphate mineralization. The assemblages were formed by pyrolytic oxidation of phosphide minerals at temperatures above 1000 °C and near-atmospheric pressure. From a crystal chemical point of view, crocobelonite is a rare example of phosphate, which exhibits unit-cell twinning (chemical twinning) — the type of polymorphism that is more common for silicates.
Tetrahedrite-(Ni), Cu6(Cu4Ni2)Sb4S13, the first nickel member of tetrahedrite group mineral from Luobusa chromite deposits, Tibet, China
https://doi.org/10.2138/am-2022-8761Wang et al. report on tetrahedrite-(Ni), ideally Cu6(Cu4Ni2)Sb4S13, the first natural Ni-member of tetrahedrite group mineral. The new species occurs with gersdorffite, vaesite, and chalcostibite, which are disseminated in a matrix of dolomite, magnesite, quartz, Cr-rich mica, and Cr-bearing clinochlore. Tetrahedrite-(Ni) may be the product of late-serpentinization at moderately high-temperature conditions (about 350 C). In this case, tetrahedrite-(Ni) and its mineral paragenesis record an entire geological process of nickel enrichment, migration, activation, precipitation, and alteration from deep mantle to shallow crust.
New Mineral Names: Heavy Metal and Minerals from China
https://doi.org/10.2138/am-2023-NMN108106September 2023
Fluorine-rich mafic lower crust in the Southern Rocky Mountains: The role of pre-enrichment in generating fluorine-rich silicic magmas and porphyry Mo deposits
https://doi.org/10.2138/am-2022-8503Amphiboles in the deep mafic crust of Colorado are enriched in fluorine (F). Numerical models and geochemical data support a mafic lower crustal source for F-rich shallow magmas in the region. Melts with greater mantle- or ancient felsic crustal-components lack F enrichment. The results of Rosera et al. suggest that ancient mafic lower crust in Colorado may have undergone multiple melting episodes in the Cenozoic, but F-rich magmas were only generated during periods of high heat flow that broke down F-rich amphibole.
Apatite in brachinites: Insights into thermal history and halogen evolution
https://doi.org/10.2138/am-2022-8712Zhang et al. propose that intergranular apatite grains in brachinites have been replaced by merrillite. They find the presence of augite in pyroxene-troilite intergrowths, which are products of sulfidization of olivine. And they find that all apatite grains in brachinites have experienced chromite exsolution. They find the presence of a fluorapatite inclusion in NWA 4969 and propose that the apatite inclusions with, or without, subhedral-to-euhedral merrillite could be relicts of the precursor materials of brachinites.
A high-pressure structural transition of norsethite-type BaFe(CO3)2: Comparison with BaMg(CO3)2 and BaMn(CO3)2
https://doi.org/10.2138/am-2022-8722This article by He et al. reported the effect of ionic radii on phase transition pressures of carbonates. The phase transition pressures of BaMg(CO3)2, BaFe(CO3)2, and BaMn(CO3)2 are 2.4(2), 2.7(5), and 3.9(2) GPa, respectively. The effective cation radii of Ba2+, Ca2+, Mn2+, Fe2+, and Mg2+ are 1.35, 1.00, 0.83, 0.78, and 0.72 Angstroms at ambient conditions, respectively. With the addition of the norsethite-type members, it is clear that a smaller metal cation tends to stabilize the trigonal structure to higher pressure in carbonates, and the phase transition pressures are much lower for norsethite-type carbonates than that of calcite- and dolomite-type carbonates. However, unlike the linear trend reported previously, the relationship tends to be nonlinear in the norsethite-type minerals. The onset of the phase transition pressures to high-pressure phases increases with cation radii for norsethite-type minerals, while the results are opposite for both calcite- and dolomite-type carbonates. This is attributed to the larger ratio between the radii of the Ba2+/(Mg2+, Mn2+, Fe2+) ions compared to that of Ca2+/(Mg2+, Mn2+, Fe2+). The effect of ionic radii on phase transition pressures has been found not only in carbonate minerals but also in other materials and can be applied to predict structural stability in isostructural materials.
An evolutionary system of mineralogy, Part VII: The evolution of the igneous minerals (>2500 Ma)
https://doi.org/10.2138/am-2022-8539Part VII of the evolutionary system of mineralogy by Hazen et al. catalogs, analyzes, and visualizes relationships among 919 natural kinds of primary igneous minerals, which are associated with the wide range of igneous rock types through more than 4.5 billion years of Earth history. A systematic survey of the minerals in 1850 varied igneous rocks from around the world reveals that 115 of these mineral kinds are frequent major and/or accessory phases. Patterns of coexistence among these minerals, revealed by network, Louvain community detection, and agglomerative hierarchical clustering analyses, point to four major communities of igneous primary phases, corresponding in large part to different compositional regimes: (1) quartz- and/or alkali feldspar-dominant rocks, including rare-element granite pegmatites; (2) mafic/ultramafic rock series with major calcic plagioclase and/or mafic minerals; (3) rocks with major feldspathoids and/or analcime, including agpaitic rocks and their distinctive rare-element pegmatites; and (4) carbonatites and related carbonate-bearing rocks. Igneous rocks display characteristics of an evolving chemical system, with significant increases in their minerals' diversity and chemical complexity over the first two billion years of Earth history. Earth's first igneous rocks (>4.56 Ga) were ultramafic in composition with 122 different minerals, followed closely by mafic rocks that were generated in large measure by decompression melting of those ultramafic lithologies (4.6 Ga). Quartz-normative granitic rocks and their extrusive equivalents (>4.4 Ga), formed primarily by partial melting of wet basalt, were added to the mineral inventory, which reached 246 different mineral kinds. Subsequently, four groups of igneous rocks with diagnostic concentrations of rare element minerals — layered igneous intrusions, complex granite pegmatites, alkaline igneous complexes, and carbonatites — all appeared less than 3 billion years ago. These more recent varied kinds of igneous rocks hold more than 700 different minerals, 500 of which are unique to these lithologies. Network representations and heatmaps of primary igneous minerals illustrate Bowen's reaction series of igneous mineral evolution, as well as his concepts of mineral associations and antipathies. Furthermore, phase relationships and reaction series associated with the minerals of a dozen major elements, as well as minor elements, are embedded in these multi-dimensional visualizations.
Oriented secondary magnetite micro-inclusions in plagioclase from oceanic gabbro
https://doi.org/10.2138/am-2022-8784In the paper by Bian et al., oriented needle-shaped magnetite micro-inclusions in plagioclase from oceanic gabbro occur in two generations. Primary inclusions are elongated perpendicular to seven important plagioclase lattice planes. During hydrothermal processing, they recrystallize into secondary magnetite needles aligned parallel to the plagioclase c-axis. This ensures a good match between the oxygen sublattices and a good linkage between crystal structure elements across magnetite-plagioclase boundaries.
A multi-methodological study of the bastnäsite-synchysite polysomatic series: Tips and tricks of polysome identification and the origin of syntactic intergrowths
https://doi.org/10.2138/am-2022-8678Bastnäsite-synchysite fluorcarbonates have been investigated by several techniques, including micro-Raman spectroscopy, electron backscattered diffraction (EBSD), and high-resolution transmission electron microscopy (HRTEM) in this contribution by Conconi et al. EBSD was effective in establishing the sample orientation and to ascertain the syntactic relationship among the detected fluorcarbonates but failed to distinguish among different polysomes. Raman spectroscopy, which offers the advantage of being a non-destructive technique, allowed the distinction of different polysomes, but it could not distinguish between ordered and disordered polysomes with the same composition. HRTEM was confirmed as the ultimate technique for polysome identification, but unfortunately, it is destructive. Several ordered polysomes were detected in addition to the basic ones, including a B2S and a long-range polytype with a 32 nm repeat distance along the c-axis. Overall, the detected microstructure is indicative of a growth mechanism in which fluorcarbonates crystallize from a fluid close to thermodynamic equilibrium, whose conditions quickly and repeatedly crossed the parisite-bastnäsite stability boundary.
Petrogenesis of Chang'E-5 mare basalts: Clues from the trace elements in plagioclase
https://doi.org/10.2138/am-2022-8570In this contribution by Tian et al., they found that (1) The melt inverted from the Chang'E-5 plagioclase has higher incompatible element concentrations than the Apollo samples but close to the KREEP-rich rocks. (2) The enrichment of trace elements reflects a high degree of fractional crystallization. (3) The parental melt’s TiO2 content estimated from the earliest crystallized plagioclase is ~3.3 wt%, suggesting a low-Ti origin for Chang'E-5 basalts.
Experimental investigation of trace element partitioning between amphibole and alkali basaltic melt: Toward a more general partitioning model with implications for amphibole fractionation at deep crustal levels
https://doi.org/10.2138/am-2022-8536Bonechi et al. present an experimental study performed on a K-basalt at 0.8 GPa and 1030-1080 °C to provide new data on the partitioning of trace elements between amphibole and melt. Indeed, despite numerous investigations on the partitioning of trace elements between crystals and melts, there are still some mineral phases, including amphibole, for which data are limited or missing. These new data allowed them to estimate the ideal radius, the maximum partition coefficient, and the apparent Young's modulus of the A, M1-M2-M3, and M4-M4' sites of amphibole. Moreover, the influence of melt and amphibole composition, temperature, and pressure on the partition coefficients between amphiboles and glasses has also been investigated by comparing their data with a literature dataset spanning a wide range of pressures (0.6-2.5 GPa), temperatures (780-1100 °C), and compositions (from basanite to rhyolite). Finally, Bonechi et al. modeled a deep, fractional crystallization process using the amphibole-melt partition coefficients determined in this study, observing that significant amounts of amphibole crystallization (>30 wt%) well reproduce the composition of an andesitic melt similar to that of the calc-alkaline volcanic products found in Parete and Castelvolturno bore-holes (NW of Campi Flegrei, Italy).
Grain-scale zircon Hf isotope heterogeneity inherited from sediment-metasomatized mantle: Geochemical and Nd-Hf-Pb-O isotopic constrains on Early Cretaceous intrusions in central Lhasa Terrane, Tibetan Plateau
https://doi.org/10.2138/am-2022-8508Combined zircon U-Pb, Hf, and O isotope investigations are widely used to address the contribution of mantle and sediment components in the source of igneous rocks. In particular, zircon Hf isotopic variabilities in a single sample beyond the analytical uncertainty are commonly interpreted as the mixing of magmas derived from two isotopically different reservoirs. However, subducted sediments may have profound impacts on the Hf budget of the mantle, theoretically adding complexity to interpreting zircon Hf data. Nevertheless, compared with granitoids, Hf isotopic variation is rarely observed in zircons of mafic rocks. In this study, Li et al. report integrated data of whole-rock geochemistry (major, trace elements, and Sr-Nd isotopes), zircon U-Pb, Hf and O isotopes and trace elements, and in situ clinopyroxene major and trace elements and Pb isotopes, for some newly recognized gabbro-diorite rocks in central Lhasa Terrane, Tibetan Plateau. The magmatic zircons present dramatically heterogeneous Hf isotopes (from +13 to -4) even in the same individual grain, yet their O isotopes are relatively uniform and are slightly higher than that of the mantle value. Combined with their relatively constant clinopyroxene Pb isotopes and whole-rock geochemistry, they contend that the zircon Hf isotope heterogeneities were inherited from a depleted asthenospheric mantle metasomatized by 1-4% terrestrial sediments. Their study, therefore, emphasizes caution when using zircon Hf isotopes as arguments of involvement of two end-member magmas at the crustal level without comprehensive mineral and geochemical investigations.
Mechanism and kinetics of the pseudomorphic replacement of anhydrite by calcium phosphate phases at hydrothermal conditions
https://doi.org/10.2138/am-2022-8592This study by Roza-Llera et al. focuses on the kinetics of the replacement of anhydrite single crystals by mixtures of the calcium phosphate phases, beta-tricalcium phosphate, and hydroxyapatite via an interface-coupled dissolution-precipitation reaction in the temperature range between 120 to 200 °C. Both the Avrami and the iso-conversion methods yield an empirical activation energy Ea (kJ/mol) of about 40 kJ/mol for this replacement reaction. The dissolution of anhydrite appears to be the rate-limiting process, and the overall kinetics of the replacement reaction is controlled by the rate of diffusion of dissolved species through the pore network. These results open a window for the development of new strategies for the recovery of P, a scarce element in the Earth's crust, through the precipitation of phosphate phases through dissolution-crystallization reactions that involve a pre-existing mineral.
Vacancy infilling during the crystallization of Fe-deficient hematite: An in situ synchrotron X-ray diffraction study of non-classical crystal growth
https://doi.org/10.2138/am-2022-8379The work by Chen et al. highlights a non-classical crystallization pathway involving vacancy infilling by cations during nanoparticle growth. Incipient hematite nanocrystals nucleated with Fe deficient concentrations as high as 40 mol%, and the Fe occupancy increased as Fe3+ cations replaced H+ during crystal growth until reaching a steady state. The steady-state vacancy concentration in the final product could be controlled by the reaction environment, including pH, temperature, and time.
Simulated diagenesis of the iron-silica precipitates in banded iron formations
https://doi.org/10.2138/am-2022-8758Iron-silica precipitates were once deposited across the ocean, but the legacy of this time now only lives on in iron and silica-rich minerals hosted in rocks known as Banded Iron Formations (BIFs). Hinz et al. developed a new experimental method to partially oxidize iron under ancient ocean-like conditions and initially formed a precursor iron silicate, similar to the mineral greenalite recently proposed as the original BIF sediment, as well as subsidiary iron oxides. After simulated post-depositional aging, they observed crystallization of Mg-rich greenalite and magnetite, a common BIF mineral, as well as some persistent iron oxides, suggesting new ways to identify alteration and extract primary information held in BIFs.
Wave vector and field vector orientation dependence of Fe K pre-edge X-ray absorption features in clinopyroxenes
https://doi.org/10.2138/am-2022-8547The pre-edge energy range of X-ray absorption spectra is commonly used to quantify redox ratios of multivalent elements. In contrast with anisotropy observed as optical pleochroism in petrographic thin sections, pre-edge absorption is largely the product of quadrupole transitions. With this difference in mind, Steven et al. find that in different analytical geometries, the absorption anisotropy is as much a function of the propagation direction as is the polarization direction for the pre-edge absorption of clinopyroxenes.
Structure and compressibility of Fe-bearing Al-phase D
https://doi.org/10.2138/am-2022-8559Criniti et al. studied the crystal structure and compressibility of Fe-bearing Al-phase D, a possible major water carrier in Earth's mantle transition zone and shallow lower mantle. They found that the symmetry and crystal structure of this phase are intermediate between those of pure Mg-phase D and Al-phase D, while its bulk modulus is in agreement with some previous studies on Mg-phase D. Additionally, no change in the compression behavior due to the symmetrization of H-bonds was found, suggesting the elasticity of phase D is relatively insensitive to the chemical composition and degree of order.
Synthesis of boehmite-type GaOOH: A new polymorph of Ga oxyhydroxide and geochemical implications
https://doi.org/10.2138/am-2022-8568A new polymorph of GaOOH (γ-GaOOH) was synthesized with boehmite as a structure template. The results provide insight into Ga in boehmite and indicate that boehmite can act as a template to enrich free Ga and epitaxially induce nucleation and growth of γ-GaOOH. This study by Liu et al. also provides a potential migration, enrichment, and mineralization mechanism of Ga, which will improve the understanding of the geochemical processes and occurrence of Ga in nature.
Scheelite U-Pb geochronology and trace element geochemistry fingerprint W mineralization in the giant Zhuxi W deposit, South China
https://doi.org/10.2138/am-2022-8495Skarn-type tungsten deposits dominate the world's W supply, however, the temporal relation between the W mineralization, causative intrusions, and the sources of ore-forming fluids and metals is still a matter of great debate. In this contribution, Zhao et al. report in situ LA-ICP-MS U-Pb dating and trace element compositions of scheelite from the world's largest tungsten deposit to address the above issues. Their study highlights that, compared to other hydrothermal accessory minerals (such as molybdenite, muscovite, apatite, etc.), scheelite LA-ICP-MS U-Pb dating is a robust technique to determine the mineralization age of skarn W deposits. This study also discovered that the destruction of early-formed garnet could produce a high Y/Ho ratio for the subsequent ore-forming fluid and could provide metals such as W, Cu, and Sn to form skarn-type ore deposits. The new results from this study contribute to a better understanding of the metal source and fluid evolution for the skarn-type ore deposits.
A rare sekaninaite occurrence in the Nenana Coal Basin, Alaska Range, Alaska
https://doi.org/10.2138/am-2022-8698In this contribution by Reidel and Ross, coal-seam fires represent an unusual and relatively unexplored natural environment of mineral formation in pyrometamorphic rocks. The Mystic Creek coals burned with an extremely high temperature and produced sekaninaite, a relatively rare mineral as well as a yet unidentified Al-Fe-TI opaque mineral. This coal-seam fire is a natural laboratory for observing low-pressure, high-temperature fractional crystallization paths in magmas. Thus, pyrometamorphic rocks like that at the Mystic Creek coal basin provide a valuable natural laboratory for exploring magmatic processes and new minerals for future mineralogical studies.
Slyudyankaite, a new sodalite-group mineral from the Malo-Bystrinskoe lazurite deposit, Baikal Lake area
https://doi.org/10.2138/am-2022-8598The article by Sapozhnikov et al. is a contribution to the crystal chemistry of sodalite-group minerals. It describes the new mineral slyudyankaite, approved by the IMA CNMNC. Slyudyankaite is a very unusual new member of the sodalite group containing S6 and CO2 molecules as species-defining components. This mineral is an example of the separation of extra-framework components in cages of two types. Cages of the first type contain cations (Na+ and Ca2+) and SO42- anions, whereas cages of the second type are occupied by neutral molecules (S6, S4, CO2, and H2O). Based on a multimethodic approach involving six spectroscopic methods, it was shown that the variable color of slyudyankaite is related to the presence of polysulfide chromophores (S6 molecules as well as trace amounts of S2-, S3-,·and S4·;- radical anions). Extra-framework components in slyudyankaite and other sodalite-group minerals are important markers of volatile species in the mineral-forming medium.
Ruizhongite, a thiogermanate mineral from the Wusihe Pb-Zn deposit, Sichuan Province, Southwest China
https://doi.org/10.2138/am-2023-9000The discovery of ruizhongite has significant implications for the occurrence and enrichment mechanism of Ge in sphalerite and other metallic minerals. Ruizhongite, a thiogermanate mineral, was identified in the Wusihe Pb-Zn deposit in Sichuan Province, Southwest China, during an investigation of the mineralogy of this deposit. In the present study by Meng et al., polarized optical microscopy, scanning electron microscopy, electron microprobe, μ-X-ray diffraction, and Raman spectroscopy analyses were utilized to characterize the occurrence, optical property, chemical composition, and crystal structure of ruizhongite. Both the mineral and its name have been approved by the IMA-CNMNC (2022-066). Type specimens are preserved in the Geological Museum of China, Beijing, China (Catalog number M16138).
August 2023
Experimental apatite-fluid interaction and partitioning of rare earth elements at 150 and 250 °C
https://doi.org/10.2138/am-2022-8589Chappell et al. present new hydrothermal experiments at 150 and 250 °C and examine the role of pH, temperature, and NaCl on the solubility of apatite. The mobility of rare earth elements (REE) and formation of secondary REE phosphates have been investigated under acidic and mildly acidic conditions by studying the fluid chemistry and apatite alteration textures. These experiments indicate that apatite alteration is dissolution-controlled at low temperature, and NaCl plays a key role in mobilizing the REE. The experiments also highlight the necessity of appropriately screening igneous apatite grains using backscattered electron and cathodoluminescence imaging for signs of hydrothermal alteration textures as they show apatite is susceptible to a strong alteration overprint down to a temperature of 150 °C.
Assimilation of xenocrystic apatite in peraluminous granitic magmas
https://doi.org/10.2138/am-2022-8668High-temperature, high-pressure experiments by Clarke et al., involving two or three different apatite compositions immersed in a synthetic granite melt, show that dissolution in, and diffusive chemical exchange through, the medium of the melt result in different rates of equilibrium. The halogens in the apatite equilibrate before either the rare-earth elements or the texture of the apatite grains. A natural example of two types of apatite in one granitic rock illustrates that physical and chemical equilibrium is also only partial.
Cathodoluminescence of iron oxides and oxyhydroxides
https://doi.org/10.2138/am-2022-8843In this Open Access paper, Galili et al. used scanning electron microscope cathodoluminescence (SEM-CL) to study iron oxides and oxyhydroxides (goethite, hematite, and magnetite) and gained the following insights: (1) Iron oxides and oxyhydroxides display unique SEM-CL spectra; (2) Subtle variations in mineral-specific spectra reflect mineral formation conditions; (3) Mineral-specific spectra are affected by the substitution of various metals for iron; and (4) SEM-CL is a non-destructive, in-situ tool to characterize iron (oxyhydr)oxides.
The effect of elemental diffusion on the application of olivine-composition-based magmatic thermometry, oxybarometry, and hygrometry: A case study of olivine phenocrysts from the Jiagedaqi basalts, northeast China
https://doi.org/10.2138/am-2022-8567Olivine compositions have been widely used to constrain magmatic thermodynamic conditions such as magmatic temperature, oxygen fugacity, and H2O content. Zhang et al. studied olivine phenocrysts and olivine-hosted spinel from the Jiagedaqi (JGD) alkaline basalts, and their results show that diffusions of Mg, Ca, Fe, Sc, and Y in the JGD olivines have a strong influence on magmatic temperature calculated using a Sc/Y-in-olivine thermometer, magmatic oxygen fugacity calculated using an olivine-spinel oxybarometer, and magmatic H2O content calculated using a hygrometer based on Ca partitioning between olivine and melt. The compositional plateaus in olivine cores, which were not influenced by elemental diffusion, can preserve the magmatic temperature, oxygen fugacity, and H2O content applied at the formation of the JGD olivines. This study clearly shows that the use of olivine geochemistry to investigate magmatic thermodynamic conditions must consider the effects of elemental diffusion.
Characterization of nano-minerals and nano-particles in supergene rare earth element mineralization related to chemical weathering of granites
https://doi.org/10.2138/am-2022-8543Yi et al. used hollow fiber flow field-flow fractionation and inductively coupled plasma-mass spectrometry, scanning, and transmission electron microscopy to characterize the REE nano-particles in different granite regolith horizons (REEmax = 1201 ppm) and the association between REEs and clay minerals. Two different types of REE-bearing nano-particles were observed: Ce oxides which are mainly enriched in the upper horizon and occur as cerite, and amorphous La, Nd, and Y phosphates, which are mainly enriched in the lower horizons. These nano-particles are commonly attached to the surfaces of clay minerals. Compared with platy kaolinite, tubular halloysite occurs mainly in the lower horizons, has a higher adsorption capacity for the REEs, and thus is responsible for the REE enrichment in ion adsorption-type REE deposits. The findings indicate that there are various types of nano-minerals and -particles that affect REE enrichment and fractionation during granite weathering.
Atomic-scale interlayer friction of gibbsite is lower than brucite due to interactions of hydroxyls
https://doi.org/10.2138/am-2022-8561Okuda et al. computed the atomic-scale shear stress to deform layers of gibbsite (Al(OH)3) on its [001] plane using the first-principles method based on density functional theory. They extended the atomic-scale properties to macroscopic shear properties using adhesion theory and then compared the result with previous theoretical and experimental studies on other layered structure minerals. They found that the interlayer deformation yielded a clearly lower friction coefficient compared with experimental values, highlighting the critical role of deformation between layers.
The spatial and temporal evolution of mineral discoveries and their impact on mineral rarity
https://doi.org/10.2138/am-2022-8491Ponomar et al. developed a model to access the relationships between Nickel-Strunz classes, country of discovery, and locality counts at a time scale. According to the minerals discovery rate, three major periods were identified: the ancient period (up to 1800), the Sustainable development period (1800-1950), and the Modern period (1950-present). The bulk of rock-forming minerals was discovered until the 1980s, while the discovery rate of rare and endemic species still progresses.
The role of parent lithology in nanoscale clay-mineral transformations in a subtropical monsoonal climate
https://doi.org/10.2138/am-2022-8740Zhao et al. investigated four soils developed on different rock types in subtropical China to determine the role of parent lithology in clay-mineral evolution at the atomic scale. High-resolution TEM images demonstrated solid-state transformations among clay minerals during weathering. Secondary clay-mineral assemblages depended strongly on the starting materials at weak to moderate degrees of weathering but were controlled mainly by climate under conditions of intense alteration. Thus, nanoscale analysis is critical to evaluation of mineralogic changes that can be obscured by bulk-rock analytical methods.
Discovery of terrestrial andreyivanovite, FeCrP, and the effect of Cr and V substitution in barringerite-allabogdanite low-pressure transition
https://doi.org/10.2138/am-2022-8647Galuskin et al. discovered iron phosphides with significant variations of Cr and V contents on the Earth, which had been known only in meteorites. Cr-V-bearing phosphides may form in the process of replacing fish bone remains. Investigations of the composition and structure of Fe2P phosphides showed that when V+Cr content is higher than 0.26 apfu, a transition from the hexagonal barringerite to the orthorhombic allabogdanite takes place at low pressure. The orthorhombic phosphide with the highest Cr and V contents belongs to andreyivanovite species with the FeCrP end-member formula.
Microstructural changes and Pb mobility during the zircon to reidite transformation: implications for planetary impact chronology
https://doi.org/10.2138/am-2022-8604Impact events modify and leave behind a complex history of rock metamorphism on terrestrial planets. Szumila et al. explore laboratory shock-induced physical and chemical changes to zircon and feldspar. The authors prepared a mixture of sanidine-zircon, 97-3% by weight percent, and shocked the mixture via a flat plate accelerator with an average peak pressure of ~35 GPa. Characterization of unshocked and shocked materials showed that the starting zircon material had abundant metamict regions and the conversion of the feldspar to glass in the post-shock material. Analyses of the shocked product also yielded multiple occurrences of the high-pressure ZrSiO4 polymorph, reidite, with some domains up to 300 μm. The possibility of U-Pb system disturbance was evaluated via LA-ICP-MS and SIMS. The isotopic data reveal that disturbance of the U-Pb geochronometer in the reidite was minimal (<2% for the main U-Pb geochronometers). Numerical simulation confirmed that portions of the shock experiment may have reached pressures where zircon would be expected to transition to reidite but without reaching temperatures that would have reverted reidite to zircon.
Thermal equation of state of ice-VII revisited by single-crystal X-ray diffraction
https://doi.org/10.2138/am-2022-8554Ice-VII is a high-pressure polymorph of H2O ice, which was identified as a mineral recently after its discovery as inclusions in superdeep diamonds. It may exist in the interiors of icy planetary bodies, in some cold subducted slabs, and as inclusions in diamonds of deep origin. Lai et al. synthesized single-crystal ice-VII using an externally heated diamond anvil cell at high pressure and high temperature. They further used this device to reproduce the environments in Earth's and planetary interiors up to 78 GPa at 300-1000 K and investigated the phase stability and thermoelastic properties of ice-VII by synchrotron-based single-crystal X-ray diffraction. There is no structural change or unit-cell volume discontinuity in the investigated pressure-temperature range. The thermal equation of state of ice-VII was determined. The melting point of ice-VII was also found to be consistent with the relatively high melting curve determined in previous studies. These results may be used for modeling the inner structure of icy bodies, determining the entrapment pressure of ice-VII inclusions in superdeep diamonds, and understanding the water cycles in deep Earth.
Empirical electronic polarizabilities for use in refractive index measurements at 589.3 nm: Hydroxyl polarizabilities
https://doi.org/10.2138/am-2022-8717Shannon et al. investigate the relationship between optical properties and the strength of hydrogen bonding in hydroxyl-containing minerals and inorganic compounds. Specifically, they compare the electronic polarizabilities of OH (derived from observed refractive indices) with intrinsic polarizabilities from quantum chemical cluster calculations as a function of the bond length of the OH...O hydrogen bond. It is demonstrated that hydrogen bonding reduces the polarizability of hydroxyl ions, yielding lower refractive indices.
High-pressure behavior of 3.65 Å phase: Insights from Raman spectroscopy
https://doi.org/10.2138/am-2022-8515The hydrous phases often exhibit pressure-induced hydrogen-bond symmetrization, resulting in stiffening of elastic parameters. Basu et al. explored the high-pressure behavior of 3.65 Å phase, a relevant phase in cold slabs, using Raman spectroscopy. They used the pressure dependence of the vibrational modes to deduce the ratio of the bulk moduli and its pressure derivative. These ratios are often low for and are quite diagnostic of hydrous phases and further suggest that deciphering mantle hydration at depths is likely to be challenging owing to the greater pressure dependence of elastic moduli.
High-pressure phase transition and equation of state of hydrous Al-bearing silica
https://doi.org/10.2138/am-2022-8546Criniti et al. investigated the high-pressure structural and vibrational properties of Al,H-bearing SiO2 stishovite and post-stishovite, the silica phases that are stable in subducted basaltic crust in Earth's lower mantle. They found that, in the Al,H-bearing samples, the soft optic mode that characterizes the transition in pure SiO2 is decoupled from the structural phase transition. This may explain some seismic anomalies observed in the proximity of subducted slabs in the topmost lower mantle.
Memorial of Maryellen Cameron
New Mineral Names
July 2023
On the origin of fluorine-poor apatite in chondrite parent bodies
https://doi.org/10.2138/am-2022-8623In this contribution by McCubbin et al., they note that the phosphate mineral apatite exhibits F/Cl ratios in chondrite meteorites that are broadly reflective of the F/Cl ratio of the solar system. Moreover, the halogen geochemistry of extra-terrestrial apatite can provide important constraints on the thermochemical evolution of its parent body. In particular, the F/Cl ratio of apatite can be used as a diagnostic tool to assess whether a parent body has undergone processes such as planetary differentiation or volatile depletion.
Fluorine behavior during experimental muscovite dehydration melting and natural partitioning between micas: Implications for the petrogenesis of peraluminous leucogranites and pegmatites
https://doi.org/10.2138/am-2022-8663Sallet et al. determined experimentally F partitioning between biotite and melt and the natural F partitioning between biotite and muscovite. Along with published F partitioning between fluid and peraluminous melt by Webster and Holloway (1990), they modeled the F behavior during the anatexis of aluminous metapelites and the F distribution between muscovite, biotite, and peraluminous melts. The results indicate that peraluminous melts may be generated by anatexis at the muscovite dehydration isograd, 725-750 °C and 0.4 to 0.6 GPa. The measured melting fractions are below 20 wt%, compatible with the formation of metatexite migmatites. The F behavior deduced for the muscovite dehydration melting of two-mica protoliths show that the more Fe-rich the anatectic residual biotite -- reequilibrated or neoformed -- the more F-rich is the generated peraluminous leucosome. Such compositional control on the partition coefficient is due to the MgO content of residual biotite for biotite/melt partitioning and by the initial F content of the melted rock on the fluid/melt F partitioning. The results of this study show that detailed F in biotite systematics in migmatitic terrains could be a useful tool for the petrogenesis and rare metal potential assessment of peraluminous granites and pegmatites.
Telescoped boiling and cooling mechanisms triggered hydrothermal stibnite precipitation: Insights from the world’s largest antimony deposit in Xikuangshan China
https://doi.org/10.2138/am-2022-8490Whether the transition of Sb complexes occurs in hydrothermal systems and its role in stibnite precipitation are unknown. In this work by Yu et al., δ123Sb for stibnite from the Xikuangshan deposit shows a trend of first decreasing and then increasing from proximal to distal ore bodies. The opposite trends are induced by the transition of Sb complexes. Different stable Sb complexes result in different precipitation mechanisms (fluid boiling and fluid cooling). This study highlights that metal complexes in hydrothermal fluids may change and affect metal precipitation mechanisms.
Correlations between cathodoluminescence intensity and aluminum concentration in low-temperature hydrothermal quartz
https://doi.org/10.2138/am-2022-8471Yuan et al. have integrated scanning electron microscope cathodoluminescence (SEM-CL), electron microprobe (EMP), electron backscatter diffraction (EBSD), X-ray single crystal diffraction (XRD), fourier transform infrared spectroscopy (FTIR), and transmission electron microscope (TEM) data of quartz from the auriferous quartz-sulfide vein type Shihu and Rushan Au deposits in the North China Craton (NCC) to evaluate the genesis of CL-aluminum zoning in low-temperature hydrothermal quartz and the implications for the evolution of auriferous hydrothermal system. Their results provide a new understanding of the genesis of CL-aluminum zoning in low-temperature hydrothermal quartz.
Behavior of hydrogen defect and framework of Fe-bearing wadsleyite and ringwoodite at high temperature and high pressure
https://doi.org/10.2138/am-2022-8544Jin et al. performed experiments to assess hydrogen defect and framework behavior in Fe-bearing wadsleyite and ringwoodite using in situ infrared spectroscopic (IR) and Raman spectroscopic techniques at high temperatures and high pressures. The results provide new knowledge about hydrogen effects and framework of the two minerals.
What is mineral informatics?
https://doi.org/10.2138/am-2022-8613Minerals store an enormous amount of information about the evolution of our planet and other planetary bodies in space. To better understand the evolution of our planet and beyond, we must extract and interpret the information in these minerals. The increased interest in the development and application of data science and informatics methods combined with the increase in mineral data resources implies that we are at a tipping point. In this paper by Prabhu et al., they introduce the concept of “Mineral Informatics,” which will help researchers utilize data science and informatics methods to study minerals and mineral data from various new perspectives and in combination with many other fields of study. The intention of this paper is not to create a new specific field or a sub-field as a separate silo but to create a path for researchers studying minerals to answer important inter-disciplinary scientific questions.
Metal source and hydrothermal evolution of the Jiaoxi quartz vein-type tungsten deposit (Tibet): Insights from textural and compositional variations of wolframite and scheelite
https://doi.org/10.2138/am-2022-8440This paper by Wang et al. evaluates the controlling factors of hydrothermal wolframite and scheelite precipitation in the quartz-vein type Jiaoxi tungsten deposit (western part of the Lhasa terrane, Tibet, China) using texture, mineral major, and trace element geochemistry, and sulfur stable isotope geochemistry. Major and trace element contents and in-situ S isotope compositions of pyrite and chalcopyrite show that an early magmatic fluid which is characterized by the enrichment of W, Mn, Zr, Ti, Sc, and Sn and the depletion of Fe. This magmatic fluid mixed with meteoric water inducing dilution and interacted with the biotite monzogranite porphyry to leach Fe, Mg, and Zn into the system to form wolframites with variable Fe/(Fe+Mn) ratios. The late, Fe-enriched magmatic fluid released from the muscovite granite mixed with meteoric water that leached minor Fe and S from shale to form the Fe-enriched wolframite The compositional zoning of wolframite and scheelite grains as revealed by trace element mapping results indicate a pulsatory magmatic fluid. This study highlights that multiple Fe sources were present in the quartz vein-type tungsten deposit hydrothermal system.
Geochemical processes and mechanisms for Cs enrichment in a hot-spring system
https://doi.org/10.2138/am-2022-8532Wang et al. systematically investigated hot-spring Cs enrichment processes, including migration-precipitation-enrichment-dissolution in the Targejia geothermal system in southern Tibet, China. This system contains large-scale hot spring Cs-deposits with a Cs resource of 1.446 x 104 t (ore grade up to 2.89 wt%). The investigation of the Cs distribution in ore in a micro-scale and ore-forming process models allowed investigation of the dominant mineralization mechanism controlling Cs enrichment, which still remains controversial. The main findings are (1) the observed Cs distribution is heterogeneous in the latest stage of ore (0-4 ka), and Cs is dominantly enriched in clay minerals (illite), which is different from those previous reports that Cs mainly exists in amorphous silica opal; (2) Computed model based on physical and chemical parameters of the present activity geothermal fluid indicate that illite forms at pH of around 7 and >50 °C. Amorphous silica likely precipitated by complex processes, like the formation of ice or directly precipitation; (3) at the early stages of mineralization, initially Cs during dissolution-precipitation can be reset, depending on the elemental supply and mineral dissolution rate, which is further related to climate change of Tibet (rainfall) in Holocene and Pleistocene. Meteoric water-rock interaction modeling implies that clay disappears and cannot re-form at the given physical and chemical conditions, whereas amorphous silica can precipitate again.
Identifying xenocrystic tourmaline in Himalayan leucogranites
https://doi.org/10.2138/am-2022-8615Isotope geochronology and mineral chemistry may, in some cases, be incapable of discriminating inherited from autocrystic crystals. Electron backscatter diffraction (EBSD) analyses focusing on intracrystalline deformation and crystallographic relations can prove useful and provide critical information discriminating grains of different generations. The EBSD method is applied here by Han et al. on tourmaline and can be used in conjunction with, and in support of, in situ geochronology, and may also be used to discriminate inherited from autocrystic grains when geochronology data is not available or when the age differences are smaller than the related analytical uncertainties.
Contrasting alteration textures and geochemistry of allanite from uranium-fertile and barren granites: Insights into granite-related U and ion-adsorption REE mineralization
https://doi.org/10.2138/am-2022-8531Zhang et al. report an attempt to systematically investigate the textures and compositions of allanite during alteration from uranium-fertile and barren granites. First, textures and compositions of the Changjiang allanites combined with the occurrence of abundant fluorite suggest the presence of the superposition of F- and CO2-bearing fluids with a relatively low temperature (≤300 °C) and oxidized nature. Second, U-Pb isotopes in allanite have been used to determine the ages of regional mineralization/hydrothermal events. In this study, in situ U-Pb dating on the darker (secondary) domains of the Changjiang allanite grains yielded a weighted mean U-Pb age of 141.4 +/- 5.6 Ma, consistent with the timing of a uranium mineralization event in the Changjiang uranium ore field (~140 Ma, Zhong et al. 2019) and the age of a crustal extension event (140-135 Ma) in South China. These results provide temporal evidence for the link between uranium release from source rocks and regional crustal extension events in South China. In addition, this study also emphasizes the role of the regional crustal extension in the formation of ion-adsorption REE deposits in South China. Third, this study helps understand the mobilization processes of REE and U from primary minerals during alteration, usually a key step in the formation of an ion-adsorption REE deposit or a uranium deposit.
Synthesis and stability of feiite with implications for its formation conditions in nature
https://doi.org/10.2138/am-2022-8633The mineral feiite is a high-pressure Fe-Ti oxide that was recently discovered in a Martian meteorite. In synthesizing the mineral in the laboratory, Prissel et al. determined the pressures and compositions required to form feiite. These conditions can be related back to impact processes on Mars and suggest feiite might also form on Earth.
Thermal equation of state of Fe3O4 magnetite up to 16 GPa and 1100 K
https://doi.org/10.2138/am-2022-8571Siersch et al. performed X-ray diffraction measurements on natural magnetite using resistive-heated diamond anvil cells up to 16 GPa and 1100 K. A thermal equation of state (EoS) was then fitted to the obtained data set. Moreover, it was possible to explore the structural evolution of magnetite in detail using single-crystal measurements. Over the studied pressure and temperature range, they found no evidence of a transformation from an inverse to a normal spinel structure. The EoS parameters obtained in this study will allow implementation into currently available databases for self-consistent thermodynamic modeling. In particular, the results are used to model and compare the sound wave velocities of a magnetite-bearing and magnetite-free Martian upper mantle assemblages. Siersch et al. observe that the incorporation of magnetite reduces the sound wave velocities, however, the magnitude of the effect is below the current seismic detection limit of the InSight mission on Mars at the low abundance of magnetite expected in the Martian mantle.
UHP eclogite from western Dabie records evidence of polycyclic burial during continental subduction
https://doi.org/10.2138/am-2022-8446(1) Eclogites from western Dabie have garnets showing systematic variation in grossular end-members. Based on phase equilibrium modeling using compositional isopleth thermobarometry, Xia et al. show that P first increased from 23.0 to 28.5 kbar, then decreased to 24.0 kbar, before increasing again to a maximum of 30.5 kbar concomitant with a small increase in T from 580 to 605 °C at the late prograde stage. These data are interpreted to indicate polycyclic burial and partial exhumation of eclogite during ongoing continental subduction. Furthermore, fluid contributing to widespread retrogression of eclogite during exhumation could be both internally and/or externally sourced.
CO2 quantification in silicate glasses using μ-ATR FTIR spectroscopy
https://doi.org/10.2138/am-2022-8477Schanofski et al. introduce a new and easy to use ATR-FTIR technique for the quantification of CO2 in silicate glasses. Clear linear dependencies of the normalized ATR peak heights of the CO2=related bands (carbonate as well as molecular CO2) on the CO2 content measured by independent methods (CSA, FTIR transmission spectroscopy) were found, and linear correlation coefficients for the quantitative determination of CO2 concentrations in leucititic and rhyolitic glasses were calculated. As previously demonstrated by Lowenstern and Pitcher (2013), μ-ATR FTIR is a convenient technique for measuring H2O (and now also CO2) concentrations in melt inclusions and partially crystallized samples due to the small penetration depth of the evanescent wave as well as the good spatial resolution.
Local structure determination of Zn-smectite
https://doi.org/10.2138/am-2022-8591The traditional XRD refinement method faces great challenges to obtain accurate crystal structures of smectites, due to their turbostratically disordered layers. In this study by Tao et al., a Zn-smectite (zincsilite) was synthesized under hydrothermal conditions. Its layer charge was generated by the octahedral Zn vacancies (defects), as shown by cationic exchange capacity and vacant site determination by Hofmann-Klemmen tests. Its intralayer structure was further determined by Pair Distribution Function (PDF) analysis. Based upon this, the whole crystal structure of Zn-smectite was built and optimized by Material Studio software and density functional theory (DFT) based calculations, respectively. This study shows that although turbostratic layers make building the 3-D crystal structure of smectites impossible, their local structure (such as intralayer structure) can be reliably determined by PDF methods.
A new UHP unit in the Western Alps: First occurrence of coesite from the Monviso Massif (Italy)
https://doi.org/10.2138/am-2022-8621Ghignone et al. report the occurrence of coesite in the meta-ophiolitic Monviso Massif (Piedmont Zone, Western Alps). Previous authors have inferred metamorphic indications (mostly coming from thermodynamic modeling) of the probable occurrence of coesite on the Monviso. Ghignone et al.’s finding confirm such a hypothesis, supported by a detailed description of coesite inclusion features, μ-Raman characterization, and a thermodynamic modeling (pseudosection) of the coesite-related metamorphic P-peak. The results have been discussed within the wide literature present on the topic (UHPM) and on the Monviso Massif, proposing new observations and interpretations. This finding is of fundamental importance for constraining geodynamic models in subduction-accretion systems and on the exhumation processes that created the present day setting of the Western Alps.
Mineral evolution and mineral niches of ammonium sulfates: The case of Pastora mine (Aliseda, Spain)
https://doi.org/10.2138/am-2022-8488Ammoniojarosite, (NH4)Fe3(SO4)2(OH)6, and tschermigite, (NH4)Al(SO4)2·12H2O, have been identified in the abandoned Pastora iron mine, Aliseda (Cáceres, Spain). These minerals appear in association with other sulfates, and they form as a result of the oxidation of pyrite contained in slates and sandstone host rocks. Their formation in Pastora mine is the result of complex processes in which acid mine drainage plays an important role. In addition, the NH4+ ion required for the precipitation of ammonium sulfates seems to be supplied by a large colony of greater horseshoe bats (Rhinolophus ferrumequinum) which roosts in Pastora mine. López et al. show that the formation of ammonium jarosite, tschermigite, and associated sulfate minerals in Pastora mine is controlled by a series of dissolution-precipitation reactions responding to seasonal variations in ambient temperature and relative humidity. Accordingly, the observed association of ammonium sulfates can be considered as a stage of mineral evolution in which new minerals crystallize and “adapt” to spatiotemporal changes in physicochemical conditions. In this sense, the concept of “mineral ecology” introduced by Hazen et al. (2015) is adequate to interpret the diversity and distribution of sulfate minerals in Pastora mine. Furthermore, Pastora mine provides a unique opportunity to further investigate the formation of ammonium sulfates, a group of minerals, to the best of their knowledge, had not been found in an iron mine to date. This investigation of the formation of hydrated sulfates, and particularly of ammonium sulfates, might be relevant to identify potential conditions for the existence of life.
Discrete late Jurassic Sn mineralizing events in the Xianghualing Ore District, South China: Constraints from cassiterite and garnet U-Pb geochronology
https://doi.org/10.2138/am-2022-8412The genetic relationships between the distal skarn and vein-type orebodies and the causative plutons are difficult to determine, which is compounded by the fact that the ages of distal skarns and their associated mineralization are rarely known to the required degree of accuracy. In this contribution by Wang et al., new cassiterite and garnet LA-ICP-MS U-Pb ages coupled with field observations and core-logging support the interpretation that there were two Sn mineralization events in the Xianghualing ore district, South China. The early event occurred between 160.3 +/- 1.1 and 158.1 +/- 1.2 Ma based on the cassiterite U-Pb geochronology, and was likely caused by a hidden granitic intrusion coeval with the nearby Jianfengling intrusion. This event only accounts for less than 5% of the total Sn resource. The main Sn mineralization event occurred between 154.1+/- 2.8 and 151.1 +/- 1.6 Ma based on cassiterite and garnet LA-ICP-MS U-Pb ages and is genetically related to the Laiziling granitic intrusion (zircon U-Pb age of 152.8 +/- 1.2 Ma). The main Sn mineralization event contributes approximately 95% of the total Sn resources. Our new geochronological results indicate a superimposed Sn mineralizing system in the Xianghualing ore district, which is of great significance to regional Sn exploration.
Ryabchikovite, CuMg(Si2O6), a new pyroxene group mineral, and some genetic features of natural anhydrous copper silicates
https://doi.org/10.2138/am-2022-8620Ryabchikovite, CuMg(Si2O6), is the first natural pyroxene with species-defining copper. This find by Shchipalkina et al. shows that pyroxenes can be a principal silicate host of Cu2+ in special geological settings, characterized by high oxygen fugacity and gas transport of metals such as Cu, Zn, Pb, and Mo in a temperature range of 400 to 900 °C. Review of previous results and their new data reveals the formation of H-free, Cu-enriched silicates due to high oxygen fugacity coupled with relatively low pressures (close to atmospheric) and, presumably, Cu-halides as transport agents during silicate crystallization in post-volcanic systems where the sulfur is fixed mainly in the sulfate form, and local supersaturation in Cu occurs during the deposition of silicates.
June 2023
A shallow salt pond analog for aqueous alteration on ancient Mars: Spectroscopy, mineralogy, and geochemistry of sediments from Antarctica's dry valleys
https://doi.org/10.2138/am-2022-8381Understanding mineral formation on Mars is critical to understanding the planet's geochemical evolution, climate, and history of liquid water, as well as potential habitability. However, the formation processes behind mineral assemblages observed on Mars remain elusive. Here, Burton et al. examined aqueous alteration products developed in shallow sediments at a transient Antarctic brine pond as an example of the formation of clays, sulfates, and chlorides in extremely cold, water-poor, Mars-like settings.
Incorporation of chlorine in nuclear waste glasses using high-pressure vitrification: Solubility, speciation, and local environment of chlorine
https://doi.org/10.2138/am-2022-8599Chlorine is one of the most important halogens involved in magmatic systems, representing a major troublesome element in the immobilization of nuclear waste, for which, however, there is a lack of fundamental understanding. Jolivet et al. investigated how chlorine dissolves in the structure of high-pressure borosilicate glasses using advanced spectroscopic techniques XPS and XAS. The speciation and the local environment of chlorine species have been determined and clarified. Furthermore, they propose an approach for predicting the chlorine solubility in a wide range of glass compositions.
Experimental constraints on miscibility gap between apatite and britholite and REE partitioning in an alkaline melt
https://doi.org/10.2138/am-2022-8535Stepanov et al. performed synthesis of britholite and apatite from felsic melt, which showed that the addition of NaCl to the granite melt could have a pronounced effect on the behavior of REE. The change of the mineral association from monazite to apatite + britholite with the addition of NaCl illustrates the importance of halogens. REE entered the apatite structure via REE-Si substitutions. The results have implications for the interpretation of the phosphate associations in alkaline volcanic and plutonic rocks.
Thermal expansion of minerals in the tourmaline supergroup
https://doi.org/10.2138/am-2022-8580The minerals in the tourmaline supergroup are widespread in Earth's crust, typically occurring in granites and granitic pegmatites, as well as in certain sedimentary and metamorphic rocks. In addition, tourmalines are the primary boron-bearing minerals in the earth. Tourmaline thermal-expansion data, therefore, are essential to the thermodynamic modelling not only of pegmatitic environments, but of all high-temperature B-rich mineral assemblages. Here, Hovis et al. provide an updated high-T dataset for the tourmaline mineral supergroup that will serve thermodynamic databases and will also be a valuable tool for better understanding tourmaline physical properties. Because present data demonstrate a similarity in thermal expansion among a wide variety of tourmaline compositions, there is the possibility of using end-member thermal expansion data for compositions that deviate significantly from those studied here.
Viscosity of Earth's inner core constrained by Fe-Ni interdiffusion in Fe-Si alloy in an internal-resistive-heated diamond anvil cell
https://doi.org/10.2138/am-2022-8541Diffusivity in iron alloys at high pressures and temperatures imposes constraints on transport properties of the inner core, such as viscosity. Because silicon is among the most likely candidates for light elements in the inner core, the presence of Si must be considered when studying diffusivity in the Earth's inner core. Park et al. conducted diffusion experiments under pressures up to about 50 GPa using an internal-resistive-heated diamond anvil cell (DAC) that ensures stable and homogeneous heating compared with a conventional DAC heating method and thus allows them to conduct more reliable diffusion experiments under high pressures. They determined the coefficients of Fe-nickel (Ni) interdiffusion in the Fe-Si 2 wt.% alloy. The upper limit of the viscosity of the inner core inferred from these results is low, indicating that the Lorentz force is a plausible mechanism to deform the inner core.
The distribution of carbonate in apatite: the environment model
https://doi.org/10.2138/am-2022-8389The location of carbonate in the structure of carbonated apatite, the closest analog to the inorganic portion of bones and teeth, is of importance in the biological function of this compound, especially the possibility of the involvement of structural carbonate in acid-base regulation. Yoder et al. demonstrate that the environment model originally proposed by Fleet (2017) for apatites prepared at high temperature and pressure is also valid for apatites produced synthetically or biologically at ambient pressures and low temperatures. The model suggests that considerably more carbonate is sequestered in the apatite channel than was previously thought and brings a greater focus on the roles of both types of carbonate in the biological function of carbonate in bone mineral.
Low-temperature crystallography and vibrational properties of rozenite (FeSO4·4H2O), a candidate mineral component of the polyhydrated sulfate deposits on Mars
https://doi.org/10.2138/am-2022-8502The current generation Mars Rovers use Raman spectroscopy to search for minerals that are thought to have formed early in the martian history, when Mars was able to sustain surface waters. Raman spectroscopy is a powerful analytical tool but relies on accurate fingerprint reference spectra to identify minerals. Meusburger et al. show that the Raman fingerprint of rozenite was misinterpreted in the literature and suggest a workflow for the construction of a reliable Raman spectroscopic database for planetary exploration.
Hydrothermal fluid signatures of the Yulong porphyry Cu-Mo deposit: Clues from the composition and U-Pb dating of W-bearing rutile
https://doi.org/10.2138/am-2022-8453Rutile enriched in V, W, Mo, Sn and Sb can be used as a geochemical fingerprint of a hydrothermal fluid associated with mineralization. Chen et al. investigated the geochemical composition and U-Pb dates of hydrothermal rutile from the Yulong porphyry Cu-Mo deposit in east Tibet, China. The analysis of rutile crystals indicates that the patchy or sector zonation imparts a marked control on the distribution of W, which substitutes for Ti by a coupled substitution with the trivalent cations in the rutile. In situ LA-ICP-MS U-Pb dating shows that the age of the W-bearing rutile grains overlaps the previously reported molybdenite Re-Os ages and provides the ability to directly determine the age of the Cu-Mo mineralization. The mechanism of the enrichment of tungsten is effectively driven by the halogen-rich (F, Cl) aqueous fluids during hydrothermal mineralization in the Yulong deposit. Consequently, the chemical or isotopic composition recorded in rutile provides comprehensive information for better understanding of the nature of hydrothermal fluids during the deposition of porphyry deposits.
Magnetic contributions to corundum-eskolaite and corundum-hematite phase equilibria: A DFT cluster expansion study
https://doi.org/10.2138/am-2022-8584Magnetic contributions to mixing phase diagrams are challenging to compute, yet may have significant impact on phase stability. Pope et al. develop a protocol that includes spin orientation as an additional configurational component within multi-component cluster expansions between magnetic and non-magnetic metal oxide alloys. Two systems are compared, one where magnetic contributions to the calculated phase diagram are inconsequential (corundum-eskolaite) and one where they are absolutely essential (corundum-hematite) to accurately determine the solvus.
Microchemistry and magnesium isotope composition of the Purang ophiolitic chromitites (SW Tibet): New genetic inferences
https://doi.org/10.2138/am-2022-8392Xiong et al. investigated the textural and compositional characteristics of silicate sulfide and exotic UHP-SuR mineral inclusions in Purang ophiolitic chromitite grains, suggesting a complex evolution of this Jurassic ophiolitic massif in SW Tibet. Evolution of these rocks included subduction of lithospheric slabs into the transition zone, crystallization of magnesiochromite and some massive chromitite at depth, incorporation of UHP and highly reduced phases into the chromitites, and entrapment of oceanic lithospheric slabs in a supra-subduction zone (SSZ) setting, where they undergo varying degrees of reaction with hydrous, SSZ melts.
Pyrite geochemistry in a porphyry-skarn Cu (Au) system and implications for ore formation and prospecting: Perspective from Xinqiao deposit, Eastern China
https://doi.org/10.2138/am-2022-8527Xiao et al. investigated the geochemistry of pyrite in a porphyry-skarn Cu (Au) system in Xinqiao deposit, eastern China. Geological, trace element and S isotope analyses show that the colloform pyrite formed during early skarn mineralization. Colloform pyrite was produced by rapid decompression that triggered fluid boiling and cooling when the Cretaceous magmatic-hydrothermal fluid flowed along the Devonian-Carboniferous unconformity. Stratabound sulfide ore is sourced from Cretaceous magmatic-hydrothermal fluids, and formed from multi-stage magmatic-hydrothermal pulses. Co and Ni in pyrite increase towards porphyry and skarn ore, whereas As, Sb, Pb, Ag and Bi are enriched in pyrite in distal stratabound ore. The results indicate pyrite chemistry can be effective in discriminating the genesis of different deposit types related to porphyry-skarn systems and can potentially be used as a vectoring tool during exploration in the MLYB and elsewhere.
UV/Vis single-crystal spectroscopic investigation of almandine-pyrope and almandine-spessartine solid solutions: Part I. Spin-forbidden Fe2+,3+ and Mn2+ electronic-transition energies, crystal chemistry and bonding behavior
https://doi.org/10.2138/am-2022-8499Investigation of the solid-solution behavior of aluminosilicate garnets is a long, continuing work in progress and much still needs to be studied. A fundamental problem is to understand the nature of local structural heterogeneity that arises through the exchange of different atoms of varying sizes and electronic properties. Geiger et al. measured natural almandine-pyrope, {Fe2+3x,Mg3-3x}[Al2](Si3)O12, and almandine-spessartine, {Fe2+3x,Mn2+3-3x}[Al2](Si3)O12, crystals using UV/Vis/NIR (~29000 to 10000 cm-1) optical absorption spectroscopy. The spectra and changes in energy of a number of Fe2+ and Mn2+ spin-forbidden electronic transitions were analyzed as a function of garnet composition across both binaries in order to understand the underlying bonding behavior.
Single-crystal UV/Vis optical absorption spectra of almandine-bearing and spessartine garnet: Part II. An analysis of the spin-forbidden bands of Fe2+, Mn2+, and Fe3+
https://doi.org/10.2138/am-2022-8500Almandine and spessartine are common occurring aluminosilicate garnets with the ideal end-member crystal-chemical formulas {Fe2+3}[Al2](Si3)O12 and {Mn2+3}[Al2](Si3)O12. Garnet is a remarkable phase in several ways and much research in the Earth Sciences has focused on the different rock-forming species. Taran et al. demonstrate that a crystal-chemical aspect of significance is the eight-fold or triangular dodecahedral coordination of Fe2+ (d6) and Mn2+ (d5) by oxygen anions and the various physical properties arising from this. Different types of electronic transitions in garnet, as expressed in the NIR/Vis/UV regions of the electromagnetic spectrum, can result from the Fe2+ and Mn2+ cations. The transitions can be studied by optical absorption spectroscopy, which yields information on chemical bonding.
Single-crystal UV/Vis absorption spectroscopy of aluminosilicate garnet: Part III. {Fe2+} + [Fe3+] {Fe3+} + [Fe2+] intervalence charge transfer
https://doi.org/10.2138/am-2022-8756The various intervalence charge transfer (IVCT) mechanisms that can occur in silicate garnets, with a general crystal-chemical formula {X3}[Y2](Z3)O12, are not fully understood. Geiger and Taran conducted spectroscopic measurements on aluminosilicate garnets, with a focus on understanding IVCT behavior in almandine-rich, spessartine-rich and grossular-rich crystals and one intermediate composition almandine-pyrope garnet. The authors also reviewed and analyzed various possible IVCT mechanisms in both Ca-Ti-bearing and aluminosilicate garnets. The goal is to achieve a better and more complete understanding of the electronic transition behavior of an important rock-forming mineral group.
A novel method for experiments in a one-atmosphere box furnace
https://doi.org/10.2138/am-2022-8867This letter by Linzerova et al. presents a new high-temperature experimental method for one-atmosphere box furnace, utilizing commercially available low-cost materials. The method was designed to facilitate textural analysis of voluminous samples, with no need to use noble-metal capsules.
May 2023
Eu speciation in apatite at 1 bar: An experimental study of valence-state partitioning by XANES, lattice strain, and Eu/Eu* in basaltic systems
https://doi.org/10.2138/am-2022-8388Tailby et al. describe three new redox calibrations for apatite in basaltic systems. The first calibration includes a methodology that uses the intensity of a negative Eu anomaly relative to neighboring Sm and Gd. The second measures the intensity of the negative Eu anomaly relative to the lattice strain. The third employs a direct XANES measurement on the Eu L3 XANES spectral features.
The effect of composition on chlorine solubility and behavior in silicate melts
https://doi.org/10.2138/am-2022-8450Thomas and Wood used a sliding chlorine buffer (a mixture of AgCl/AgI and Ag) to determine the fugacities (and activities) of chlorine in many silicate melts. They show that for a wide range of compositions, from basalt, phonolite, andesite, dacite, and rhyolite that Henry's Law is obeyed for the Cl contents of interest in natural volcanic systems. They quantified the effects of different oxides, CaO, FeO, etc., on Cl solubility and determined conditions of NaCl saturation at high pressure and temperature.
High-temperature phase relations of hydrous aluminosilicates at 22 GPa in the AlOOH-AlSiO3OH system
https://doi.org/10.2138/am-2022-8429Aluminous hydrous phases are key minerals for understanding the Earth's deep water cycle since they have been found as inclusions in superdeep diamonds. In this study, Takaichi et al. investigated the phase relations of hydrous aluminosilicates under pressure conditions corresponding to Earth's interior. They found the formation of two unknown hydrous aluminosilicates. These results indicate that various hydrous aluminosilicates can widely exist within subducting crustal materials in the mantle transition zone to the lower mantle.
Crystallization of spinel from co-existing silicate and sulfide immiscible liquids: an equilibrium case with post-cumulus reactions
https://doi.org/10.2138/am-2022-8473Mao et al. find that the Kalatongke deposit is an example of where the same mineral phase from two immiscible liquids exhibits approximately equivalent compositions.They determined that the chalcophile element concentration in Cr-magnetite/magnetite of magmatic Ni-Cu deposits results from element exchange with the coexisting sulfide melt, and that the texture and composition of spinel may serve as an indicator for not only the compositional evolution of silicate/sulfide melts but also the percolation and enrichment history of sulfide liquid.
X-ray absorption spectroscopy study of Mn reference compounds for Mn speciation in terrestrial surface environments
https://doi.org/10.2138/am-2022-8236Zahoransky et al. analyzed a broad range of inorganic and organic Mn compounds by bulk Mn K-edge X-ray absorption spectroscopy (XAS); they evaluated the accuracy of XAS to quantify Mn oxidation states, and explored spectral similarities between and within different Mn species groups. Their study provides the first comprehensive framework for the analysis and interpretation of bulk Mn XAS spectra of natural samples and, thus, for understanding the role of Mn in environmental processes.
Heterogeneous and retarded phase transformation of ferrihydrite on montmorillonite surface: The important role of surface interactions
https://doi.org/10.2138/am-2022-8520These novel findings enrich our knowledge of the phase transformation characteristics of ferrihydrite under various environmental conditions and advance the understanding of the importance of mineral surface interactions in stabilizing nanominerals and mineral nanoparticles in nature. The findings by Wei et al. also have important implications for assessing mineral composition and the associated ecological environment in wildfire-affected regions. The occurrence of maghemite as an intermediate during the transformation of ferrihydrite in the heteroaggregates also provides an explanation of the origin of magnetism in soils.
Atomic-scale characterization of the oxidation state of Ti in meteoritic hibonite: Implications for early solar system thermodynamics
https://doi.org/10.2138/am-2022-8311Calcium-aluminum-rich inclusions (CAIs) in meteorites are the oldest solar system solids. Among the CAI phases, hibonite, nominally CaAl12O19, holds particular interest because it is among the first few phases to form from the solar gas and it can incorporate significant amounts of Ti in both Ti3+ and Ti4+. The relative amounts of these cations can reflect the redox conditions under which the grain formed or last equilibrated. Here Zanetta et al. develop a new method for the quantification of Ti oxidation states using electron energy-loss spectroscopy (EELS) in order to apply it to a hibonite grain sampled in the NWA 5028 CR2 chondrite. The aim is to constrain the conditions under which the hibonite grain formed and demonstrate viability of the EELS technique.
Structural behavior of C2/m tremolite to 40 GPa: A high-pressure single-crystal X-ray diffraction study
https://doi.org/10.2138/am-2022-8278The metastability/compression behavior of tremolite is studied by Ott et al. up to ~40 GPa. The metastable persistence of C2/m tremolite implies that chemically bound water may be carried much deeper than 90 km in steeply subducting cold slabs. This broad metastability range and an increasingly isotropic high-P compressibility contrasts with other clinoamphibole observations, implying that the size of cations interconnecting the I-beams of the clinoamphibole structure exerts a strong control on metastability.
Optimizing Raman spectral collection for quartz and zircon crystals for elastic thermobarometry
https://doi.org/10.2138/am-2022-8423This study systematically assesses the reproducibility of Raman spectra collected on quartz and zircon crystals and quantifies uncertainties in calculations of entrapment pressures and temperatures as determined using elastic thermobarometry. Three key new contributions include identifying different types of peak drift that operate on different time scales, assessing the reproducibility of different methods for calculating entrapment pressures, and proposing a new method for monitoring peak drift using an external light source. Some methods are resistant to peak drift errors (propagated errors much less than ~1 kbar), while others are highly sensitive (up to several kbar). Ultimately, comprehensive recommendations are provided for analysis that should improve intralaboratory reproducibility and interlaboratory comparisons.
Measuring H2O concentrations in olivine by secondary ionization mass spectrometry: Challenges and paths forward
https://doi.org/10.2138/am-2022-8247Trace concentrations of water in olivine strongly affect diverse mantle and magmatic processes. Accurate measurements of these low concentrations are challenging and even more so due to a ~40% disagreement between the two main studies that have produced independent water calibrations. In this study, Towbin et al. resolved the differences between these calibrations and offer recommendations for an improved ion microprobe calibration using a readily available set of standards. In light of their finding, they reevaluate published partition coefficients for water between olivine and melt. By revising the measured olivine concentrations, they improve agreement between experimentally determined partition coefficients and those determined from olivine-hosted melt inclusions.
Arsenic clustering in arsenian pyrites: A combined photoemission and theoretical modelling study
https://doi.org/10.2138/am-2022-8415The photoemission spectra from samples of arsenian pyrite collected by Mahjoub et al. using synchrotron soft X-ray spectroscopy are compared to a series of first principles simulations of the four experimentally observed surfaces of pyrite in the presence of As atoms on the top layers that substitute for superficial S. A comparison between the spectrum of bulk As-3d in the samples with its bulk counterpart in arsenopyrite revealed a 0.6 eV shift towards lower binding energies. The calculated partial charges of atoms located at the surface of arsenian pyrite indicated that while the electron density on the As atom of As-S dimers in arsenian pyrite is less negative than the As in bulk arsenopyrite, it is more negative than the As atom of As-As dimers, which were only seen in the superficial As clusters. This firmly validated the description of As presence in arsenian pyrite as local clusters inducing localized lattice strain due to increased bond distances.
High-pressure electrical conductivity and elasticity of iron-bearing δ-AlOOH
https://doi.org/10.2138/am-2022-8393In this contribution by Su et al., the electrical conductivity and elasticity were determined for δ-(Al,Fe)OOH with 5 and 48 mol.% FeOOH at pressures up to 75 GPa. They found that conductivity of δ-(Al,Fe)OOH may be slightly affected by high iron content and spin transition at high pressure, and concluded that subduction of Fe-bearing δ-AlOOH may account for some high conductivity regions in the lower mantle, e.g., the North Philippine Sea slab.
Nudged Elastic Band calculations of the (4H)SiX hydrogarnet type defect in Mg2SiO4 forsterite
https://doi.org/10.2138/am-2022-8449The Nudged Elastic Band (NEB) method is used by Poe and Perna to investigate configurational transformations of the hydrogarnet defect in forsterite. Reactions involve rotational or torsional movements of OH bonds without breaking any strong chemical bonds, and activation energies do not exceed 0.6 eV. In some cases, reactions are accompanied by significant displacement of the (4H) center of mass to suggest a low-temperature means of promoting charge transport without requiring chemical dissociation of the hydrogarnet defect species.
Mn substitution and distribution in goethite and influences on photocatalytic properties from first-principles calculations and experiments
https://doi.org/10.2138/am-2022-8182This study by Liu et al. provides one simulation frame and reveals the distribution pattern of Mn impurities in goethite, i.e., the Mn cations prefer to distribute within the cation layer approximately parallel to the (001) plane. They also predict that there may be Mn-rich nano-scale clusters or lamellae in Mn-substituted goethite even with as low Mn content as 3-4 mol% based on the phase diagram and band gap analysis. The incorporation of impurity ions can significantly decrease the band gap of goethite because of the involvement of impurity bands, here, Mn substitution enlarges the potential photocatalytic range of goethite to the whole visible and partial near-infrared light. Because visible light is the main solar radiation reaching the Earth, the enlargement of photocatalytic range provides more solar energy for photocatalyze. Doped goethite may be involved in various photochemical processes, for instance, solar-induced generation of reactive oxygen species (ROSs). ROSs can initiate subsequent degradation of organic compounds, which may be one possible reason to explain abiotic oxidation of organic compounds in desert on Earth and the absence of organics on Mars. Such photocatalytic ability probably can affect the element cycling to a non-negligible extent on both early and modern terrestrial planets.
Incorporating previously neglected oxygen associated with ferric iron into matrix corrections of microprobe data from cubic and rhombohedral Fe-Ti oxides
https://doi.org/10.2138/am-2022-8447Dungan et al. address a long-term error in the application of 'ZAF' matrix corrections to iron-rich minerals, particularly those with high ferric iron. This mistake has prevented up to ~7 wt% oxygen from being considered as an absorbing element for Fe. Redressing this omission leads to increases in reported FeO+Fe2O3 up to 1.1 wt% for end-member Fe3O4. The routine for accomplishing this as a part of microprobe data collection is now available to users of Probe for EPMA software. Old or new data not collected with this version of the program may be recalculated with CalcZAF, an app which is available on-line.
Recycled carbonates in the mantle sources of natural kamafugites: A zinc isotope perspective
https://doi.org/10.2138/am-2022-8382Kamafugites are extremely silica-undersaturated melts that are difficult to produce by partial melting of volatile-free peridotites, but can be experimentally yielded with CO2 being added. Nevertheless, there is still rare evidence for a CO2-rich mantle source and possible recycled carbonates in the source of natural kamafugites. Here Ma et al. take Cenozoic kamafugites from the West Qinling orogen, China, as an example to address the origin of this rare volcanic rock. Their results indicate that the West Qinling kamafugites represent the products of low-degree partial melting of a carbonated (CO2-rich) peridotite source, which provides evidence for an important role of recycled carbonates in the origin of natural kamafugite suites.
Raman analysis of octocoral carbonate ion structural disorder along a natural depth gradient, Kona coast, Hawai'i
https://doi.org/10.2138/am-2022-8406Calcitic octocorals collected along a natural depth gradient were analyzed by Conner et al. using Raman spectroscopy to better understand the individual effects of environmental and growth rate kinetic factors on carbonate ion structural disorder. Structural disorder displayed unique trends across the environmental gradient and across varying skeletal growth rates, respectively. The results of this study have implications for Mg paleo-proxies and potentially quantifying biogenic calcite solubility using Raman.
Memorial of Charles Wilson Burnham, 1933-2021
https://doi.org/10.2138/am-2023-1016Erratum
https://doi.org/10.2138/am-2023-E10854April 2023
Mineralogy and geochemistry of hot spring deposits at Namafjall, Iceland: Analog for sulfate soils at Gusev crater, Mars
https://doi.org/10.2138/am-2022-8364Carson et al. report on the mineralogy and geochemistry of hot spring deposits at Namafjall in northern Iceland. The acidic hot springs produce patterns of mineralization in zones from most reducing (Fe-sulfide bearing) to most oxidized (Fe3+-sulfate and oxide bearing), with intermediately oxidized phases (elemental sulfur, Fe2+-sulfates) found in between. The Fe-sulfate assemblage is similar to that interpreted for the Paso Robles soils at the Mars Exploration Rover (MER) Spirit landing site, which could have undergone a similar process.
The iron spin transition of deep nitrogen-bearing mineral Fe3N at high pressure
https://doi.org/10.2138/am-2022-8434Lv and Liu performed in-situ X-ray emission spectroscopy measurements on Fe3N up to 45.8 GPa at room temperature. The results show that 1) pressure-induced spin transition of iron in Fe3N starts at relatively low pressures and completes at ~28.7 GPa; (2) the iron spin transition pressure is highly related to the nitrogen concentration of hexagonal iron nitrides; and (3) the identity and concentration of light elements, together with crystal structure, take charge of the spin transition pressure of iron-rich alloys.
Hydrogen occupation and hydrogen-induced volume expansion in Fe0.9Ni0.1Dx at high-PT conditions
https://doi.org/10.2138/am-2022-8348The density of the Earth's core is notably lower than that of iron-nickel alloy; hydrogen (H) is one of the most promising elements accounting for the density deficit. This study by Shito et al. first clarified the hydrogenation mechanism of FeNi alloy at high pressures and temperatures up to 12 GPa and 1000 K by in-situ X-ray diffraction and neutron diffraction measurements. Their results suggest that only 10% of nickel in iron drastically changes the behaviors of H. The maximum H content in the Earth's inner core is estimated to be one to two times the amount of H in the oceans.
Volumes and spin states of FeHx: Implication for the density and temperature of the Earth's core
https://doi.org/10.2138/am-2022-8237This study by Yang et al. show that: (1) Hhdrogen stabilizes the magnetic properties of fcc Fe to higher pressure; (2) spin transition and volume collapse of fcc FeH are predicted at ~40 GPa; (3) the HS to LS transition in fcc FeH dramatically increases its elastic modulus and sound velocities.; and (4) the presence of hydrogen induces a relatively low core temperature.
Thermodynamic characterization of synthetic lead-arsenate apatites with different halogen substitutions
https://doi.org/10.2138/am-2020-7452Puzio et al. determined the enthalpies of formation (ΔH°f,el) of lead arsenate apatites (synthetic analogs of mimetites: Pb5(AsO4)3X, X= OH, Cl, Br, I) using high-temperature oxide melt calorimetry. The values of ΔH°f,el exhibit a linear increase with the molar mass of the apatite and show a strong linear correlation with the electronegativity of the halogen anion. On the basis of this observation, the values of ΔH°f,el for F-mimetite Pb5(AsO4)3F and I-pyromorphite Pb5(PO4)3I, which were previously unknown, were calculated from regression. The theoretically predicted values agree well with the observed systematic trends and can be used with confidence in thermodynamic calculations pending experimental confirmation.
Structural changes in shocked tektite and their implications to impact-induced glass formation
https://doi.org/10.2138/am-2022-8149Sekine et al. investigated shock-recovered tektites using XRD, Raman, and Ti K-edge XAFS methods. Tektite holds the local structures around Ti in melt even after annealing in the release process. Some tektites with Ti3+ indicate intensive impact heating over 3000 K and reduction. Impact glasses observed near impact craters suggest mild or oxidizing impact conditions.
Characterization of vandenbrandeite: A potential alteration product of spent nuclear fuel
https://doi.org/10.2138/am-2022-8272Frankland et al. discuss the potential of using laser-based spectroscopic techniques to characterize the alteration products of spent nuclear fuel in settings where the use of more traditional analytical methods is impracticable. This is demonstrated through the investigation of two copper-bearing uranyl oxy-hydroxide phases, which may form on the surface of the spent fuel should a KBS-3 type copper canister fail or suffer from initial defects. In this study, two vandenbrandeite samples were characterized using multiple-wavelength laser Raman and luminescence spectroscopy, with the results interpreted using ab initio modeling. As alteration phases may be poorly crystalline or amorphous, the two vandenbrandeite specimens were chosen to exhibit varying degrees of crystallinity. Good agreement was obtained between experimental and simulated Raman spectra.
The NaCl-CaCO3 and NaCl-MgCO3 systems at 6 GPa: Link between saline and carbonatitic diamond forming melts
https://doi.org/10.2138/am-2022-8403Carbonatitic and saline melts or high-density fluids (HDFs) are the most common among inclusions in natural diamonds. However, a genetic link between saline and carbonatitic endmembers remains controversial and experimentally unresolved. The results obtained by Shatskiy et al. do not support the hypothesis of the formation of chloride and carbonate melts as a result of liquid immiscibility. The continuous trend in composition from carbonatitic to saline HDFs entrapped as microinclusions in natural diamonds can be explained by (1) fractional crystallization of the Cl-bearing carbonatite melt; (2) melting of carbonated mantle domains induced by saline HDF; and (3) reduction of Cl-bearing carbonatitic melt.
Single-crystal elasticity of (Al,Fe)-bearing bridgmanite up to 82 GPa
https://doi.org/10.2138/am-2022-8435Fu et al. report high-pressure single-crystal elasticity of (Al,Fe)-bearing bridgmanite up to 82 GPa, corresponding to the mid-lower mantle depth. The results are used in thermoelastic modeling to evaluate velocity profiles of lower-mantle mineral aggregates at relevant pressure-temperature conditions and then applied to help better constrain the lower-mantle composition.
Single-crystal X-ray diffraction of fluorapatite to 61 GPa
https://doi.org/10.2138/am-2022-8410Rucks et al. used synchrotron-based single-crystal X-ray diffraction coupled with diamond anvil cell techniques to probe the structural response of fluorapatite under room temperature compression to 61 GPa. Previous investigations of fluorapatite are limited to relatively low pressures. In this study, the hexagonal fluorapatite structure was examined up to 32.4 GPa, after which point the structure transforms to a triclinic phase. This is the first observation of a pressure-induced triclinic apatite. Additionally, the authors provide Birch-Murnaghan equation of state parameters for both phases.
Iron and aluminum substitution mechanism in perovskite phase in the system MgSiO3-FeAlO3-MgO
https://doi.org/10.2138/am-2022-8457From multi-anvil high-pressure experiments, the open access paper (available to all readers) by Ishii et al. found that the A-site vacancy component of Fe2/3SiO3 may exist by 1-2 mol% at 1700-2000 K in FeAlO3-bearing MgSiO3 perovskite phase, whereas 1 mol% of the oxygen vacancy component of MgFeO2.5 appears at higher temperatures. The A-site vacancy component might be more important than the oxygen vacancy component for the defect chemistry of bridgmanite in slabs and for average mantle conditions when the FeAlO3 charge-coupled component is dominant.
Ultrasonic studies of alkali-rich hydrous silicate glasses: Elasticity, density, and implications for water dissolution mechanisms
https://doi.org/10.2138/am-2022-8451Liu et al. determined the compressional and shear wave velocities of three series of alkali-rich hydrous silicate glasses with up to 5.25 wt% H2O using ultrasonic interferometry at ambient conditions. The results show that the sound velocities and calculated elastic properties are nearly linear functions of dissolved water content. On the other hand, the addition of Na2O content might weaken the effect of water on acoustic velocity. In addition, the similar behavior of Al-free and Al-bearing glasses implies that water depolymerizes melts in both Al-free and Al-bearing systems. The decrease of water content derivatives of the Poisson's ratio &91;∂(v)/∂(XH2O)&93; with increasing Al/(Al + Si) indicates that water interaction with Al-O bonds depolymerizes peralkaline silicate melts less effectively than Si-O bonds with increasing Q4(4Al) abundance. Hence, water may prefer to interact with Si-O bonds rather than Al-O bonds in peralkaline silicate systems.
Gadolinium-dominant monazite and xenotime: Selective hydrothermal enrichment of middle REE during low-temperature alteration of uraninite, brannerite, and fluorapatite (the Zimna Voda REE-U-Au quartz vein, Western Carpathians, Slovakia)
https://doi.org/10.2138/am-2022-8418Ondrejka et al. investigated a very unusual assemblage of Gd (MREE)-rich minerals formed during the interaction of primary hypogene ore minerals with low-temperature hydrothermal fluids. From detailed microanalytical and spectroscopic data, along with textural observations, they provide evidence for gradual remobilization-fractionation of U and REE in aqueous media. This study also demonstrated the continuous evolution of the W-type tetrad effect during the progressive crystallization of monazite towards Ce→Nd→Sm→Gd rich species and the evolution of xenotime towards Y→Gd species. Moreover, this study shows that low-T hydrothermal alteration and replacement reactions of MREE-selective, but nominally REE-free minerals, e.g., uraninite, brannerite, and fluorapatite, can produce an enhanced MREE signature in chemically-closed systems.
Nucleation of Th-rich cerianite on halloysite surface in a regolith-hosted rare earth elements deposit in South China
https://doi.org/10.2138/am-2022-8391Cui et al. (1) revealed the effect of halloysite on Ce enrichment and nucleation in a weathering granite profile; (2) characterized aggregates of Th-rich cerianite nano-particles with a preferred orientation on the halloysite surface; and (3) interpreted the selective nucleation of cerianite on basal surface of halloysite based on lattice match analyses.
MSA Presentations of Awards 2022
New Mineral Names
March 2023
Mineralogy and bulk geochemistry of a fumarole at Hverir, Iceland: Analog for acid-sulfate leaching on Mars
https://doi.org/10.2138/am-2022-8363In this paper by Carson et al., an Icelandic fumarole interacting with basalt produced a pattern of alteration minerals (silica, sulfates, oxides, and clays) that informs their understanding of potential hydrothermal alteration on Mars. The high silica and titanium acid-sulfate leached deposit, coupled with only minor sulfates, could be analogous to silica-rich soils at Gusev crater on Mars, while more distal samples contain varied sulfates and oxides, potentially relevant to the formation of Martian jarosite and hematite.
The crystal structure and chemistry of natural giniite and implications for Mars
https://doi.org/10.2138/am-2022-8138Giniite is a mineral that may occur on Mars and be important as a resource for future extended human missions to Mars. However, there are little data available on the mineral and the last characterization of the structure was over 40 years ago. Here Adcock et al. revisit the chemistry and crystal structure of natural giniite. Their results improve upon what was previously known and support the potential of giniite as a potential in situ resource for fuel, energy, and water on Mars.
Solid solution of CaSiO3 and MgSiO3 perovskites in the lower mantle: The role of ferrous iron
https://doi.org/10.2138/am-2022-8356MgSiO3 and CaSiO3 perovskites have near identical chemical and crystal structures and thus at some point must form a single phase in the lower mantle. By using ab-initio calculations Zhang et al. investigated Fe partition coefficients between perovskite phases and the role of iron on their mixture. They found iron increases the miscibility of Ca and Mg perovskite phases and reduces the mixing temperature, but this effect is highly nonlinear. Iron is found not a significant factor in determining the phases of the lower mantle and there is no strong thermodynamic gradient to push iron into a mixed phase.
Secondary ion mass spectrometer analyses for trace elements in glass standards using variably charged silicon ions for normalization
https://doi.org/10.2138/am-2022-8362Microanalyses using secondary ion mass spectrometry can detect elements (e.g., Li, Be, B) at low concentrations (e.g., <µg/g). Matrix effects appear to be minimized if (1) the ion signal of interest is normalized to Si+ (as opposed to Si2+ or Si3+) and (2) low-energy (maximum signal) ions are selected. Carlson and Hervig explored the advantages and disadvantages of using multiply charged Si and Al ions, as well as O+ ions, to create calibration curves for application to different matrices.
Raman shifts of c-BN as an ideal P-T sensor for studying water-rock interactions in a diamond anvil cell
https://doi.org/10.2138/am-2022-8372Ren et al. report Raman spectra of cubic boron nitride (c-BN) at simultaneous high pressure and high temperature (>700 K) conditions. The Raman shift of the c-BN is shown to be a coupled function of pressure and temperature and cannot be ignored, as was assumed in previous studies. Ren et al. propose that the Raman shift of c-BN is an ideal P-T sensor for diamond anvil cell experiments, especially for studying water-rock interactions in subduction zones.
Resetting of the U-Pb and Th-Pb systems in altered bastnasite: Insight from the behavior of Pb at nanoscale
https://doi.org/10.2138/am-2022-8318The study by Zhang et al. highlights that because of the different orders of magnitude between the U and Th contents in bastnasite, the mobilization of radiogenic and non-radiogenic Pb during alteration may have significantly different impacts on the U-Pb and Th-Pb system.
X-ray diffraction reveals two structural transitions in szomolnokite
https://doi.org/10.2138/am-2022-8147This work by Pardo et al. contributes to high-pressure experimental data on hydrated sulfates relevant to planetary interiors on Earth, Mars, and the icy satellites. They find two crystal structure transitions occurring in szomolnokite (FeSO4.H2O) under 20 GPa. Using equation of state fitting with existing data, they illustrate that the relative structural and elastic properties of szomolnokite and other hydrated sulfate phases have complex chemical and pressure-dependent behaviors.
Contamination of heterogeneous lower crust in Hannuoba tholeiite: Evidence from in situ trace elements and strontium isotopes of plagioclase
https://doi.org/10.2138/am-2022-8254Su et al. found that 87Sr/86Sr of the studied plagioclase crystals varied from 0.70333 to 0.70557, more significant than the whole rock of Hannuoba alkaline and tholeiitic basalts reported previously and consistent with that of Cenozoic basalts in North China. The compositional heterogeneity of Hannuoba tholeiitic basalt is caused by the mixing of heterogeneous lower crust rather than different mantle-derived melts, which indicates that the contribution of the continental lower crust to continental basalt is more complicated than previously recognized.
Trace element partitioning between anhydrite, sulfate melt, and silicate melt
https://doi.org/10.2138/am-2022-8345This article by Hutchinson et al. presents experimentally derived trace element partition coefficients between anhydrite, sulfate melt, and silicate melt at conditions typical for arc magmas. Partitioning behavior for 2+ and 3+ cations in anhydrite is controlled by exchange reaction with the anhydrite Ca2+ site and is modeled using a lattice strain approach. Modeled partitioning is then compared to measured strontium concentrations in several examples of natural magmatic anhydrite to assess the conditions of anhydrite crystallization.
Chemical reaction between ferropericlase (Mg,Fe)O and water under high pressure-temperature conditions of the deep lower mantle
https://doi.org/10.2138/am-2022-8390Ferropericlase (Mg,Fe)O is the second most abundant phase in a pyrolitic lower mantle model. Chemical reaction between ferropericlase and water was investigated by Yang et al. under high pressure-temperature conditions of the deep lower mantle. The pyrite-structured phase FeO2Hx (x≤1, Py-phase) containing a negligible amount of Mg (<1 at%) was formed at the expense of iron in the Fp-phase through the reaction between (Mg,Fe)O and H2O, thus serving as a mechanism of water storage in the deepest lower mantle.
Composition-dependent thermal equation of state of B2 Fe-Si alloys at high pressure
https://doi.org/10.2138/am-2022-8067Iron-silicon alloys are classically considered to be major components of planetary cores, in particular for planets having formed under reducing conditions. Such conditions are believed to be particularly relevant to the case of Mercury. However, both the phase diagram and thermoelastic properties of Si-rich alloys in the Fe-FeSi system are not well understood under high pressure for a wide range of compositions. This study by Yokoo et al. reports experimental results of pressure-temperature-volume relations in iron-silicon alloys with various silicon contents obtained over a large pressure (up to ~120 GPa) and temperature (up to ~3000 K) range. Based on the collected data, Yokoo et al. constructed an empirical thermal equation of state for the B2 phase. The silicon content of the B2 phase showed that the phase boundary is temperature independent and moves towards the Si-rich side with increasing pressure. The results have been applied to evaluate the buoyancy of crystallizing solids within Mercury's core. While density contrast between solid and liquid is small in alloys with ~10 wt% silicon or less, a Si-rich core can crystallize B2 alloys dense enough to gravitationally sink to form a solid inner core.
Effects of thermal annealing on water content and δ18O in zircon
https://doi.org/10.2138/am-2022-8444Primary water contents and δ18O in zircon have proved potentially important proxies for magmatic processes and magma source regions. However, the assignment of primary water is not straightforward because secondary water can enter zircon after crystallization, especially in metamict zircons due to U-Th radiation damage. Many established screening criteria for the least metamict zircons may not be applicable to ancient samples, in which nearly all zircons are metamict. This study by Yang et al. shows that annealing at 600°C for >4 hours can effectively recover primary water and oxygen isotopes from metamict zircons and significantly improves the oxygen isotope measurement accuracy by eliminating secondary water and matrix effects from the metamict zircons. The effective recovery of primary water and oxygen isotope information from metamict zircons through thermal annealing extends the application of these proxies to geologically ancient samples.
Tourmaline and zircon trace the nature and timing of magmatic-hydrothermal episodes in granite-related Sn mineralization: Insights from the Libata Sn ore field
https://doi.org/10.2138/am-2022-8357This contribution by Vincent et al. focuses on granite-hosted Sn and associated veins from Northwestern Nigeria, which forms part of an extensive Sn-W-Nb-Ta metallogenic belt across western Nigeria. In this paper, major, trace element, and boron isotopic compositions of tourmaline and in-situ geochronological and Lu-Hf isotopes studies of zircons hosted in granites and wall rocks from the Libata Sn ore field are used to constrain the origin, formation mechanism, and evolution of this ore field. This paper highlights the utility of both tourmaline and zircon as tracers for formation conditions, as well as nature and timing of magmatic-hydrothermal episodes in Sn bearing ore systems. They propose a Pan-African magmatic-hydrothermal event for the ore mineralization in the Libata ore field based on their zircon U-Pb data, which constrained overlapping magmatic and hydrothermal ages of ~650 Ma for the Libata granites. Magmatic and hydrothermal zircon grains from the unmineralized and altered rock suites have overlapping εHf(t) and 176Hf/177Hf but distinct 176Lu/177Hf and 176Yb/177Hf ratios, which suggest a magmatic source for the hydrothermal fluids that are responsible for the mineralization. The tourmalines from the two studied veins in the Libata ore field are of schorls and have high alkali, high Al content, and moderate X-site vacancies as well as Li/Sr and Ca-Fe-Mg ratios that constrain a magmatic source for the Libata tourmalines. T These findings have widespread implications for the genesis for granite-related Sn deposits in other parts of the world with similar geological setting.
Cation ordering, twinning, and pseudo-symmetry in silicate garnet: The study of a birefringent garnet with orthorhombic structure
https://doi.org/10.2138/am-2022-8455Silicate garnets are common rock-forming minerals that occur in Earth's crust and mantle. In general, silicate garnets have cubic (Ia3(bar)d) symmetry. Many studies on rare birefringent garnet structures are performed with powder XRD. However, the lattice parameters of birefringent garnets are very close to a cubic structure, which would result in significant overlap between symmetrically non-equivalent reflections in the powder XRD pattern. Information regarding the extinction conditions and slight deviation from the cubic lattice would be completely lost, and many unsupported assumptions have to be made in those studies, resulting in questionable conclusions. It is only with single crystal-XRD and high-resolution TEM can we extract the details in the cation ordering and twin domain structures. The results from Huifang Xu et al. imply that Fe-Al ordering states in the grandite garnets are related to their crystallization temperatures. Well-ordered grandite garnet with compositions close to Adr0.5Grs0.5 was crystallized at low-temperature. Possible lower symmetries in reported grandite garnets can be explained with the determined structure and twins in different proportions. Coexisting grandite garnets and Al-Fe ordering states in the orthorhombic structure can be used as an indicator of their crystallization temperature.
The occurrence of monoclinic jarosite in natural environments
https://doi.org/10.2138/am-2022-8276Jarosite and related subgroup minerals are of high importance in mineral processing, as sources and sinks for metals and acidity in the environment, and they have the potential to preserve elemental and isotopic biomarkers relevant to the search for life in the solar system. The crystal structures and chemistry of jarosite minerals influences their stability and reactivity and thus the roles they play in natural and engineered systems. Whitworth et al. report the occurrence of monoclinic symmetry in a natural natrojarosite sample investigated using synchrotron powder X-ray diffraction (PXRD), thermogravimetric analysis (TGA), and electron backscatter diffraction (EBSD). The results of this study suggest that monoclinic symmetry in natural jarosites may be more prevalent than previous studies suggest. The splitting of peaks in monoclinic jarosites can be subtle so it is recommended that high-resolution XRD data is collected when studying the crystal structure of jarosites.
Niobium speciation in minerals revealed by L2,3-edges XANES spectroscopy
https://doi.org/10.2138/am-2022-8293This paper from Bollaert et al. provides the first Nb L2,3-edges XANES spectroscopy study on a selected set of Nb minerals and synthetic oxides. Spectral features mirror the sensitivity to the average Nb–O distance, local site symmetry, and bond covalence, which are used to constrain Nb speciation in minerals. Deciphering Nb speciation will improve our understanding of Nb enrichment in primary and lateritic deposits and may help to diversify the range of primary sources and to upgrade the efficiency of ore processing of this critical metal.
The first occurrence of the carbide anion, C4-, in an oxide mineral: Mikecoxite, ideally (CHg4)OCl2, from the McDermitt open-pit mine, Humboldt County, Nevada, U.S.A.
https://doi.org/10.2138/am-2022-8408Cooper et al. describe mikecoxite, ideally (CHg4)OCl2, the first oxide-chloride mineral to be discovered that also contains the carbide ion: C4-, a rather unexpected discovery that extends the range of anions in oxide-halogen-oxysalt minerals to (F, Cl, Br, I)-, (O, S)2-, N3-, and C4-. The source of carbon for mikecoxite in the volcanic, high-desert environment of the type locality seems to be methane, with the reaction catalyzed by microbiota through full mercuration of carbon atoms, beyond the first stage that produces the volatile and highly mobile methylmercury, [CH3Hg]+.
Hydrothermal alteration of Ni-rich sulfides in peridotites of Abu Dahr, Eastern Desert, Egypt: Relationships among minerals in the Fe-Ni-Co-O-S system, fO2 and fS2
https://doi.org/10.2138/am-2022-8217Abdel-Halim demonstrate that the sulfide mineralogy reflects the magmatic and post-magmatic evolution of Abu Dahr complex and that serpentinization-related Ni released from magmatic olivine upgraded the tenor of Ni in disseminated magmatic ore and the Ni-abundance in rocks. They also found that the hydrothermal alteration processes change the mineralogy of the Ni-bearing minerals and compare the high-Ni sulfide and metallic phases of Abu Dahr serpentinites to global Ni-rich serpentinites.
New Mineral Names
https://doi.org/10.2138/am-2023-NMN10831February 2023
Analyses under the curve, identifying how invisible gold is held in pyrite
https://doi.org/10.2138/am-2022-8791This Highlight and Breakthrough paper by Daniel Gregory shows how the paper by Ehrig et al. (see below) traces how amounts of gold are held in the mineral pyrite. It has long been debated whether gold occurs dissolved in the pyrite or occurs as inclusions of other mineral species. This paper shows that even when traditional techniques would indicate gold is held within the crystal structure, it is actually held within nanoinclusions of other minerals.
Titanite geochemistry and textures: Implications for magmatic and post-magmatic processes in the Notch Peak and Little Cottonwood granitic intrusions, Utah
https://doi.org/10.2138/am-2022-8241Henze et al. demonstrate that titanite compositions and textures provide important insights into the origins of granitic rocks and can be used to distinguish separate batches of magma, gauge the evolution of magmatic rocks, assess mixing processes, and infer compositions of mixing components. Because titanite also forms hydrothermally, it retains hints about the composition, temperature, and oxygen fugacity of the hydrothermal fluids and reveals details about titanite-forming reactions.
Gismondine-Sr, Sr4(Al8Si8O32)·9H2O, a new strontium dominant, orthorhombic zeolite of the gismondine series from the Hatrurim Complex, Israel
https://doi.org/10.2138/am-2022-8376Skrzynska et al. show that, in addition to high pressure and dehydration, the elliptical deformation of the channels in GIS also arises as a consequence of the extraframework cations and H2O content. The extraframework content, i.e., the type of extraframework cations in t-gsm cages of gismondine-Sr, influences the aluminosilicate framework leading to the orthorhombic symmetry.
Lifting the cloak of invisibility: Gold in pyrite from the Olympic Dam Cu-U-Au-Ag deposit, South Australia
https://doi.org/10.2138/am-2022-8395Ehrig et al. employed a combination of micrometer- and nanoscale analysis to define the location and physical form of so-called “invisible gold” in pyrite from one of the Earth’s largest ore systems, Olympic Dam, South Australia. They recognize nanostructures associated with local enrichment in Te, Bi, and Pb and the presence of Au-Ag-telluride nanoparticles. The data reveal new layers of complexity with respect to the trace/minor element incorporation in mineral matrices and their subsequent release during overprinting.
Paragenesis and precipitation stages of Nb-Ta-oxide minerals in phosphorus-rich rare-element pegmatites (Buranga dike, Rwanda)
https://doi.org/10.2138/am-2022-8201Araujo et al. present the application of high-resolution Raman imaging techniques, coupled with petrography and EPMA analyses, to define the paragenesis and crystallization stages of Nb-Ta-oxide minerals (NTO) in the phosphorus-rich Buranga pegmatite. The results show that NTO precipitates as (1) the Fe-Nb-Ta-rich rutile in the magmatic stage; and (2) columbite group minerals (CGM) in the magmatic-hydrothermal stage. Additionally, the CGM precipitation is linked with the destabilization of primary minerals after crystal-fluid-melt interactions and depends on the availability of Fe and Mn in the system, which in turn is controlled by the crystallization of phosphate minerals in these flux-rich pegmatites.
3D zoning of barium in alkali feldspar
https://doi.org/10.2138/am-2022-8139Lubbers et al. used computed X-ray microtomography (microCT) to observe 3D chemical zoning in natural sanidine minerals. The results allow us to better quantify mineral zoning geometries and volumes while mitigating sectioning effects related to looking at minerals in 2D. These results have implications for petrological tools that interpret mineral zoning (e.g., diffusion chronometry, mineral growth/dissolution).
In situ Raman vibrational spectra of siderite (FeCO3) and rhodochrosite (MnCO3) up to 47 GPa and 1100 K
https://doi.org/10.2138/am-2022-8343Wang et al. report the Raman vibrational spectra of siderite and rhodochrosite at simultaneously high-P and high-T conditions for the first time. They found that the frequency shifts of the carbonate modes are a coupled function of both P and T, and the effect of the P-T cross-derivative term cannot be neglected. A method of using high P-T Raman scattering data to calculate the mode (macroscopic) Gruneisen parameters (γi) and the bulk modulus parameter (K(T)) is established.
Isotopic responses of Mg to two types of dissolution-reprecipitation processes for the growth of the double-carbonate mineral norsethite
https://doi.org/10.2138/am-2022-8386Liu et al. used Mg isotopes to probe the trace element behavior during the formation and growth of norsethite ['BaMg(CO3)2]. They observed two different types of dissolution-reprecipitation process during carbonate growth, including the transformation of amorphous carbonate to crystalline carbonate (fast dissolution-reprecipitation) and recrystallization of crystalline carbonate (slow dissolution-reprecipitation). The study finds significantly different Mg isotope fractionation behaviors during these two processes and highlights the importance of understanding the formation mechanism and post-depositional history of carbonate for interpreting the isotopic data in carbonate minerals.
Fluid-rock interaction and fluid mixing in the large Furong tin deposit, South China: New insights from tourmaline and apatite chemistry and in situ B-Nd-Sr isotope composition
https://doi.org/10.2138/am-2022-8310Chen et al. show that the in situ elemental and B-Sr-Nd isotopic compositions of tourmaline and apatite at the Furong Sn deposit define the greisen-type mineralization to a dominantly magmatic-hydrothermal environment and the skarn-type mineralization to an open system with fluid mixing and fluid-rock reactions. This work highlights that coupled hydrothermal minerals such as tourmaline and apatite have great potential to fingerprint the nature, source, and evolution of fluids in granite-related ore systems.
A neutron diffraction study of boussingaultite, (NH4)2[Mg(H2O)6]SO4)2
https://doi.org/10.2138/am-2022-8385Boussingaultite is a magnesium ammonium sulfate hexahydrate with the ideal formula (NH4)2&91;Mg(H2O)6](SO4)2. It occurs as a sublimate formed under fumarolic conditions, geysers, or from coal gas at burning coal dumps. There is an increasing interest in boussingaultite, or its synthetic counterpart, generated by some potential industrial and agricultural utilization of this material. Using a multi-methodological approach, this study by Gatta et al. sheds new light on the mechanisms that promote solid solution with substitutions in the crystalline edifice A+2B2+(SO4)2(H2O)6 at the A (e.g., K+ vs. NH4+) and at the B (e.g., Mn2+ vs. Mg2+) sites.
Zn-clays in the Kihabe and Nxuu prospects (Aha Hills, Botswana): A XRD and TEM study
https://doi.org/10.2138/am-2022-8439Putzolu et al. conducted powder XRD and TEM-AEM experiments of Zn-rich clays in the Kihabe and Nxuu prospects. The obtained data allowed describing fraipontite and sauconite as the main Zn carriers. The study of the paragenetic evolution of the ore showed that Zn-clays formed at the expense of early precipitated barren phyllosilicates (i.e., kaolinite and beidellite) of hydrothermal origin that were fertilized by Zn2+ and Al3+-rich fluids of mixed meteoric and low-T hydrothermal nature. The nanoscale TEM analyses revealed the alteration of previously formed willemite and sphalerite sourced Zn during the clay formation process. Furthermore, the detection of low tetrahedral charge sauconite, with a stoichiometry close to synthetic Zn-stevensite, is valuable to future studies on metals enrichment in 2:1 clays and also has implications on the clay systematics.
Finchite, Sr(UO2)2(V2O8)·5H2O, a new uranyl sorovanadate with the francevillite anion topology
https://doi.org/10.2138/am-2022-8365Spano et al. describe a new mineral, finchite, Sr(UO2)2(V2O8)·5H2O. This is the first uranium mineral known to contain essential Sr and represents a possible immobilization species for both uranium and the hazardous radionuclide 90Sr because of the relative insolubility of uranyl vanadate minerals in water.
Multi-stage metasomatic Zr mineralization in the world-class Baerzhe rare-earth-element Nb-Zr-Be deposit, China
https://doi.org/10.2138/am-2022-8336Wu et al. show that metasomatic zircon in the agpaitic granite was not subjected to metamictization or intense alteration, with trace elements accommodation largely following a xenotime substitution mechanism. Most zircon in the miaskitic-agpaitic complex at Baerzhe was the product of autometasomatism that was constrained to their parental granites. Some REE- and Be-rich zircon that replaced magmatic amphibole crystallized as a result of a reaction with an REE- and Be-rich fluid that most likely was responsible for REE-Nb-Be mineralization that affected both the miaskitic and agpaitic granites. The multi-stage metasomatic zircon-forming events present at Baerzhe reveal a more complex history of zircon formation in alkaline complexes than previously thought. Such complexity in the formation of metasomatic zircon could apply to similar alkaline settings that contain high Zr endowments.
January 2023
Nickel in olivine as an exploration indicator for magmatic Ni-Cu sulfide deposits: A data review and re-evaluation
https://doi.org/10.2138/am-2022-8327Nickel and cobalt are essential battery metals, increasingly in demand as the world moves away from fossil fuels. We need to find new deposits, but exploration is becoming increasingly difficult. One of the techniques that have been applied for decades to identify possible target bodies of igneous rock (intrusions) is to look at the nickel contents of the common silicate minerals, particularly olivine, on the assumption that ore-forming processes will leave a signal of nickel depletion that extends beyond the ore deposits themselves. A large body of data has been collected worldwide over several decades, including thousands of analyses of olivines in volcanic rock as well as in intrusions, but the multiple factors that control the variability of nickel in olivine have not been systematically considered and evaluated to test whether the signals of magmatic sulfide formation can be reliably identified. Barnes et al. begin by defining the “baseline” of olivine compositions in non-ore-bearing rocks in different settings, then show that the natural variation in ore-bearing rocks shows complex signals, including enrichment as well as depletion in nickel relative to these baselines. Through empirical investigation and quantitative modeling, they attempt to unravel the multiple factors that lead to these observations. Nickel in olivine can indeed be a useful ore deposit indicator in some circumstances, but many confounding factors need to be considered. Nevertheless, useful information about ore-forming processes can certainly be derived.
Repeat, fast, and high-resolution mapping of fine-scale trace element distribution in pyrite and marcasite by LA-Q-ICP-MS with the Aerosol Rapid Introduction System (ARIS)
https://doi.org/10.2138/am-2022-8168Minor and trace element compositions of sulfides provide critical insights into a variety of geological processes. Reynolds et al. detail a new method for mapping minor and trace element distribution in pyrite and marcasite by LA-Q-ICP-MS using the Aerosol Rapid Introduction System (ARIS) and novel operating conditions. This approach allows for increased mapping speed, decreased limits of quantification, and decreased cost of consumables. As a result, extraordinarily high-resolution maps can be created without an excessive compromise of speed, cost, or limits of quantification.
Continuous Be mineralization from two-mica granite to pegmatite: Critical element enrichment processes in a Himalayan leucogranite pluton
https://doi.org/10.2138/am-2022-8353Beryllium is a critical metal typically concentrated in highly fractionated granitic rocks. Liu et al. study beryl mineralization in a typical leucogranite pluton at Pusila in the central Himalaya, a striking example of continuous Be mineralization. (1) Beryl is the most common beryllium mineral and is ubiquitous at Pusila. The beryl compositions trend to Na-Li-Cs enrichment and Fe-Mg depletion and show continuous decreasing Na/Li and Mg/Fe, and increasing Cs/Na ratios, which are positively correlated with increasing whole-rock Rb/Sr ratios. Mineral chemistry characteristics highlight beryl as a monitor of magmatic evolution processes. (2) The enrichments of Be and other critical elements are associated with strongly fractionated granites or their equivalents. Despite the low to moderate fractionation, the biotite- and two-mica granites contain unusually high Be contents, indicative of an initial magma relatively enriched in Be. The gneisses of GHS, considered as the protolith, also show high Be abundances, which could be the source reservoir of Be. Therefore, a high degree of fractionation is likely not the only factor for beryl saturation. A source relatively enriched in Be could effectively reduce the dependence on the degree of fractionation to concentrate Be sufficiently to reach saturation. (3) This study reveals the differing behaviors of incompatible elements (e.g., Be, Cs) in melt. Once the system is beryl saturated, beryllium is no longer incompatible, and the Be concentrations would be controlled by multiple processes such as fractional crystallization and the buffering action of beryl crystallization. This combination of processes could be applicable to other trace and incompatible elements. (4) The term “delayed crystallization,” describing the process of beryl crystallization at the late stage of granite solidification in two-mica granite and muscovite granites, is the crucial part of the continuous crystallization model. This continuous crystallization model reveals the evolutionary mechanisms of continuous Be mineralization with a progressive increase in whole rock Be contents and concurrently could provide new insights into the enrichment processes of other critical metal elements, like Cs, Li, Zr, etc., which are commonly rare and incompatible in granitic melts.
An evolutionary system of mineralogy, Part VI: Earth’s earliest Hadean crust (>4370 Ma)
https://doi.org/10.2138/am-2022-8329Part VI of the evolutionary system of mineralogy, by Morrison et al., imagines Earth’s earliest mineralogy from a time before the oldest known mineral grain, more than 4.37 billion years old. Based on the evidence of minerals from rocky asteroids, planets, and moons, amplified by experimental petrology, geochemistry, and models of planetary evolution, they postulate 262 mineral species that formed within a few tens of millions of years of the Moon’s violent formation.
Oxidation or cation re-arrangement? Distinct behavior of riebeckite at high temperature
https://doi.org/10.2138/am-2022-8073Fe-rich amphiboles are important rock-forming silicates, and their composition, in particular their Fe3+/Fetot ratio, may provide insight into the redox state of the geological system where they grew. Della Ventura et al. studied the oxidation process at high temperature in riebeckite by combining Mossbauer spectroscopy and powder XRD and showed that riebeckite follows two distinct paths depending on the external environment. Under oxidizing conditions, riebeckite is stable in the hydrous form up to relatively low temperatures (400-450 °C), and then it undergoes a rapid transformation into an oxo-amphibole which is stable up to ~900 °C. Under vacuum conditions, no Fe oxidation is observed; riebeckite undergoes a significant cationic re-arrangement and is stable in the hydrous form up to much higher temperatures (750-800 °C). These findings imply that characterization of the oxidation state of iron in riebeckite does not necessarily provide the redox and thermal conditions of formation.
Fe3+/FeT ratios of amphiboles determined by high-spatial resolution single-crystal synchrotron Mossbauer spectroscopy
https://doi.org/10.2138/am-2022-8115This work by Ratschbacher et al. presents the first documentation of amphibole Fe3+/FeT ratios determined by single-crystal synchrotron Mossbauer spectroscopy (SMS). SMS provides a high-spatial resolution and precision to avoid analyzing inclusions and alteration features and thus allows detecting intra-grain compositional variations in Fe3+/FeT ratios, which then can be interpreted in the context of changing crystallization condition and/or post-crystallization oxidation. SMS does not require reference spectra, therefore eliminating that source of uncertainty in the final results.
How clay delamination supports aseismic slip
https://doi.org/10.2138/am-2022-8195Delamination between clay layers due to interlayer water lubrication was suspected of supporting aseismic slip but lacked a mechanistic insight. The variable hydration state of a clay mineral adds to the complexity. Here Zhou et al. use atomic-scale simulations to investigate the role of clay minerals in aseismic slip. The reported shear behaviors of clay minerals at different temperatures and pressures provide constraints on fault mechanics. Increased pore fluid pressure leads to more intercalated water, resulting in lower frictional strength and enhanced velocity-strengthening behavior. This work suggests that knowledge of the hydration state of a clay mineral is a necessity when studying fault mechanics.
The influence of Al2O3 on the structural properties of MgSiO3 akimotoite
https://doi.org/10.2138/am-2022-8257Akimotoite, a MgSiO3 polymorph present in Earth’s lower transition zone, will contain some Al, raising the question of how this will affect its crystal structure and properties. Siersch et al. investigate MgSiO3 akimotoite - Al2O3 corundum solid solutions by single-crystal X-ray diffraction in order to examine their crystal chemistry. Results show a strong non-linear behavior of the a- and c-axes as a function of Al content, which arises from fundamentally different accommodation mechanisms in the akimotoite and corundum structures and suggests that an immiscibility gap may be present around the 50:50 compositions in this solid solution. Furthermore, samples belonging to the akimotoite - corundum solid solutions were investigated at high pressure in order to determine the different compression mechanisms associated with the cation substitution. Al2O3-bearing akimotoite becomes more compressible at least up to 20 mol% Al2O3, which is likely driven by an increase in compressibility as the Al cation is incorporated into the SiO6 octahedron. This observation is in strong contrast to the stiffer corundum end-member with respect to that of the akimotoite end-member. These findings have important implications for mineral physics models of elastic properties, which have in the past assumed linear mixing behavior between the MgSiO3 akimotoite and Al2O3 corundum end-members in order to calculate sound wave velocities for Al-bearing akimotoite at high pressure and temperature.
Atomistic insight into the ferroelastic post-stishovite transition by high-pressure single-crystal X-ray diffraction refinements
https://doi.org/10.2138/am-2022-8458Zhang et al. studied atomistic information and distortion modes across the ferroelastic transition from stishovite to post-stishovite. Using the synchrotron single-crystal X-ray diffraction technique, they determined crystal structures of the stishovite and post-stishovite phases in a diamond-anvil cell up to 75.3 GPa. Across the transition at ~51.4 GPa, the oxygen coordinate splits, the apical and equatorial Si-O bond lengths cross over, and a rotational lattice distortion occurs. This structural information has been correlated with shear modulus softening and Landau parameters, including spontaneous strains and order parameters reported in the literature. Importantly, they found that the symmetry-breaking spontaneous strain and the order parameter are proportional to the SiO6 rotation angle due to the appearance of the rotational mode. Together with atomistic insights of other types of ferroelastic transitions (such as the proper-type in albite and the improper-type in CaSiO3 perovskite), Zhang et al. found that the ferroelastic transition always occurs through changes of structural angle and an occurrence of a distortion mode. This finding could further shed light on the abnormal seismic properties and geodynamics in the regions where the ferroelastic transition occurs.
Epidote as a conveyor of water into the Earth’s deep mantle in subduction zones: Insights from coupled high pressure and high-temperature experiments
https://doi.org/10.2138/am-2022-8252Li et al. found that epidote begins to decompose at 1223 K and fully breaks down at 1373 K at atmospheric pressure. Furthermore, there is no phase transition in epidote at temperatures and pressures up to 1172 K and 14.0 GPa. These results suggest water can be conveyed downward into the mantle transition zone through epidote in cold, subducting mafic oceanic crust.
Potential link between antigorite dehydration and shallow intermediate-depth earthquakes in hot subduction zones
https://doi.org/10.2138/am-2022-8271The dehydration of antigorite has been associated with intermediate-depth earthquakes in subduction zones. However, the reaction of antigorite → forsterite + talc has not yet been considered to explain some of these earthquakes because this reaction was not thought to occur in subducting mantle. In the hot subducting mantle beneath Cascadia and southwestern Japan, however, the reaction fits shallow intermediate-depth earthquakes. To explore whether the reaction can trigger these earthquakes, Shao et al. conducted a series of isothermal experiments on the kinetics of antigorite dehydration. Avrami modeling of the experimental data combined with microstructural observations of run products suggests that the reaction is controlled by a heterogeneous nucleation and growth mechanism. The fast fluid production rate of the reaction may lead to high fluid pressure in the subducting mantle but does not necessarily cause embrittlement. Superplasticity or velocity weakening of fine-grained forsterite and velocity weakening of antigorite by water and/or talc may be responsible for earthquake nucleation and propagation in a heterogeneous system, which can be either antigorite and its dehydration products within a serpentinized fault zone or the mixture of antigorite and surrounding peridotite in hot subduction zones (<2 GPa).
Stability of Fe5O6 and its relation to other Fe-Mg-oxides at high pressures and temperatures
https://doi.org/10.2138/am-2022-8370Woodland et al. report new results on the stability of high-pressure Fe and Mg-Fe oxide phases. Such information is important for understanding the mineralogy of the Earth’s mantle, particularly in certain domains that appear to be iron-rich. The existence of such domains is documented by certain mineral inclusions found in diamonds and, by inference, may also be involved in the process of diamond formation. From these results, they conclude that either Fe4O5 or Fe5O6 could occur in such Fe-rich environments. However, the observed limited solubility of Mg in Fe5O6 implies that it will not be present as an accessory mineral in a peridotite bulk composition. Rather a Fe4O5 phase would be more likely to be stable in the “average” mantle composition.
From schwertmannite to natrojarosite: Long-term stability and kinetic approach
https://doi.org/10.2138/am-2022-8288Jimenez et al. find that schwertmannite cannot be considered an efficient phase for the immobilization of contaminating metals in the short term, whereas natrojarosite remains stable under conditions of high acidity for long reaction times and can be considered more reliable for the retention of heavy metals. Moreover, an exhaustive spectroscopic study confirms the mineralogical results and suggests that the crystal structure of jarosite-like minerals may offer interesting geochemical information about the aqueous solutions in which they were formed.
Trace element and isotopic (S, Pb) constraints on the formation of the giant Chalukou porphyry Mo deposit, NE China
https://doi.org/10.2138/am-2022-8142Porphyry-type Mo deposits have supplied most of Mo to the world. However, the source of the Mo and the controls on its enrichment in such deposits are still a matter of great debate. Zhao et al. present in situ trace element and isotopic data for a giant porphyry Mo deposit and use these data to address this issue. The trace element chemistry of pyrite and S and Pb isotopic compositions have made it possible to interpret the evolution of hydrothermal fluids during the formation of the Chalukou porphyry Mo deposit. The in situ LA-ICP-MS results lead to the conclusion that the absolute content of metal in the ore fluid was not the key factor controlling metallic mineral deposition and that molybdenite saturation was controlled by the system temperature. Another important conclusion of the study is that magmas unusually enriched in Mo due to crustal contamination, and crystallized as the syn-ore intrusions, were the source of Mo for the Chalukou deposit. This shows, contrary to previous studies, that magma fertility (enrichment in Mo) may play an essential role in generating large porphyry Mo deposits. Finally, the study highlights the importance of in situ compositional analyses (including S and Pb isotopes and trace elements) of ore sulfides as tools for constraining the origin and chemical evolution of ore fluids.
Textural and chemical evolution of magnetite from the Paleozoic Shuanglong Fe-Cu deposit: Implications for tracing ore-forming fluids
https://doi.org/10.2138/am-2022-8400Zhang et al. identified three types of magnetite (MA, MB, and MC) in the Shuanglong Fe-Cu deposit, modified by the mushketovitization, as well as the dissolution and reprecipitation. Scheelite and wolframite inclusions in MA-type magnetite formed by the W expulsion during mushketovitization. Textural and chemical information reveals that the synchronous oscillatory zoning in magnetite and coexisting chlorite was caused by the variation of fluid temperature.
Jingwenite-(Y) from the Yushui Cu deposit, South China: The first occurrence of a V-HREE-bearing silicate mineral
https://doi.org/10.2138/am-2022-8373Liu et al. report the occurrence of jingwenite-(Y), the first V-HREE-bearing silicate mineral discovered in nature, and its structure represents a new structural type of nesosilicate and can be classified as a new mineral group.
Wenjiite and kangjinlaite: New minerals in the ternary Ti-P-Si system from the Luobusa ophiolite, Tibet, China
https://doi.org/10.2138/am-2022-8226Xiong et al. demonstrate that wenjiite and kangjinlaite are more than just another pair of new minerals. Wenjiite has implications for understanding polymorphism in intermetallic compounds. It is isostructural with over 175 compounds, including two other minerals, but this structure type is only one of four principal types reported for the 5:3 stoichiometry. Kangjinlaite has not been previously reported either in nature or as a product of synthesis. Discovering and characterizing kangjinlaite and wenjiite was only possible thanks to the recently developed 3-dimensional electron diffraction technique. The new technique enables the collection of in situ structural data suitable for ab-initio solution and refinement of crystal structures from single grains of less than 1 micrometer in size.
Evaluating the physicochemical conditions for gold occurrences in pyrite
https://doi.org/10.2138/am-2022-8207Gold extraction from other minerals is often of great interest to the mining industry. The incorporation mechanism of gold in the host mineral is a chief factor affecting the extraction of gold. For a long time, various incorporation mechanisms have been recognized through spectroscopy, but with little theoretical consideration. In this study, He et al. consider such incorporation mechanisms from an energetic perspective; they propose a theoretical framework investigating the occurrence of elements in mineral structures and employing DFT calculations to examine the occurrence of gold in pyrite.
American Mineralogist Volume 107
December 2022
Highlights and Breakthroughs: Oxidation of arcs and mantle wedges: It’s not all about iron and water
https://doi.org/10.2138/am-2022-8802Callum Hetherington wrote a commentary on Song et al. (2022) Oxidation of arcs and mantle wedges by reduction of manganese in pelagic sediments during seafloor subduction. American Mineralogist, 107, 1850-1857.
Paragenesis of Li minerals in the Nanyangshan rare-metal pegmatite, Northern China: Toward a generalized sequence of Li crystallization in Li-Cs-Ta-type granitic pegmatites
https://doi.org/10.2138/am-2022-8285Yang et al. describe a generalized crystallization sequence of lithium minerals in LCT-type pegmatites. As the typical and the largest one of hundreds of LCT pegmatite dikes in eastern Qinling orogenic district, North China, the Nanyangshan pegmatite is strongly Li-mineralized with a clear series of lithium minerals, including spodumene, montebrasite, lithiophilite, elbaite, lepidolite, and possible former petalite. They discuss the potential factors (mostly volatile activity) controlling the stability of lithium minerals under conditions of pegmatite crystallization. They collect Li-mineral data of 58 Li-Cs-Ta pegmatites worldwide, which are used to infer a general consensus of successive evolution of Li minerals with crystallization of LCT pegmatites.
The new mineral tomiolloite, Al12(Te4+O3)5[(SO3)0.5(SO4)0.5](OH)24: A unique microporous tellurite structure
https://doi.org/10.2138/am-2022-8368Missen et al. describe a new mineral, tomiolloite, which has a unique structure and chemical composition. It was found in the oxidation zone of a tellurium-gold mine near Moctezuma, Sonora, Mexico. Tomiolloite has a microporous structure containing channels. The chemical composition of tomiolloite includes aluminium cations and four anions: the tellurite anion, the sulfate and sulfite anions (unusual to find in the same structure), and hydroxide anions.
Authigenic anatase nanoparticles as a proxy for sedimentary environment and porewater pH
https://doi.org/10.2138/am-2022-8330Titanium has long been considered to be immobile during weathering and diagenetic processes, and it is widely used for normalization of elemental concentrations in weathering profiles. However, Hong et al. demonstrate that authigenic titania is commonly formed in fine-grained siliciclastics of a wide variety of facies through weathering of Ti-bearing silicates, authigenic euhedral anatase is present ubiquitously as nanoparticles, and its morphology varies in a predictable manner over a range of depositional environments. The crystal habit of authigenic anatase nanoparticles is controlled primarily by porewater pH during early diagenetic alteration of Ti-bearing silicates. Their findings suggest that authigenic anatase could be a sensitive proxy for the sedimentary environment and sedimentary porewater chemistry and will likely prove useful in depositional facies analysis.
Color effects of Cu nanoparticles in Cu-bearing plagioclase feldspars
https://doi.org/10.2138/am-2022-8325This study by Jin et al. bridges the gap between colloidal optics in material science and the color effects in minerals and gemstones. The different colors and pleochroism in Cu-bearing feldspars are explained quantitatively for the first time. The result also has implications for understanding the crystal chemistry of feldspars, as well as the igneous processes that create them, by providing a way to quantify the diffusion of Cu in feldspars through optical spectroscopy. It also inspires designing and engineering novel optical materials.
Expanding the speciation of terrestrial molybdenum: Discovery of polekhovskyite, MoNiP2, and insights into the sources of Mo-phosphides in the Dead Sea Transform area
https://doi.org/10.2138/am-2022-8261Britvin et al. describe a new mineral, polekhovskyite, MoNiP2, the first terrestrial Mo phosphide and a phosphorus-rich homologue of meteoritic monipite, MoNiP. Polekhovskyite represents a novel terrestrial Mo speciation. Having 44 wt% Mo in its composition, the mineral is a striking example of selective Mo enrichment. The origin of Mo in phosphides of the Southern Levant is likely related to the processes of the Dead Sea Rift formation.
Sound speed and refractive index of amorphous CaSiO3 upon pressure cycling to 40 GPa
https://doi.org/10.2138/am-2022-8081Geballe et al. investigate amorphous silicate, CaSiO3, that is created through several different pressure-temperature routes. The samples exhibit highly reproducible properties at room temperature and at pressures up to 40 GPa, especially in comparison to other silicates. This suggests that the amorphous solid may mimic the liquid over the pressure range investigated. The basis of this assessment is three complementary data sets: sound-speed measurements, refractive index measurements, and previously published NMR and Raman spectroscopy.
Calorimetric study of skutterudite (CoAs2.92) and heazlewoodite (Ni3S2)
https://doi.org/10.2138/am-2022-8337Thermodynamic properties of chalcogenide minerals are important for modeling and understanding ore deposits. In particular, nickel and cobalt arsenides, sulfarsenides, and sulfides occur in many hydrothermal ore deposits but their thermodynamic properties of these phases are not well known. Majzlan et al. determined a full set of thermodynamic properties for heazlewoodite and skutterudite using high-temperature oxide-melt solution calorimetry and low-temperature relaxation calorimetry.
Melting phase equilibrium relations in the MgSiO3-SiO2 system under high pressures
https://doi.org/10.2138/am-2022-8004Moriguti et al. determined the melting relations in the system MgSiO3-SiO2 at 13.5 GPa and up to 2900 °C using the Kawai-type of multi-anvil apparatus. At such extreme temperatures, generated pressure calibration was also carried out. The mass-balance calculations on E-chondrites model indicate that Si content in the core would be between 2.7 to 8.6 wt%, which is within the range of 2 to 9 wt% Si in the core as predicted by metal-silicate element partitioning. Thus, the E-chondrite model could still have high potential to explain the Bulk Earth composition if the Si depletion in the core has worked well through Earth's history.
Effects of hydrostaticity and Mn-substitution on dolomite stability at high pressure
https://doi.org/10.2138/am-2022-8248Wang et al. collected high-pressure Raman spectra of natural Mg-dolomite CaMg(CO3)2 and Mn-dolomite kutnohorite Ca1.11Mn0.89(CO3)2 samples up to 56 GPa at room temperature in a diamond anvil cell using helium or neon as a pressure-transmitting medium (PTM). Phase transitions in CaMg(CO3)2 were observed at 36.1(25) GPa in helium and 35.2(10) GPa in neon PTM for dolomite-II to -III, respectively. Moreover, the onset pressure of Mn-dolomite Ca1.11Mn0.89(CO3)2-III occurs at 23−25 GPa, about 10 GPa lower than that of Mg-dolomite-III. These results reveal a significant effect of hydrostaticity and cationic radius on the kinetics of the pressure-induced structure transformations in the dolomite group. The results provide new insights into deep carbon carriers within the Earth's mantle.
Crystallization of bastnaesite and burbankite from carbonatite melt in the system La(CO3)F-CaCO3-Na2CO3 at 100 MPa
https://doi.org/10.2138/am-2022-8064Nikolenko et al. present the first experimental data on the crystallization of bastnaesite and two other REE carbonates, burbankite and lukechangite, from carbonatitic melt in the model system La(CO3)F-CaCO3-Na2CO3 at 100 MPa and temperatures between 625 and 850 ºC. Liquidus phases in the run products are calcite, nyerereite, Na carbonate, bastnaesite and burbankite solid solution and lukechangite. Addition of 10 wt% Ca3(PO4)2 to a ternary mixture resulted in massive crystallization of La-bearing apatite and monazite, and complete disappearance of bastnäsite and burbankite. Thus, primary magmatic crystallization of REE carbonates and fluorocarbonates from natural carbonatitic melts is unlikely (with a possible exception of the uniquely REE-rich and P2O5-poor carbonatites at Mountain Pass).
Crystal shapes, triglyphs, and twins in minerals: The case of pyrite
https://doi.org/10.2138/am-2022-8280Arrouvel analyzed pyrite samples using XRD, SEM and EDS coupled with atomistic simulations to study their crystal growth, twinning and anisotropy. The findings include: (1) {120} and {210} pyritohedral pyrite can be distinguished by the orientation of the striations on their surfaces. (2) The striations are linked to kinetic growth with a higher rate. (3) The directions of the striations are compatible to the sulfur network orientated along the 6 <001> directions. (4) The so-called negative striated {120} pyrite crystals are rare specimens that can be encountered in geothermal areas. (5) Striations help identify [uvw]° rotations in merohedral twinning. (6) Simulations confirm that mirror (001) twin and [001]90° iron-cross twinning are energetically the most favorable grain boundaries.
Nanostructure reveals REE mineral crystallization mechanisms in granites from a heavy-REE deposit, South China
https://doi.org/10.2138/am-2022-8309Shi et al. report unusual nanostructure of Ce-poor bastnasite-(La,Nd,Y) from the South China granites, whose weathering crusts form HREE deposits. In-situ SRXD and high-resolution TEM analyses show the REE-mineral grew as disordered nanocrystals, and coaligned, nearly coaligned nanoparticle aggregations. They provide a novel nonclassical crystallization by particle attachment, and further support that the HREE-rich mineral precipitation in the parental granites underwent sudden, rapid crystallization in a highly oxidized environment. This work has practical implications for mineral exploration, as it provides criteria for the identification of similar HREE-rich deposits elsewhere.
Paratobermorite, Ca4(Al0.5Si0.5)2Si4O16(OH)˙2H2O˙(Ca˙3H2O), a new tobermorite-supergroup mineral with a novel topological type of the microporous crystal structure
https://doi.org/10.2138/am-2022-8284Pekov et al. discovered a new mineral, paratobermorite, ideally Ca4(Al0.5Si0.5)2Si4O16(OH)˙2H2O˙Ca˙3H2O), a member of the tobermorite supergroup, at the Bazhenovskoe chrysotile asbestos deposit, Central Urals, Russia. Paratobermorite significantly differs in the topology of crystal structure [mutual arrangement of (Si,Al)O4 tetrahedra and Ca polyhedra] from tobermorite and other tobermorite-supergroup minerals. Due to the original structure type and the presence of a significant amount of Al which substitutes Si, paratobermorite can be considered a novel microporous material and a perspective cation-exchanger.
Morphological and chemical characterization of secondary carbonates in the Toki granite, central Japan, and the evolution of fluid chemistry
https://doi.org/10.2138/am-2022-8229Yuguchi et al., in serial works (Yuguchi et al., 2015, 2019, 2021, and this study), provide new insights into the mass transfer due to hydrothermal alterations and groundwater-rock interactions. This study focuses on the petrography and mineral chemistry of calcites in the Toki granite, central Japan. It explores the sequential variations in mass transfer during the sub-solidus stages in granite, which were overlooked or not fully appreciated in earlier studies.
Characteristics and formation of corundum within syenite in the Yushishan rare metal deposits in the northeastern Tibetan Plateau
https://doi.org/10.2138/am-2022-8223Liu et al. present newly discovered in-situ corundum in syenite and discuss the characteristics, origin, and geological processes of corundum. Abundant mineral inclusions were found and categorized into primary and secondary based on morphological and reacting characteristics. They indicate two geological processes. Trace-element characteristics and oxygen isotopes are also consistent with corundum of magmatic origin. The implication is that corundum crystallized in melts with the involvement of Al-rich and Si-poor crustal material.
Hydrogen solubility in FeSi alloy phases at high pressures and temperatures
https://doi.org/10.2138/am-2022-8295Light elements alloying with metallic Fe can greatly affect its properties and play a key role in dynamics of planetary cores. H and Si are two candidate light elements in cores. However, the H storage in the Fe-Si system under relevant pressure and temperature is still unclear. Here Fu et al. found dramatic decrease of H content in FeSi alloys when Si is present. Their experiments indicate that H remains in the structure of FeSi alloys when recovered to 1 bar. The unusual property of FeSiHx alloys will help understand important geochemical processes involving hydrogen in future studies.
First evidence of dmisteinbergite (CaAl2Si2O8 polymorph) in high-grade metamorphic rocks
https://doi.org/10.2138/am-2022-8505Wannhoff et al. present the first finding and characterization of dmisteinbergite inside crystallized anatectic melt inclusions (MI) in garnet from three locations on two continents. These MI were originally entrapped inside a garnet growing or recrystallizing in presence of crustal melts during orogenesis continental collision under high T and highly variable P. The work on dmisteinbergite in anatectic MI, combined with previous results reported in literature, helps (a) to identify more precisely which factors and P-T conditions control the formation of this particular polymorph and (b) to better constrain the crystallization behavior of silicate melts in small pores. Overall, these new findings contribute to shed light on high T minerals which increasingly appear to be a common occurrence in the metamorphic evolution of the continental crust.
New Mineral Names
https://doi.org/10.2138/am-2022-NMN1071218November 2022
The Zn, S, and Cl isotope compositions of mare basalts: Implications for the effects of eruption style and pressure on volatile element stable isotope fractionation on the Moon
https://doi.org/10.2138/am-2022-8290The stable isotope compositions of volatile elements are sensitive tracers for the loss of volatiles throughout planetary evolution. Gargano et al. measure three distinct volatile element isotope systems of Zn, S, and Cl in a suite of lunar mare basalts to better understand the causal mechanisms for the Moon's depletion of volatile elements and wide-ranges in volatile element stable isotope compositions. Their data show that the Moon is enriched in the heavy isotopes of Zn, S, and Cl when compared to the Earth — likely resulting from vaporization associated with the Moon-forming Giant Impact. Additionally, they show that the Zn, S, and Cl isotope compositions of lunar low and high-titanium mare basalts are similar despite sampling reservoirs that crystallized early and late, respectively, from the Lunar Magma Ocean. As such, they conclude that the Giant Impact likely dominated the fractionation of these volatile-element stable isotope systems in the lunar interior, with minimal contribution from later mare volcanism.
An ab-initio study on the thermodynamics of disulfide, sulfide, and bisulfide incorporation into apatite and the development of a more comprehensive temperature, pressure, pH, and composition-dependent model for ionic substitution in minerals
https://doi.org/10.2138/am-2022-8250Recent studies of natural and experimental apatite demonstrate that the oxidation state of S (S6+, S4+, S1– and S2–) in apatite varies systematically as a function of oxygen fugacity (fO2). To establish a theoretical understanding of S in apatite, Kim et al. adopted quantum-mechanical methods to explore the thermodynamics and geometry of incorporation of disulfide (S22–), sulfide (S2–), and bisulfide (HS–) as possible forms of reduced S species in apatite.
Experimental partitioning of fluorine and barium in lamproites
https://doi.org/10.2138/am-2022-8289The halogens (F, Cl, Br, and I) are elements essential to life and are cycled by exchanges between the Earth's interior and the biosphere, hydrosphere, and atmosphere. Whilst some attention has been paid to the recycling of these elements on the surface of our planet, little is understood about their long-term storage within the Earth and how the exchange between the deep Earth and surface takes place over geological time. Lamproites, ultrapotassic melts, have the highest known F and Ba content of any primary mantle melts, but little is known about the source mineralogy that generates these unusual melts. Ezad and Foley experimentally determined the partitioning of F and Ba between phlogopite mica, a primary halogen-bearing mineral in the mantle, and lamproite melts. Using their partition coefficients, they investigate several mantle lithologies to determine which upon melting can produce lamproitic melts.
Nb and Ta intracrustal differentiation during granulite-facies metamorphism: evidence from geochemical data of natural rocks and thermodynamic modeling
https://doi.org/10.2138/am-2022-8260Both continental crust and depleted mantle are characterized by subchondritic Nb/Ta, leading to a mass imbalance compared to the bulk Earth. Even though several potential high Nb/Ta reservoirs in Earth's core and mantle have been proposed, little attention has been given to those in the crust. Huang et al. present bulk-rock and rutile geochemical data for samples of lower crustal pelitic granulite from the North China Craton, which exhibit systematic variations in Nb and Ta contents. High-temperature (HT) and ultra-high temperature (UHT) granulite residues exhibit Nb/Ta ratios that are close to chondritic and subchondritic, respectively; whereas leucosomes from UHT granulites mostly have higher Nb/Ta. These variations are best explained via competition for Nb and Ta between biotite and rutile during metamorphism, although initial bulk-rock Nb/Ta values also have an effect. When significant amounts of biotite remain in the residue, biotite dehydration melting generates a high-Nb/Ta residue and low-Nb/Ta melt because biotite preferentially incorporates Nb over Ta. However, geochemical modeling suggests that once biotite is depleted, the Nb/Ta ratio of the system is instead controlled by rutile growth, which promotes formation of a lower Nb/Ta residue and higher Nb/Ta melt, even though the amount of these melts might be lower. Huang et al. propose that in-situ to in-source leucosome and leucocractic veins in UHT terranes may maintain a high Nb/Ta geochemical signature, whereas residual crustal-derived A-type granite that experienced significant fractionation of Nb- or Ta-bearing minerals or contamination from other low-Nb/Ta sources could not maintain high Nb/Ta ratio, even though these ratios would still be higher than S-type granite.
Complexions and stoichiometry of the 60°//[100](011) symmetrical tilt grain boundary in Mg2SiO4 forsterite: a combined empirical potential and first-principles study
https://doi.org/10.2138/am-2022-8420Furstoss et al. show that multiple stoichiometric and non-stoichiometric grain boundary (GB) structures are stable for one particular GB in forsterite. Vacancies are, in most cases, attracted by the GBs. The atomic structures found here have never been described before and could explain the high diffusivity of oxygen at GB as well as part of the water storage in forsterite.
Effects of electronegativities and charge delocalization on vibrational properties of silicate crystals and glasses: Raman, XPS and simulation results
https://doi.org/10.2138/am-2022-8172Nesbitt et al. investigate Raman shifts of the A1 symmetric stretch of silicate Q2 species vary over a range of ~90 cm-1 in crystals and glasses containing alkali and alkaline earth oxides. The shifts display a striking, sympathetic relationship with the electronegativity of the alkali and alkaline earth metals (M), with the highest frequency observed for Mg-silicate glasses and crystals and the lowest frequency for Cs-bearing glasses.
The nyerereite crystal structure: a possible messenger from the deep Earth.
https://doi.org/10.2138/am-2022-8106Zucchini et al. studied the crystal structure of synthetic nyerereite [Na2Ca(CO3)2]. Na2Ca(CO3)2 likely has a wide stability field at pressure conditions higher than those of the synthesis experiments (100 MPa). New scenarios, where nyerereite would have a wide stability field at high-pressure conditions, strongly influence the mobility of carbonate melts in the deep upper mantle/transition zone and have significant implications for the deep carbon cycle associated with carbonatites.
Crystal structures and high-temperature and high-pressure vibrational spectra of synthetic fluorine-bearing brucites
https://doi.org/10.2138/am-2022-8215Miao et al. synthesized two F-bearing brucite samples, Mg(OH)1.78F0.22 and Mg(OH)1.16F0.84, at 9.5 GPa and 1373 K. Single-crystal X-ray diffraction measurements indicate that both the phases still crystallize in the symmetry of P3m1, and the fluorine effect on the crystal structure has been carefully characterized. Additional IR-active OH-stretching bands are observed at 3660, 3644, and 3513 cm-1 for the F-bearing samples, besides the original one at 3695 cm-1. In situ high-temperature and high-pressure Raman and Fourier transform infrared (FTIR) spectra were systematically collected on both the F-bearing samples and a natural one with 0.7 mol% F-. Miao et al. observed that the dehydration points of the synthetic F-bearing brucite samples are 800–850 K at the ambient pressure, which is 100–150 K higher than that for F-free samples in the atmosphere. In the presence of fluorine, hydrous minerals, including brucite, could be stable at higher P-T conditions in subduction zones, which will provide us new insight into the combination of hydrogen and fluorine cycling in the subduction slabs. Fluorine substitution generates new H positions at lower energies (frequencies) and stabilizes the lattice structure, which has a pronounced impact on the thermoelastic properties, and fluorine could promote hydrogen bond symmetrization in hydrous minerals at high pressures. And, fluorine incorporation will affect the hydrogen isotopic fractionation between hydrous minerals and water.
The influence of OH content on elastic constants of topaz [Al2SiO4(F,OH)2]
https://doi.org/10.2138/am-2022-8159Topaz might play a significant role in transporting water and fluorine into the Earth's interior in subduction zones. Seismological detection of topaz gives us insights into the transport of water and fluorine and requires a thorough understanding of its elastic properties. The influence of OH content on elastic constants of topaz has not been fully understood, though experimental and theoretical studies have been done on topaz with various OH contents. Aradachi et al. determined elastic constants of topaz for five natural single-crystal specimens with different OH content (x=0.28~0.72) via the sphere resonance method at ambient conditions. Their measured elastic constants are significantly higher (>3%) than theoretically determined values, especially in diagonal components (Cii). The theoretical lower values must be related to the lattice parameters used in the theoretical studies, which are systematically larger than measured lattice parameters. The theoretical approach should be modified to reproduce measured lattice parameters and lead to agreement of theoretical and experimental elastic constants. These results provide a clue to a better understanding of elasticity of topaz and a basis for the seismological detection of subducted oceanic sediments.
Experimental calibration of an Fe3+/Fe2+-in-amphibole oxybarometer and its application to shallow magmatic processes at Shiveluch Volcano, Kamchatka
https://doi.org/10.2138/am-2022-8031Goltz et al. present an experimentally calibrated amphibole-melt oxybarometer applicable to mafic and felsic systems with no coexisting oxides. Using the partitioning of Fe3+ and Fe2+, the oxygen fugacities of magmas at Shiveluch Volcano are calculated. Some samples with oxidized amphibole reflect pre-eruptive dehydrogenation; diffusion modeling of H is used to determine the timing of mafic recharge events. Not all recharge events are directly linked to eruption.
The crystal structure of mineral magadiite, Na2Si14O28(OH)2*8H2O
https://doi.org/10.2138/am-2022-8156Marler et al. describe the complex structure of mineral magadiite, a mineral known for 54 years and discovered at Lake Magadi, Kenya. Magadiite has also frequently been synthesized in the lab to act as a precursor material for a subsequent transformation into porous silicates. Its structure, however, remained obscure until now due to a limited crystallinity, small particle size, and complex structure. Magadiite is a layered silicate containing thick silicate layers with intercalated bands of hydrated sodium cations.
Tin isotopes as geochemical tracers of ore-forming processes with Sn mineralization
https://doi.org/10.2138/am-2022-8200Zhou et al. show that kinetic Rayleigh fractionation is likely to lead to isotope fractionation of tin during the formation of the ores, that liquid-vapor partitioning or vaporization, and fluid-rock interaction have strong effect on Sn isotope fractionation, and that Sn isotopes can be a robust tool for tracing mineralization centers, fluid pathways, migration mechanisms of tin, and a powerful probe of redox processes.
The role of graphite in the formation of unconformity-related uranium deposits of the Athabasca Basin, Canada: A case study of Raman spectroscopy of graphite from the world-class Phoenix uranium deposit
https://doi.org/10.2138/am-2022-8158Unconformity-related uranium (URU) deposits in the Proterozoic Athabasca Basin (Canada) represent the richest and one of the most important uranium endowments in the world. Song et al. use the Phoenix uranium deposit in the southeastern Athabasca Basin as a case study to address the association of URUs with graphitic basement faults. Petrographic studies indicate that there is no direct contact between graphite and uraninite at microscopic scales, and the content of graphite in the graphitic metapelite along the ore-controlling WS Shear Zone does not show a systematic change with the distance from the unconformity surface. Raman spectroscopic studies of graphite suggest that the degree of structural disorder of graphite does not change systematically with the distance from the unconformity surface either. The minor irregularities in these parameters near the unconformity are better explained by paleo-weathering related to the unconformity and/or diagenetic processes than by hydrothermal activity related to uranium mineralization. Based on these observations and interpretations, the role of graphite as an in situ reducing agent, either directly or as a provider of hydrocarbons, is discounted. It is proposed that hydrocarbons derived from graphite at depth, tapped by episodic reactivation or seismicity of the basement faults that were facilitated by graphite as a lubricant, were responsible for URU mineralization.
Pomite and pseudopomite, two new carbonate-encapsulating mixed-valence polyoxovanadate minerals
https://doi.org/10.2138/am-2022-8335The discovery of POM minerals is one of the specific landmarks of descriptive mineralogy and mineralogical crystallography of our time. In this paper by Kampf et al., the most recently discovered POM minerals, pomite and pseudopomite, are the first to incorporate carbonate as the central heteropolyhedral group of the POM. Synthetic carbonate-bearing POMs have been suggested as useful for the capture of atmospheric carbon, and naturally occurring phases, such as pomite and pseudopomite, may prove to be important guides in that respect.
Book Review: Triple Oxygen Isotope Geochemistry
https://doi.org/10.2138/am-2022-B1071117October 2022
Experimentally derived F, Cl, and Br fluid/melt partitioning of intermediate to silicic melts in magmatic systems
https://doi.org/10.2138/am-2022-8109Understanding the behavior of halogens in magmas is of importance for a range of crustal processes, including magma evolution and degassing and ore mineralization, by influencing the physical and chemical properties of melts, fluids, and minerals. To this end, the study by Cassidy et al. uses experiments and chemical analyses to define under what conditions these halogens prefer the fluid (or gas phase) relative to the melt. These data provide a framework for interpreting halogens gases measured within volcanic plumes.
Spectroscopic study on the local structure of sulfate (SO42–) incorporated in scorodite (FeAsO4∙2H2O) lattice: Implication for understanding the Fe(III)-As(V)-SO42–-bearing minerals formation
https://doi.org/10.2138/am-2022-8184Incorporation of SO42- in the scorodite structure is an important process during its formation in mining-related settings such as acid mine drainage and hydrometallurgical tailings. In addition, the incorporation of SO42- in scorodite is also an important mechanism during the immobilization of arsenic in waste sulfuric acid streams that use scorodite as an As-carrier. The incorporated structural SO42- can reduce the scorodite stability and then affect the mobility and fate of arsenic. The study by Ma et al. indicates that the SO42- may form Fe2(SO4)3-like local structure in scorodite. Hence, higher solubility of local Fe2(SO4)3 (vs. FeAsO4∙2H2O) trapped in scorodite may be the main reason for the higher solubility of SO4-scorodite. Furthermore, the structure of SO4-scorodite solid solution can also help understand the evolution and crystallization processes of its precursors, i.e., amorphous ferric arsenate.
Oxidation of arcs and mantle wedges by reduction of manganese in pelagic sediments during oceanic subduction
https://doi.org/10.2138/am-2022-8171Song et al. investigated high-pressure metamorphosed Mn-rich cherts in ancient seafloors in the Qilian subduction zone, and demonstrated that reductive reactions of Mn-rich cherts in the subduction zone are an important factor for mantle wedge oxidization and oxygen recirculation. They propose that the oxygen flux generated by progressive metamorphism of Mn-rich pelagic sediments is high enough for oxidizing the sub-arc mantle, while the ferric iron plays very little role in the oxidization of the sub-arc mantle wedge.
Raman scattering and Cr3+ luminescence study on the structural behavior of δ-AlOOH at high pressures
https://doi.org/10.2138/am-2022-8005Wang et al. conducted Raman scattering and luminescence spectroscopic measurements on δ-AlOOH at pressures up to 34.6 GP and 22.1 GPa, respectively. From the Raman spectra, the Raman-active B2 modes at 390 cm-1 and 416 cm-1 show a continuous hardening behavior but become abnormally stiffened at ~8 GPa, in agreement with the structural transition from P21nm to Pnnm. These two modes are highly sensitive to pressure in the P21nm phase but less so in the Pnnm phase, which are consistent with the different compressibilities of two phases. Additionally, several softening modes were observed in the P21nm phase, which show a gradual decrease in intensity and finally disappear before the P21nm-to-Pnnm phase transition. The luminescence data also show systematic abrupt changes in the slopes or pressure dependences of the R-lines and sidebands across the phase transition. The evolution of R2-R1 splitting with pressure indicates continuously suppressed distortion of AlO6 octahedron under high pressure.
Jadeite and related species in shocked meteorites: Limitations on inference of shock conditions
https://doi.org/10.2138/am-2022-8220Jadeite has been frequently reported in heavily shocked meteorites. It displays a variety of textures and grain sizes, and many researchers identified it solely on the basis of its Raman spectra. Here Baziotis et al. show that additional techniques are required in order to confidently identify jadeite and distinguish it from related species. A reassessment of reported jadeite occurrences puts in a new light on many previous conclusions about the shock histories preserved in particular meteorites.
Pressure-induced C23-C37 transition and compression behavior of orthorhombic Fe2S to Earth's core pressures and high temperatures
https://doi.org/10.2138/am-2022-8187Fe2S was recently identified as a plausible iron-rich sulfide stable at Earth's inner core boundary conditions. Zurkowski et al. present the mechanism for a pressure-induced phase transition in Fe2S around Earth's core-mantle-boundary pressures. Up to 120 GPa, Fe2S exhibits the Co2P structure type (C23), and a highly compressible a axis compared to the b and c axes. In the 120 to 150 GPa pressure range, a change in compressibility is observed, such that the C23 Fe2S structure evolves towards a more Co2Si-like structure (C37). Above 150 GPa, this transition is complete and C37 Fe2S is observed to at least 306 GPa based on previous studies. An equation of state of the C37 Fe2S phase is presented and compared to the density of Earth's Fe-rich outer core. Based on these results, an Fe-S alloy alone cannot match the geophysical constraints of Earth's core and likely S plus other light elements such as Si, O, C, and H are present.
Estimation of ferric iron contents in clinopyroxene by machine learning models
https://doi.org/10.2138/am-2022-8189Huang et al. demonstrate that: (1) machine learning models provide reliable predictions of clinopyroxene ferric iron content; (2) supervised training models can significantly minimize temperature uncertainties of clinopyroxene-garnet geothermometers; and (3) the distribution of Fe3+/ΣFe values differs slightly between dehydrogenated and non-dehydrogenated clinopyroxenes.
Pyradoketosite, a new, unexpected, polymorph of Ag3SbS3 from the Monte Arsiccio mine (Apuan Alps, Tuscany, Italy)
https://doi.org/10.2138/am-2022-8136Biagioni et al. describe the new mineral pyradoketosite, a new polymorph of the well-known compound Ag3SbS3. This compound has been known for two centuries as the trigonal and monoclinic polymorphs pyrargyrite and pyrostilpnite, respectively, that represent important Ag ore minerals and, more recently, are used for their thermoelectric and optical properties. It is worth noting that previous synthesis experiments in the Ag-Sb-S system were not able to obtain the synthetic analogue of pyradoketosite. The study of natural occurrences of Ag-sulfosalts can not only contribute to the knowledge of Ag crystal chemistry, but also may have interesting technological implications.
Pyrite geochemistry and its implications on Au-Cu skarn metallogeny: An example from the Jiguanzui deposit, Eastern China
https://doi.org/10.2138/am-2022-8118This study by Zhang et al. highlights the significance of wallrock hydraulic fracturing, fluid-rock reaction, and the fluid physicochemical evolution in the skarn ore-forming process, and suggests the potential use of pyrite trace element geochemistry for exploring Au-Cu skarn mineral systems.
Synthesis of ferrian and ferro-saponites: Implications for the structure of (Fe,Mg)-smectites synthesized in reduced conditions
https://doi.org/10.2138/am-2022-8231The smectite found at the Yellowknife Bay of Mars, surprisingly, may be characterized as a ferrian saponite, which has unique octahedral sheets based on the traditional clay mineralogy. Here, Sakuma et al. synthesized ferrian saponites using a hydrothermal method under reduced conditions. The stability of ferrian saponite can be explained by the desorption of hydrogen in the structure to neutralize the charge. The valence state of part of the iron cations in the octahedral sheets is reversible. These results provide a basis for estimating the paleoenvironments of Mars and Ceres using iron smectites.
Natural cubic perovskite, Ca(Ti,Si,Cr)O3-δ, a versatile potential host for rock-forming and less-common elements up to Earth's mantle pressure
https://doi.org/10.2138/am-2022-8186Britvin et al. report the discovery of a natural cubic perovskite. This is the first example of natural ABO3 perovskite bearing disordered oxygen vacancies. The mineral contains more than 20 mol% of davemaoite component (cubic perovskite-type CaSiO3) and is abnormally enriched in Cr. It remains stable upon compression to at least 50 GPa. Cubic perovskite can serve as a structural host for incorporation of different elements up to a pressure corresponding to the Earth's mantle conditions.
Nazarovite, Ni12P5, a new terrestrial and meteoritic mineral structurally related to nickelphosphide, Ni3P
https://doi.org/10.2138/am-2022-8219Britvin et al. describe Nazarovite, Ni12P5, a new natural phosphide belonging to the system Fe-Ni-P that is the main reservoir of reduced (non-phosphate) phosphorus in the Solar System. Due to the crystal-chemical features, the mineral may serve as a chemical discriminator, selectively extracting Ni from phosphide-bearing Fe-Ni metal systems.
Zinconigerite-2N1S ZnSn2Al12O22(OH)2 and zinconigerite-6N6S Zn3Sn2Al16O30(OH)2, two new minerals of the nolanite-spinel polysomatic series from the Xianghualing skarn, Hunan Province, China
https://doi.org/10.2138/am-2022-7983Rao et al. report the occurrence of two new polysomes of the N-S type polysomatic series. They describe the detail chemical and structural information of zinconigerite-2N1S and -6N6S. This discovery provides new insights into the crystal chemistry of N-S polysomatic series and their origin.
Tracing structural relicts of the ikaite-to-calcite transformation in cryogenic cave glendonite
https://doi.org/10.2138/am-2022-8162Nemeth et al. show that glendonite, a calcite pseudomorph after ikaite (CaCO3∙6H2O) and commonly interpreted as an indicator of low paleotemperature, is characterized by high intergranular micrometer-size porosity, whereas calcite crystals, the constituent of glendonite, are consistent with a relatively low-porosity material. Although the aligned mesopores, frequently occurring twins, small grain size, presence of aqueous inclusions and the observed porosity arise from the ikaite-calcite transformation, the glendonite morphology remains the most reliable feature to identify calcite derived via transformation of ikaite and thus to infer past cryogenic temperatures. These findings have implications for the emerging research field of cryogenic caves carbonates, which are commonly associated with the presence of permafrost.
Oxygen-fugacity evolution of magmatic Ni-Cu sulfide deposits in East Kunlun: Insights from Cr-spinel composition
https://doi.org/10.2138/am-2022-8050Jia et al. provide the first comparison of the magma fO2 for the Xiarihamu Ni-Cu sulfide deposit and Shitoukengde mafic-ultramafic intrusion based on olivine-spinel oxybarometry. The authors propose that the temporal evolution of oxygen fugacity of mantle-derived magma can be a key indicator for evaluating metallogenic potential of Ni-Cu sulfide deposits and that reduction processes from mantle source to shallow crust play an important role in the genesis of magmatic Ni-Cu sulfide deposits.
New Mineral Names
https://doi.org/10.2138/am-2022-NMN1071016September 2022
Apollo 15 regolith breccia provides first natural evidence for olivine incongruent melting
https://doi.org/10.2138/am-2022-8121The present-day architecture of the solar system is the result of countless collisions between astronomical objects. During impacts, the energy released by shock waves triggers pressures and temperatures comparable to those of planetary interiors. Minerals can form during impacts, and through their identification, it is possible to establish impact pressure and temperature conditions. In this study, Satta et al. describe for the first time the presence of ferropericlase — a mineral typically confined to depths about 660 km in the Earth — found in a lunar rock collected during the Apollo 15 mission. Their observations suggest that the lunar ferropericlase formed as a result of a shock-induced process never observed before in natural samples. This study provides a new understanding of impacts on the Moon's surface.
Enhanced weathering in the seabed: Rapid olivine dissolution and iron sulfide formation in submarine volcanic ash
https://doi.org/10.2138/am-2022-8057Within volcanic ash particles recovered from a seamount in 3000 m water depth, olivine dissolution and related iron sulfide formation have been observed by Kahl et al. The FeS2 precipitates are found to be conjoined and planar aggregates, occurring in the center of cavities that formed by partial to complete dissolution of olivine. Almost all occurrences of FeS2 precipitation are related to Cr-spinel inclusions in the former olivine. Iron sulfide formation involves initial heterogeneous nucleation of FeS2 on spinel surfaces and subsequent pyrite and/or marcasite precipitation as overgrowths on existing FeS2 aggregates. Rapid olivine dissolution is facilitated by sulfide-bearing fluids, which prevent the formation of hydrous ferric oxide layers and promote sequestration of iron as FeS2. These delicate structures of FeS2 within voids created by olivine dissolution may represent a transient in-seafloor alteration, as progressive dissolution of volcanic glass causes the formation of Fe-oxyhydroxides and clay minerals, which are the commonly observed alteration features in seafloor basalts.
The efficiency of copper extraction from magma bodies: Implications for mineralization potential and fluid-silicate melt partitioning of copper
https://doi.org/10.2138/am-2021-7951This study by Zhou et al. indicates that the efficiency of copper extraction from magmas plays a critical role in determining Cu mineralization potential. Their results indicate that a variety of igneous minerals with anomalously low Cu contents could potentially be used as a tool to identify a Cu-mineralizing magma body in a deposit with multiphase intrusions; nevertheless, a suite of igneous mineral compositions from a region should be analyzed for comparison. The results suggest that the inefficient copper extraction from plutons may be ascribed to the lack of reduced S species during fluid exsolution or different evolution paths of Cu and Cl during magma crystallization.
Validation of clinopyroxene-garnet magnesium isotope geothermometer to constrain the peak metamorphic temperature in ultrahigh-temperature ultramafic-mafic granulites
https://doi.org/10.2138/am-2022-8058In this study by Gou et al., on the basis of evidence from phase equilibrium modeling, mineral chemical analysis, and conventional clinopyroxene-garnet Fe-Mg exchange geothermometry and clinopyroxene-garnet Mg isotope geothermometry, they evaluate the feasibility of a clinopyroxene-garnet Mg isotope geothermometer in the Neoarchean ultramafic-mafic granulites from the Namakkal Block of the Southern Granulite Terrane, India, which underwent ultrahigh-temperature (UHT) metamorphism, and the effect of the retrograde cooling on equilibrium Mg isotope fractionation between clinopyroxene and garnet during the UHT metamorphism. They demonstrate that the inter-mineral Fe-Mg exchange between clinopyroxene and garnet during the retrograde cooling of the ultrahigh-temperature (>900 °C) did not disturb their Mg isotope fractionation equilibrium attained during the ultrahigh-temperature metamorphic condition. Therefore, clinopyroxene-garnet Mg isotope thermometry is a valid tool to constrain the peak metamorphic temperature conditions even for ultramafic-mafic granulites that have undergone ultrahigh-temperature metamorphism.
Uranotungstite, the only natural uranyl tungstate: Crystal structure revealed from 3D electron diffraction
https://doi.org/10.2138/am-2022-8112The structure of the only U(VI) tungstate known from nature has been characterized from a nano-size crystal by means of the 3D electron diffraction techniques. This work by Steciuk et al. is the first structural description of a natural uranyl-tungstate mineral and confirms the great structural and chemical flexibility of the beta-U3O8 type of sheets.
Carbon flux and alkaline volcanism: Evidence from carbonatite-like carbonate minerals in trachytes, Ulleung Island, South Korea
https://doi.org/10.2138/am-2022-8007This paper by Chen et al. presents a detailed mineral chemical study of the trachyte and trachyandesite of Ulleung Island. Petrographic and chemical properties of different types of carbonate and silicate melt products in the rocks have been thoroughly analyzed. The carbonate phases include the following types, (1) euhedral pseudomorphic ankerite grains, (2) spherical and dumbbell shape globules in a trachyte matrix, (3) irregular-shaped inclusions in biotite and ulvospinel, and (4) hexagonal shaped inclusions in biotite. The C-O isotopes have δ13C (VPDB, ‰) ranging from -3.98 to -5.76, δ18O (SMOW, ‰) from 4.43 to 11.49, and exhibit igneous characteristics. Based on their appearances, the carbonate phases were formed from carbonate melt generated by carbon flux from the subducted oceanic slab. The pseudomorph carbonate grains were altered from pyroxene in the lower crust or the bottom of the fractionated magma chamber. The volcanic system of Ulleung Island preserves extraordinary evidence of carbon activity within the lower crust. The recycled carbon from the subducted slab can be stored within the lower crust through the replacement of silicate minerals. The calculation of carbon circulation in the earth system may need to consider adding this parameter in the modeling.
Controls on the formation of porphyry Mo deposits: Insights from porphyry (-skarn) Mo deposits in northeastern China
https://doi.org/10.2138/am-2021-7665The results presented in this study by Ouyang et al. suggest that unique magma sources are not required for the formation of porphyry Mo deposits of both arc-related and Climax-type. Although the two classes of porphyry Mo deposits were formed in distinct tectonic settings and are associated with different kinds of intrusions, their formation is fundamentally controlled by similar geological processes and/or factors. A prerequisite for the formation of porphyry Mo deposits of both arc-related and Climax-type is the emplacement of oxidized, water-saturated magmas at 1.5 to 6.0 km palaeodepth. However, the actual ore-forming event itself is considered to relate to a sudden depressurization of the magma chamber and consequent venting of voluminous fluids along focusing structures, such as small stocks or dike swarms. These results, in combination with an examination of other porphyry Mo systems, suggest that sudden depressurization of magma chambers and subsequent venting of voluminous fluids along focusing structures most likely plays a critical role in the formation of porphyry Mo deposits of both arc-related and Climax-type. The findings of this study indicate that fluid processes in the shallow crust are pivotal for the formation of porphyry Mo deposits and that settings with ideal magmatic-hydrothermal architectures are most likely to develop into productive porphyry Mo systems.
High-pressure synchrotron single crystal X-ray diffraction study of lillianite
https://doi.org/10.2138/am-2021-7765In this paper by Zucchini et al., the high-pressure (HP) synchrotron X-ray diffraction study of lillianite up to approximately 21 GPa allowed them to (1) confirm the reversible first-order phase transition to beta-Pb3Bi2S6 that was bracketed between 4.90 and 4.92 GPa; (2) analyze the phase transition mechanism, as well as the compressibility of both lillianite and beta-Pb3Bi2S6; (3), observe a pseudomerohedral twinning that disappeared on decompression making lillianite an interesting shape-memory material; and (4) conclude that the stronger the stereochemical activity of the LEP, the higher the stability of the crystal structures at HP.
Thermoelastic parameters of Mg-sursassite and its relevance as a water carrier in subducting slabs
https://doi.org/10.2138/am-2022-8034This work by Sula et al. presents new thermoelastic parameters and the structural evolution of Mg-sursassite at high-pressure and high-temperature, obtained by in-situ, synchrotron-radiation, single-crystal diffraction measurements, in relation with its mineral chemistry. Mg-sursassite is a hydrous mineral that potentially takes part, together with other hydrous minerals (e.g., Dense Hydrous Magnesium Silicates), in the mechanism of sequestration and release of water in the Earth's interior from subducted slabs.
3D crystal size distributions of pyroxene nanolites from nano X-ray computed tomography: Improved correction of crystal size distributions from CSDCorrections for magma ascent dynamics in conduits
https://doi.org/10.2138/am-2022-8039Crystal size distributions (CSDs) are commonly used in igneous petrology because they can provide important information on crystallizing conditions and syneruptive magma dynamics. Okumura et al. proposed CSDs plotted against short-axis length (S-plot CSDs) as a new method. In contrast to the prevalent CSDs plotted against long-axis length, S-plot CSDs suppressed errors and also showed differences according to eruption style more markedly (by ~20% more) than using the long-axis length. Their findings make it possible to obtain more reliable and informative CSDs and infer variations in magma ascent dynamics.
Amphibole fractionation and its potential redox effect on arc crust: Evidence from the Kohistan arc cumulates
https://doi.org/10.2138/am-2022-8141Zhang et al. discovered that Kohistan amphibole-bearing rocks are of cumulate origin; Fe3+/ΣFe ratios of Kohistan arc cumulates are mainly controlled by amphibole; amphibole fractionation leads to oxidation of arc magma, and amphibole fractionation is key to porphyry deposit formation and crustal evolution.
A comparative study of two-phase equilibria modeling tools: MORB equilibrium states at variable pressure and H2O concentrations
https://doi.org/10.2138/am-2022-8211The development and ubiquitous usage of phase equilibria represent an inflection point in our ability to understand the link between petrogenesis and tectonics. However, the choice between different thermodynamic models raises important questions when modeling systems where multiple modeling tools are applicable, such as at suprasolidus conditions. Hernandez-Uribe et al. systematically compare the results of two modeling tools commonly used in the igneous and metamorphic petrology communities: MELTS (Ghiorso et al. 2012; Ghiorso & Gualda 2015) and the metabasite set of Green et al. (2016) using the thermodynamic database ds62 (Holland & Powell 2011), respectively. They selected a N-MORB composition and modeled closed-system equilibrium phase relations as a function of temperature at 0.25 GPa and 1 GPa for compositions with 0.5 wt% and 4 wt% H2O. Their results show that phase relations do exhibit some key differences that can impact geological inferences based on the modeling tool used for calculations. Differences in modal abundances of phases and liquid compositions correlate with predicted differences in liquid trace-element signature. The critical insight of this study is to inform the users about the differences in modeling approaches and how these can have important implications for interpretations of geologic processes and their associated uncertainties.
On the occurrence of Jahnsite/Whiteite phases on Mars: A thermodynamic study
https://doi.org/10.2138/am-2022-8174(See also Graphical Abstract on the website). The thermodynamic predictions by Drouet et al. and predominance/stability calculations and experimental data strongly suggest the very unlikely presence of Jahnsites/Whiteites in Gale Crater on Mars, which was recently hypothesized from Curiosity analyses. Jahnsites are not favorable phases to form directly, contrary to other more favorable phases, and extrapolation to present and past Martian conditions also argues against their direct precipitation at the surface of Mars or their preservation if formed indirectly.
Hydroxymcglassonite-(K) the first Sr-bearing member of the apophyllite group, from the Wessels mine, Kalahari Manganese Field, South Africa
https://doi.org/10.2138/am-2022-8210In this work by Yang et al., a new mineral species, hydroxymcglassonite-(K), has been found in the Wessels mine, Kalahari Manganese Field, Northern Cape Province, South Africa. It is isostructural with hydroxyapophyllite-(K) and represents the first Sr-bearing mineral of the apophyllite group.
August 2022
Estimating kaolinite crystallinity using near-infrared spectroscopy: Implications for its geology on Earth and Mars
https://doi.org/10.2138/am-2022-8025Pineau et al. investigated the near-infrared (NIR) spectral signatures of kaolinite to see if they can be used as a proxy of kaolinite crystallinity. The authors acquired NIR reflectance spectra of terrestrial kaolinites with well-constrained geological origins and estimated their crystallinity using XRD. They then studied the NIR signature of kaolinite by using specific spectral criteria developed using the second and third derivative NIR spectra. They compared the spectral criteria values with the XRD indices and observed high correlations between these, showing that NIR signature of kaolinite can be used as a semi-quantitative proxy to estimate its crystallinity. The authors also show that the crystallinity of kaolinite can be used as a proxy to determine its geological origin: (1) well-ordered kaolinites are not restricted to hydrothermal deposits; (2) kaolinites from a similar sedimentary or pedogenetic context can have contrasting crystallinities; (3) poorly crystalline kaolinites are more likely to have a sedimentary or pedogenetic origin. This study provides a methodology to quickly estimate the crystallinity of a kaolinite sample using NIR data, which gives important information about its geology. This work has implications in diverse domains, from industrial exploitation of kaolin to planetary exploration.
The interplay between twinning and cation inversion in MgAl2O4-spinel: Implications for a nebular thermochronometer
https://doi.org/10.2138/am-2021-7874Manga et al. conducted a density-functional-theory study of the thermodynamics of spinel twins. They report a stable-cation inversion associated with the planar fault. The predicted cation-inversion near the twin is in stark contrast to the inversion that is known for the bulk spinel and has implications for different fields ranging from materials science to earth and planetary sciences. To the field of cosmochemistry, the study reports a nebular thermochronometer based on the predicted twin-induced cation-inversion.
The effect of fluorine on reaction rim growth dynamics in the ternary CaO-MgO-SiO2 system
https://doi.org/10.2138/am-2022-8123CCBYReaction rims are a border of secondary minerals formed at the margin of a primary grain in metamorphic rocks. Several features of reaction rims, such as the layer (phase) sequence, the relative layer thickness, and the internal microstructure, contain valuable information about the respective rim formation conditions, which all may be affected by the presence of volatile components. These features of reaction rims may, therefore, not only be used to reconstruct the P-T-t history of a metamorphic rock but also can tell something about the fluid composition in fluid-mediated metasomatic reactions. Frank and Joachim-Mrosko investigated the effect of fluorine on the growth dynamics and microstructure of reaction rims. Results show that natural reaction rims may have the potential to serve as "geofluidometers", which would be of great importance for samples that have lost all direct clues, such as fluid inclusions that usually allow us to unravel the chemical composition of metasomatic fluids.
Seeing through metamorphic overprints in Archean granulites: Combined high-resolution thermometry and phase-equilibrium modeling of the Lewisian Complex, Scotland
https://doi.org/10.2138/am-2022-8214CCBYThe Lewisian Complex in NW Scotland presents a record of the transition from the Neo-Archean to the Paleoproterozoic. However, this record is complicated by a long and varied history after peak metamorphism that has erased and/or partially reset much of the early history of the rocks. Such overprinting is a common feature of Archean granulites, and it poses a substantial problem when trying to understand the tectonic processes that were active prior to the onset of modern plate tectonics. By combining careful petrography with phase diagram modeling and a range of exchange thermometers, Gopon et al. obtained the peak and retrograde temperature history of the Lewisian Complex from a single, well preserved, representative sample of garnet-bearing mafic granulite. They present the application of high-resolution electron probe microanalysis (HR-EPMA) to sub-micrometer orthopyroxene exsolution lamellae in clinopyroxene. They discuss ways to mitigate issues associated with HR-EPMA including surface contamination, beam drift, standards, and the need to correct for secondary fluorescence effects. The resulting compositions from HR-EPMA analyses provide an independent measure of the retrograde temperature conditions and can also be used to back-calculate the compositions of clinopyroxene in the peak assemblage. They obtain peak metamorphic conditions for the Lewisian of > 11 kbar and >1025 °C and constrain subsequent metamorphic overprints to 850 °C (Grt-Cpx), 590 °C (Opx-Cpx), and 460 °C (Mag-Ilm).
Interphase misorientation as a tool to study metamorphic reactions and crystallization in geological materials
https://doi.org/10.2138/am-2021-7902Morales presents a new method for the study of orientation relationships in minerals based on interphase misorientation angle/axes, which can be calculated from crystallographic orientation data acquired using electron backscatter diffraction. The results using this methodology have been obtained for several examples, including clacite-aragonite, olivine-antigorite, magnetite-hematite, plagioiclase-olivine, and plagioclase-ilmenite.
Trace-element partitioning between olivine and melt in lunar basalts
https://doi.org/10.2138/am-2022-7971Chen et al. found that the partition coefficients for most elements between olivine and melt in lunar basalts (LB) fall within the range in terrestrial basalts (TB), except for three elements, Li, V, and Cr. Li partition coefficient (DLi) in LB is higher than in TB due to compositional differences between LB and TB. DV in LB is higher than in TB due to fO2 difference. DCr in LB is lower than in TB due to a combination of fO2 and compositional difference. Even though Cr is compatible and V is fairly incompatible during TB evolution, Cr is an incompatible element in primitive LB, with a similar degree of incompatibility as V, leading to nearly constant V/Cr ratio of 0.039 in lunar basalts.
Solving the iron quantification problem in low-kV EPMA: An essential step toward improved analytical spatial resolution in electron probe microanalysis-Fe-sulfides
https://doi.org/10.2138/am-2022-8027In electron probe microanalysis (EPMA), the decrease of the electron beam size permitted by Schottky field emission sources combined with the reduction of the electron interaction volume obtained by decreasing the accelerating voltage from traditional 15-20 kV to 7 kV, may allow for accurately quantifying submicrometer-sized features. Moy et al. describe two low accelerating voltage EPMA techniques to quantify Fe. The first method uses the low-intensity Fe Ll X-ray line, while the second method uses the area of the combined Fe Lalpha-Lbeta X-ray lines. The second method is based on a calibration curve that can be applied to any electron microprobe, for measurements performed at 7 kV with 40 ° takeoff angle spectrometers, without having to recalibrate. The calibration curve is used to convert experimental Fe Lalpha-Lbeta area k-ratios, calculated with a pure metallic Fe standard, into Fe concentrations. Both methods have been successfully applied to the quantification of Fe-sulfide specimens, producing substantial improvements compared to the traditional EPMA quantification method, which uses the Fe Lbeta X-ray line.
Zircon geochronological and geochemical insights into pluton building and volcanic-hypabyssal-plutonic connections: A complex intraplate alkaline volcano
https://doi.org/10.2138/am-2021-7861Scarrow et al. studied the Oki-Dozen ocean island complex intraplate alkaline volcano, Sea of Japan, which preserves temporally associated evolved intrusions, dikes, and lavas, revealing magma storage architecture and processes. SHRIMP U-Th-Pb zircon ages, 6.4 to 5.7 Ma, combined with compositional data, show the pluton formed by low flux protracted amalgamation of discrete magma pulses. The rhyolite dike preserves an evolved segregate of these, whereas the trachyte magma, derived from the same source, stalled at relatively shallow depths prior to eruption.
Using cathodoluminescence to identify oscillatory zoning of perthitic K-feldspar from the equigranular Toki granite
https://doi.org/10.2138/am-2022-8146Yuguchi et al. report on novel use of cathodoluminescence (CL) in observing oscillatory zoning in perthitic K-feldspars from the equigranular Toki granite, central Japan. Based on the CL patterns, two types of zoning were identified: single core oscillatory zoning (SCOZ) and multiple core oscillatory zoning (MCOZ). This study demonstrates that CL patterns can reveal crystal growth behavior and nucleation of magmatic K-feldspar, which are useful for revealing the spatial extent of element partitioning between the melt and crystal and that of mass transfer from the melt into the crystal during the evolution of magma chamber processes.
Influence of intensive parameters and assemblies on friction evolution during piston-cylinder experiments
https://doi.org/10.2138/am-2022-7958Piston-cylinder presses are used worldwide to mimic the pressure and temperature conditions in the planetary interiors up to ~5 GPa. However, the accuracy of targeted pressure during an experiment is highly debated because piston-cylinder assemblies exhibit inhomogeneous pressure distributions and biases compared to the theoretical pressure applied to the hydraulic press. Condamine et al. conducted almost 40 experiments to provide an overview of the cell performance for the most common pressure media and their applicability for Earth science experiments.
Formation process of Al-rich calcium amphibole in quartz-bearing eclogites from the Sulu belt, China
https://doi.org/10.2138/am-2022-7996Enami et al. studied the Al-rich and Si-poor amphibole in quartz-bearing (Si-saturated) eclogites from the Sulu ultrahigh-pressure (UHP) metamorphic belt by electron probe micro analyzer (EPMA), focused ion beam system (FIB)-transmission electron microscope (TEM), and electron backscattered diffraction (EBSD) method, and discussed why the Al-rich and Si-poor amphibole formed in quartz-bearing (Si-saturated) rocks. This amphibole occurs around garnet porphyroblast, and these two phases have characteristically very similar molecular Al/Si and Ca/Si values to each other. The Al-rich and Si-poor amphibole is a pseudomorph after garnet and formed by the preferential diffusions of Mg, Fe2+, and alkali elements between garnet and the matrix during exhumation and hydration stages of the Sulu eclogites. In contrast, Si, Al, and Ca, which have smaller diffusion coefficients, probably did not migrate much and contributed to the growth of the Al-rich and Si-poor amphibole with little change in their ratios from the garnet value. The formation process of the amphibole is discussed in relation to the very rapid exhumation rate of the Sulu UHP metamorphic belt, which has been proposed by many studies.
Helvine-danalite mineralogy of the Dulong Sn-Zn polymetallic deposit in SE Yunnan, SW China
https://doi.org/10.2138/am-2021-7919Southeastern Yunnan is a major Sn polymetallic province of China, with the Dulong large Sn-Zn polymetallic deposit being one of the most representative deposits. Liu et al. investigated helvine-danalite mineralogy of the Dulong Sn-Zn deposit and showed that (1) helvine-group minerals at Dulong include oscillatory-zoned helvine-danalite; (2) oscillatory zoning mainly formed by fluctuations of the ore-fluid fS2 and fO2; (3) helvine zone likely formed under higher fS2 and lower fO2 than the danalite zone; and 4) cassiterite U-Pb dating yielded 86.5 ± 1.6 Ma, coeval with sulfide mineralization.
Native gold enrichment process during growth of chalcopyrite-lined conduits within a modern hydrothermal chimney (Manus Basin, PNG)
https://doi.org/10.2138/am-2021-7866Seafloor hydrothermal chimneys from back-arc basins are important hosts for metals, such as Cu, Zn, Pb, Ag, and Au, and detailed knowledge of mineralogy and chimney growth history will help understand the spatial distribution and enrichment mechanism of precious metals. To unravel the mechanism of native gold precipitation during the growth of multiple chalcopyrite-lined conduits as part of a modern chalcopyrite-sphalerite chimney, Hu et al. conducted a submicrometer- to a millimeter-scale investigation of the mineralogy and microstructures with a combination of SEM-EBSD and Synchrotron XRF. Gold occurrence in chalcopyrite-lined conduit walls is relatively rare and often associated with tennantite or Bi-tellurite. This study showed that gold is closely associated with various sulfides, including chalcopyrite, sphalerite, tennantite, and pyrite, and may precipitate during different mixing processes between hydrothermal fluids and surrounding fluids. The observed board spectrum of gold-sulfide associations in chalcopyrite-lined chimney walls provides important implications for searching for native gold in more modern seafloor hydrothermal chimneys, as well as in ancient volcanogenic massive sulfide deposits.
Pliniusite, Ca5(VO4)3F, a new apatite-group mineral and the novel natural ternary solid-solution system pliniusite-svabite-fluorapatite
https://doi.org/10.2138/am-2022-8100Pekov et al. report the discovery of a new apatite-group mineral, pliniusite, ideally Ca5(VO4)3F, the second vanadate mineral of the apatite supergroup, in fumarole exhalations at the Tolbachik volcano (Kamchatka, Russia) and in a pyrometamorphic rock of the Hatrurim Complex (Israel). Pliniusite, fluorapatite Ca5(PO4)3F, and svabite Ca5(AsO4)3F form an almost continuous ternary solid-solution system with wide variations of T5+ = P, As, and V. The combination of high temperature, low pressure, and high oxidizing potential is favorable for the incorporation of V5+ into calcium apatite-type compounds and in general for the formation of fluorovanadates.
Heamanite-(Ce), (K0.5Ce0.5)TiO3, a new perovskite supergroup mineral found in diamond from Gahcho Kue, Canada
https://doi.org/10.2138/am-2022-8908Anzolini et al. report the discovery and description of a new perovskite-group mineral, heamanite-(Ce), ideally (K0.5Ce0.5)TiO3, found as an inclusion in a diamond from the Gahcho Kue mine in the Northwest Territories, Canada. Heamanite-(Ce) is the K-analog of loparite-(Ce), ideally (NaCe)Ti2O6. This finding not only represents the sixth perovskite-structured mineral to occur in Earth's mantle, along with perovskite sensu stricto, K-REE- Cr-rich tausonite, bridgmanite, CaSiO3-perovskite, and goldschmidtite, but also indicates that perovskite-structured oxides have the potential to be significant hosts for K and LREE in the mantle. To precipitate a phase with such high concentrations of incompatible elements, we suggest that extreme levels of fractionation occurred in the fractionating metasomatic fluid. Moreover, the high concentration of radiogenic elements in heamanite-(Ce) may allow for future isotopic dating, making this discovery of interest to geochemists, kimberlite petrologists, and mineralogists.
A revised analysis of ferrihydrite at liquid helium temperature using Mossbauer spectroscopy
https://doi.org/10.2138/am-2021-7802Ferrihydrite is ubiquitous in the environment and is a major component of the bioavailable global iron pool. A powerful analytical approach for studying ferrihydrite is 57Fe Moessbauer spectroscopy. However, despite the first spectrum being published several decades ago, there remain conflicting and contradicting reports in the literature on the best way to fit it. Byrne and Kappler have rigorously fitted 2-line and 6-line ferrihydrite using a range of models and multiple sites to determine the most statistically relevant approach to interpreting this enigmatic mineral.
First find of merrillite Ca3(PO4)2 in a terrestrial environment as an inclusion in lower-mantle diamond
https://doi.org/10.2138/am-2022-8175Kaminsky and Zedgenizov found merrillite as inclusions in lower-mantle diamonds from the Juina area in Brazil. Previously this rare mineral was known only in meteorites and Lunar rocks. This finding may be of importance because merrillite and tuite are considered members of the deep Earth minerals and potential hosts for rare earth elements and large ion lithophile elements.
Book Review: Deep Carbon: Past to Present
https://doi.org/10.2138/am-2022-B107814New Mineral Names: Hydrous Minerals
https://doi.org/10.2138/am-2022-NMN107813July 2022
Mineral Evolution Heralds a New Era for Mineralogy
https://doi.org/10.2138/am-2022-8414This Highlights and Breakthrough by Anhuai Lu is dedicated to the review article by Robert M. Hazen "On the paragenetic modes of minerals: A mineral evolution perspective" in this issue of American Mineralogist. As IMA President, Lu also wishes to use this contribution to congratulate Robert M. Hazen, who was honored with the IMA Medal for Excellence in Mineralogical Research 2020, for his outstanding achievements in mineral crystal chemistry, particularly in the field of mineral evolution.
Pauling's Rules for Oxide-Based Minerals — A Re-examination Based on Quantum Mechanical Constraints and Modern Applications of Bond-Valence Theory to Earth Materials
https://doi.org/10.2138/am-2021-7938Recent criticisms of Pauling's Rules of crystal chemistry based on a statistical analysis of their applicability to some 5000 crystal structures have raised serious questions about their usefulness. Gibbs et al. have addressed these questions and concerns, showing that there is a firm quantum mechanical basis for these rules and that modification of the second rule to account for variation in bond strength with bond length has led to development of bond valence theory (BVT). They conclude by showing some of the many applications of BVT to Earth materials.
A cotunnite-type New High-pressure Phase of Fe2S
https://doi.org/10.2138/am-2022-7959Sulfur is frequently observed in iron meteorites and could be a common impurity element in planetary iron cores in our solar system and beyond. Therefore, the Fe-FeS phase diagram is of great importance to understand metallic cores of terrestrial planets. Here Oka et al. examined the pressure-induced phase transitions in Fe2S and discovered a new phase with the cotunnite-type structure that is stable between 30 and 130 GPa. It has been argued that sulfur is an important light element in the Mars' core, and they discuss the possible presence of cotunnite-type Fe2S there.
Density Determination of Liquid Iron-Nickel-Sulfur at High Pressure
https://doi.org/10.2138/am-2021-7924The nature and the amount of light elements in the Earth's outer core has been one of the biggest enigmas of Earth sciences for more than 60 years. The comparison of seismic observations with sound velocity and density data measured in high pressure experiments can help reveal the lighter components in the Earth's outer core. Kawaguchi et al. determined the density of liquid (Fe,Ni)75S25 up to 74 GPa at 3400 K from pair distribution functions obtained by synchrotron X-ray diffraction measurements using diamond anvil cells. They obtained bulk modulus and its pressure derivative of liquid (Fe,Ni)75S25 using the third-order Vinet equation of state. The atomic volume change of Fe47Ni28S25 by melting was found to be approximately 10%, larger than that of pure Fe (~3%). They assumed that this large volume change was due to large anisotropy in the direction of the thermal vibrations of the Fe3S structure, based on the analysis of powder X-ray diffraction measurements at ambient pressure. Finally, assuming ideal mixing with pure Fe and S as the only light element in the core, the density profile of the outer core is best explained using 5.3-6.6 wt% S, an estimation compatible with sound velocity measurements, w.
On the Paragenetic Modes of Minerals: A Mineral EvolutionPerspective
https://doi.org/10.2138/am-2022-8099A systematic survey of the paragenetic modes of 5659 mineral species by Hazen and Morrison reveals patterns in the diversity and distribution of minerals related to their evolving formational environments. The earliest minerals in stellar, nebular, asteroid, and primitive Earth contexts were dominated by relatively abundant chemical elements, notably H, C, O, Mg, Al, Si, S, Ca, Ti, Cr, and Fe. Significant mineral diversification subsequently occurred via two main processes, first through gradual selection and concentration of rarer elements by fluid-rock interactions, and then through near-surface biologically-mediated oxidation and weathering. Most minerals form in only one way, though a few common minerals form by a dozen or more processes. A third of known mineral species form exclusively as a consequence of biological processes. By far the most significant factor in enhancing Earth's mineral diversity has been its dynamic hydrological cycle. At least 4583 minerals — 81 % of all species — arise through water-rock interactions.
Lumping and Splitting: Toward a Classification of Mineral Natural Kinds
https://doi.org/10.2138/am-2022-8105How does one best subdivide nature into kinds? All classification systems require rules for lumping similar objects into the same category, while splitting differing objects into separate categories. In the evolutionary system mineralogy Hazen et al. lump two minerals only if they have related composition, structure, and formation environment. They split two minerals if they form in two or more distinct paragenetic environments. Based on these lumping and splitting criteria, they estimate that Earth holds more than 10,000 mineral natural kinds.
Thermal Expansion of Minerals in the Amphibole Supergroup
https://doi.org/10.2138/am-2022-7988This paper by Tribaudino et al. attempts to understand thermal expansion in the amphibole group of minerals. Such work will lead to a greater ability to predict phase equilibria that involve amphiboles at high temperatures, in this case those applicable to both metamorphic and igneous conditions. As a follow-on to their recent thermal expansion work on the pyroxene system, the data also allow further testing of various thermal expansion models.
A Multi-faceted Experimental Study on the Dynamic Behavior of MgSiO3 Glass in the Earth's Deep Interior
https://doi.org/10.2138/am-2022-7969Ryu et al. take a unique, multifaceted, experimental approach by studying MgSiO3 glass using three techniques at high pressure: Raman spectroscopy, X-ray scattering, and Brillouin spectroscopy. Their results present a complete and consistent picture of the structural modification of MgSiO3 glass under high pressure. A structural evolution model is proposed, explaining all the details of structural modification in MgSiO3 glass with pressure.
Origin of Beta-cristobalite in Libyan Desert Glass -- the Hottest Naturally Occurring Silica Polymorph?
https://doi.org/10.2138/am-2021-7922Cavosie et al. report a geochemical and microstructural study of cristobalite, a silica polymorph in Libyan Desert Glass (LDG). LDG is a nearly pure silica glass found in Egypt and is thought to have formed during a meteorite impact. Our data reveal formation of oscillatory zoned beta-cristobalite at >1350 °C; during cooling it reverted to alpha-cristobalite. They propose that cristobalite in LDG represents the hottest naturally occurring silica polymorph thus far described, reflecting its unique formation during a meteorite impact.
Time-resolved Raman and Luminescence Spectroscopy of Synthetic REE-doped Hydroxylapatites and Natural Apatites
https://doi.org/10.2138/am-2022-8006Fau et al. use time-resolved Raman and luminescence spectroscopies to characterize synthetic REE-doped hydroxylapatites and natural apatites. Using a unique experimental setup that allowswork at the microscale, they show how time-resolved spectroscopy can separate the Raman and luminescence signals for such samples. Such techniques is useful to study both signals without contribution/perturbation from the other signal. They discuss the possibility of semi-quantifying the REE concentration from luminescence spectra in such samples. Such quantification may be possible in samples doped with one single REE but is challenging in samples doped with several luminescence activators because of energy transfers between the various activators.
Reexamination of the Structure of Opal-A: A combined Study of Synchrotron X-ray Diffraction and Pair Distribution Function Analysis
https://doi.org/10.2138/am-2022-8017Opals (opal-A and opal-CT) are very common in young sedimentary rocks. New information about opal structures will help us better understand and quantity the transformation from opal-A (diatomaceous clay) to opal-CT during the diagenesis . The currently accepted opal structure is composed of disordered intergrowths of cristobalite- and tridymite-like domains consisting of six-membered rings of [SiO4] tetrahedra, but ILee et al. have identified additional, coesite-like domains comprising four-membered rings of [SiO4] tetrahedra. More broadly, this study has also demonstrated that the combined approach of synchrotron XRD/PDF is a powerful tool to determine the structures of poorly crystallized minerals. The combined method will provide the detailed crystallographic information of the phase transformation of natural opal.
A First-Principles Study of Water in Wadsleyite and Ringwoodite: Implication for the 520 Km Discontinuity
https://doi.org/10.2138/am-2021-7929Wang and Wu demonstrate that water is preferentially incorporated into wadsleyite relative to the coexisting ringwoodite and the presence of water enhances the formation of the 520-km discontinuity by influencing the two-phase coexistence domain.
Inclusions in Calcite Phantom Crystals Suggest Role of Clay Minerals in Dolomite Formation
https://doi.org/10.2138/am-2020-7483Farsang et al. characterized the micro- and nano-inclusions embedded in calcite phantom crystals from Gemerska Ves, Slovakia, by a combination of Raman spectroscopy, scanning and transmission electron microscopy, X-ray powder diffraction, and C and O isotope analysis. The inner, terracotta-colored part of the crystals contains abundant cavities, dolomite, hematite, goethite, titanite, phyllosilicate, and apatite inclusions and nanostructures that have grown on the walls of cavities: hematite and goethite particles sandwiched between either two phyllosilicate crystals or a phyllosilicate and a carbonate (calcite or dolomite) crystal. Their observations suggest that a smectite-illite conversion provides the Mg necessary for the precipitation of dolomite.
Reinvestigation of Probertite, CaNa[B5O7(OH)4]x3H2O, a Mineral Commodity of Boron
https://doi.org/10.2138/am-2022-8086Probertite, CaNa[B5O7(OH)4]x3H2O, is a mineral commodity of B (with B2O3~50 wt%). Its crystal chemistry was reinvestigated by Gatta et al. with a multi-methodological approach (X-ray and neutron diffraction, EPMA-WDS, LA-ICP-MS). Excluding B, probertite does not act as geochemical trap of other industrially-relevant elements (e.g., Li, Be, or REE). The H-bonding scheme in the structure of probertite is complex and pervasive, so that the H-bonding network presumably has an important role on the structural stability. The potential utilization of probertite is discussed.
Crystal Structure Determination of Orthorhombic Variscite2O and Its Derivative AlPO4 Structure at High Temperature
https://doi.org/10.2138/am-2022-8119Variscite and metavariscite are uncommon secondary minerals, relevant in environmental and in technological applications. Although two orthorhombic modifications are known (Lucin- and Messbach-type), the fine-grained nature of the Messbach-type variscite has hampered determination of its crystal structure. Ardit et al. used XRD and NMR analyses to solve this conundrum. Structural interpretations and topological analysis demonstrate that the two orthorhombic modifications are polytypes. High-temperature XRD, NMR, and thermal analyses clarified the nature of the transformation to a new derivative AlPO4 structure.
Transformation of Fe-bearing Minerals from Dongsheng Sandstone-type Uranium Deposit, Ordos Basin, north-central China: Implications for Ore Genesis
https://doi.org/10.2138/am-2021-7888Yue et al. observe six main Fe-bearing minerals including biotite, Fe-Ti oxides, pyrite, hematite, goethite, and chlorite in four different colored sandstones. Mobilization and fixation of U are associated with alteration processes of Fe-bearing minerals. Paragenetic relationships and sulfur isotopic compositions of ore-stage pyrite suggest two distinct mechanisms for U mineralization.
Vaterite in a Decrepitated Diamond-bearing Inclusion in Zircon from a Stromatic Migmatite in the Chinese Sulu Ultrahigh-pressure Metamorphic Belt
https://doi.org/10.2138/am-2021-7940Vaterite occurs in a diamond-bearing, decrepitated inclusion of zircon from a stromatic migmatite in the Chinese Sulu UHP metamorphic terrain. Indigenous diamonds and amorphous C-Si-O and Zr-Si-O materials are associated with vaterite in this paper by Fei and Liu. The Raman spectra of highly disordered mcarbonaceous material in the amorphous materials indicate that it originated from organic matter. Inclusion decrepitation resulted in the precipitation of the amorphous materials and the formation and preservation of vaterite.
Oxygen Diffusion in Garnet: Experimental Calibration and Implications for Timescales of Metamorphic Processes and Retention of Primary O Isotopic Signatures
https://doi.org/10.2138/am-2022-7970Scicchitano et al. have uploaded a Graphical Abstract, to see online (http://www.minsocam.org/MSA/Ammin/AM_NotableArticles.html).
Oxidation State of Iron and Fe-Mg Partitioning between Olivine and Basaltic Martian Melts
https://doi.org/10.2138/am-2021-7682Olivine dominates the upper mantle, is often the first phase to crystallize from a mantle melt, and has a relatively simple chemistry. As a result, it is critical for understanding and modeling igneous processe. Fe-Mg partitioning between olivine and melt can be used as a criterion of equilibrium in experiments and for the estimation of parental liquid compositions, which allows the calculation of mantle potential temperature, as well as putative mantle compositions capable of producing observed magmas. An important consideration is whether or not the canonical exchange partition coefficient, (Fe2+O/MgO)olivine/(Fe2+O/MgO)liquid or KD, value of 0.30 is appropriate for Martian applications as Martian melts have significantly more Fe and lower Al than their terrestrial counterparts. Calculating an accurate, KD, however, is difficult due to the fact that the silicate liquid has non-negligible amounts of both Fe2+O and Fe3+O1.5. To better constrain Fe3+/Fe2+ in magmatic liquids, and the corresponding KD of martian basalts, Matzen et al. conducted a series of experiments at controlled temperature and oxygen fugacity on a synthetic analog of a proposed primary Martian basalt. After quenching, Fe3+/Fe2+ ratios of the resultant glasses were determined by Mossbauer spectroscopy. They combined their data with experiments from the literature to determine the KD appropriate for Martian magmas. They used their KD to determine which, if any, of the olivine-bearing shergottites potentially represent liquids. For each meteorite, they assumed a liquid composition equal to that of the bulk, and then compared that liquid to the most Mg-rich olivine reported. Applying their KD, leads to the possibility that bulk Yamato 980459, NWA 5789, NWA 2990, Tissint, and EETA 79001 (lithology A) represent liquids.
June 2022
Periodic and non-periodic stacking in molybdenite (MoS2) revealed by STEM
https://doi.org/10.2138/am-2022-8019Yang et al. used HAADF-STEM imaging to identify the stacking faults and domains of long-period molybdenite polytypes. Several layers of disordered domains intergrown with ordered 2H1 domain were recognized based on the contrast in HAADF image with one-dimensional lattice fringes. In addition, 30-layer long-period polytype and 2H3 polytype with three repetition domains were discovered for the first time. These results demonstrate that the presence of trace elements is strongly correlated with the existence of disordered domains in molybdenite crystals. It is suggested that non-equilibrium conditions related to compositional fluctuation of fluid during crystallization resulted in the oscillation of 2H1 and 3R polytypes and intergrowth of various disordered domains. The HAADF-STEM imaging method may be applied for studying other disordered layered crystals and twin minerals.
The effect of halogens (F, Cl) on the near-liquidus crystallinity of a hydrous trachyte melt
https://doi.org/10.2138/am-2022-8120Feisel et al. employed the diffusion couple technique to investigate the effect of F and Cl on the crystallinity of a hydrous trachyte. In essence, their experiments demonstrate a significant drop of the liquidus temperature — as manifested by a Cpx-dissolution front — in response to rising Cl and F concentration. The liquidus depression was quantified and shown to linearly correlate with the concentration of F and Cl in the melt. They propose that F and Cl are causing the observed dissolution as they complex with the main components of Cpx. With this study, the authors illustrate the strong influence that halogens can exert on the phase equilibria of a melt and that even small fluctuations of their concentrations, e.g., caused by volcanic degassing, can control the stability of Fe-Mg-Ca mineral phases.
Occurrence of tuite and ahrensite in Zagami and their significance for shock-histories recorded in martian meteorites
https://doi.org/10.2138/am-2022-8020The Zagami martian meteorite, one of the most famous martian falls, was highly shocked, as supported by the identification of many high-pressure polymorphs, e.g., stishovite, seifertite, liebermannite, and zagamiite. These phases are crucial for constraining how they were formed by asteroid impact on Mars. Gu et al. report on the discovery of two high-pressure minerals, tuite and ahrensite, identified in shock-induced melt pockets in Zagami meteorite, which could shed light on the phase decomposition under high P-T conditions. Tuite identified in this study has two formation pathways: decomposition of apatite and transformation of merrillite under high P-T conditions. Nanocrystalline ahrensite in the pyroxene clast from Zagami is likely the product of decomposition of pigeonite under high P-T conditions through a solid-state transformation mechanism. This dissociation of pigeonite to ahrensite/ringwoodite plus other phases may be widespread in other heavily shocked meteorites but potentially been overlooked in previous studies.
Zolenskyite, FeCr2S4, a new sulfide mineral from the Indarch meteorite
https://doi.org/10.2138/am-2022-8094Ma and Rubin report a new sulfide mineral, zolenskyite (FeCr2S4), discovered in the Indarch meteorite, an EH4 enstatite chondrite, and discuss its origin and significance. Zolenskyite is a monoclinic polymorph of daubréelite that may have crystallized within chondrules at high temperatures from the early solar system.
A refined estimation of Li in mica by a machine learning method
https://doi.org/10.2138/am-2022-8029Wang et al. use a compilation of >2000 mica compositions from various rock types, including Li2O and the following 10 major components: SiO2, Al2O3, TiO2, FeOT, MgO, MnO, CaO, Na2O, K2O, and F. These data are regressed using a multivariate polynomial regression (MPR) based machine learning (ML) method to refine the equation for estimating Li in micas over a wide range of chemical compositions. They then discuss the uncertainties and limitations of the refined equation and compare the performance of the MPR model with previously published empirical equations. Finally, they apply the refined equation to estimate the Li2O contents of micas from various types of rocks in central Inner Mongolia, NE China. Their results indicate that the MPR model is more precise and accurate and can be generalized to almost all types of micas. Above all, they provide a robust and convenient approach to conduct microanalysis of Li in mica, applying a combination of EMPA and machine learning methods, which can be widely used in mineralogy, petrology, and economic geology.
Olivine in picrites from Continental Flood Basalt provinces classified using machine learning
https://doi.org/10.2138/am-2022-8083Cheng et al. provide a novel method using machine learning based on a large database of olivine composition in picrites from global Continental Flood Basalts to classify olivine with two distinct sources. This method is capable of classifying the overlapping data into correct groups, which cannot be done by traditional methods in several minutes with high accuracy.
Non-Arrhenian viscosity-temperature relationships for carbonate melts
https://doi.org/10.2138/am-2021-7752Dingwell et al. report the first quantitative determination of a glass transition for a carbonate liquid. Specifically, they demonstrate the nature of the glass transition and viscosity of a mixed K2CO3-MgCO3 carbonate melt by performing scanning calorimetry and viscometry on samples of a supercooled carbonate phase. Using this low-temperature viscosity data combined with published higher temperature (high pressure) data as well as both classical and ab initio molecular dynamics simulation estimates, they determine a non-linear Vogel- Fulcher-Tammann (VFT) law fit and quantify the extent of fragility. The non-linear rate change in viscosity is an important parameter in understanding the nature of carbonatite lava flows on Earth and possibly Venus. The most widespread occurrence of carbonate melts is most likely in the deep mantles of terrestrial planets. At very small degrees of melting, alkali-rich carbonate melt may exist at lower temperatures (>1200 °C). These low temperatures are realistic for the shallow lithosphere beneath the old continental crust and are in the temperature range where a linear fit based on the high-temperature data will become inaccurate. This would have implications for modeling the transport properties of these melts to their surface expression or as metasomatizing agents.
Etching of fission tracks in monazite: Further evidence from optical and focused ion beam scanning electron microscopy
https://doi.org/10.2138/am-2022-8002Jones et al. conducted a series of experiments on monazites from Victoria, Australia, to further understand their fission-track (FT) etching properties. Monazite is a mineral currently being developed in the FT system, and a crucial first step in its development is understanding its etching properties. This enables fission tracks to be observable under the optical microscope so that they can be analyzed and used to constrain the thermal history of rocks in the Earth’s uppermost crust (1–2 km).
The low-temperature shift of antigorite dehydration in the presence of sodium chloride: in situ diffraction study up to 3 GPa and 700 °C
https://doi.org/10.2138/am-2022-7962The dehydration of serpentine mineral antigorite is regarded as the key step in metamorphic transformation of serpentinized rocks in subduction zones, which affects seismicity and feeds volcanic activity. Likhacheva et al. found that, in the presence of salt-bearing fluid, the reaction shifts to lower temperature by more than 150 °C in the pressure range of 2-4 GPa, which implies a broadened P-T region of serpentinite dehydration in subducting slab.
Chemistry-dependent Raman spectral features of glauconite and nontronite: Implications for mineral identification and provenance analysis
https://doi.org/10.2138/am-2022-8044Baumgartner et al. provide a comprehensive Raman spectral characterization of nontronite and glauconite-nontronite mixed-layer phases from seafloor hydrothermal fields. It is shown that these phyllosilicates exhibit a series of Raman peaks that are strongly dependent on the exchange between trivalent and divalent cations in the trioctahedral sheets. The importance of these findings for a variety of applications is discussed.
Experimental determination of solubility constants of saponite at elevated temperatures in high ionic strength solutions
https://doi.org/10.2138/am-2021-7827Xiong performed solubility experiments of Mg-saponite at 80 ºC to quantify the thermodynamic stability of this tri-octahedral smectite in the presence of nitrate cancrinite. In combination with the equilibrium constant at 80 ºC for the dissolution reaction of nitrate cancrinite from the literature, the author determined the solubility constant of saponite at 80 ºC to be –69.24 ± 2.08. Furthermore, the author extrapolated the equilibrium constant at 80 ºC to other temperatures (50-100 ºC) using the one-term isocoulombic method. The obtained equilibrium constants are expected to find applications in several fields, especially geological disposal of nuclear waste.
Hydrothermal troctolite alteration at 300 and 400 ºC: Insights from flexible Au-reaction cell batch experimental investigations
https://doi.org/10.2138/am-2021-7832Hansen et al. present a comprehensive experimental study on the serpentinization of olivine-rich troctolites. Dickson-type reactors were used to simulate hydrothermal troctolite alteration by reacting fresh forsteritic olivine and anorthite-rich plagioclase with seawater-like fluids at two distinct temperature levels (300, 400 °C) and an elevated pressure level (40 MPa). Time series analyses of reaction concomitant fluid samples and detailed mineralogical and geochemical characterization of the final solid-phase assemblage were combined with comparative thermodynamic modeling to enable a comprehensive characterization of the alteration process. Presented results confirm earlier hypothesis that silica activity levels set by temperature-dependent coupled olivine serpentinization and plagioclase desilication reactions constitute a key control. They further demonstrate the crucial effect of variable reaction rates in different portions of a solid reactant that are closer to or further away from the solid-fluid interface.
Timescales and rates of intrusive and metamorphic processes determined from zircon and garnet in migmatitic granulite, Fiordland, New Zealand
https://doi.org/10.2138/am-2022-7967Stowell et al. investigated the timing of pluton emplacement and metamorphism in the lower crust of the Fiordland magmatic arc using zircon and garnet geochronology. The isotope data provide precise dates which clearly indicate that high-temperature granulite facies metamorphism occurred less than 3 million years after intrusion of voluminous mantle-derived magmas. No heat source has been documented for the metamorphism; however, they speculate that underplating of magma caused heating. Subsequent cooling for this granulite orthogneiss at 15-25 °C/million years was considerably slower than previously estimated. The new isotope data provide precise ages and rates of cooling for a single outcrop and underline the importance of multiple chronometers and careful textural characterization for assigning meaningful ages to lower-crustal rocks.
In situ chemical and isotopic analyses and element mapping of multiple-generation pyrite: evidence of episodic gold mobilization and deposition for the Qiucun epithermal gold deposit in Southeast China
https://doi.org/10.2138/am-2022-8030Ma et al. investigated the origin of high-grade ore shoots and visible gold in gold deposits by analyzing the multiple generations of pyrite in a representative epithermal gold system in SE China using a number of in situ analytical techniques (including LA-ICP-MS trace element analysis and mapping and LA-MC-ICP-MS sulfur isotope analysis). The results demonstrated that multiple remobilization and reprecipitation of gold were taken place in this epithermal gold deposit. The pressure-driven hydrothermal processes play an important role not only in initial invisible (refractory) gold enrichment but also in the re-enrichment of visible gold and some other trace metals during episodic deposition, replacement, and hydrothermal alteration of auriferous pyrite in gold deposits. This study demonstrates that detailed textural examination and in situ chemical and sulfur isotope analysis under robust geological and petrographic frameworks can effectively constrain the complex gold mobilization and deposition processes in hydrothermal ore deposits.
Hydrothermal mineralization of celadonite: Hybridized fluid-basalt interaction in Janggi, Korea
https://doi.org/10.2138/am-2022-8045Park et al. discovered an exceptional celadonite mineralization in the Miocene lacustrine Janggi basin in the southeastern Korean Peninsula. This celadonite occurs as greenish earthy/vitreous materials filling the E-W trending fault zones in basaltic flows. Detailed fieldwork and whole-rock major, trace, and oxygen isotope analyses indicate that celadonites were formed in an open system of ~120 °C via the interaction of the hybridized fluid and basalts along with the physicochemical fault brecciation of the host rock. The cations required for the celadonite formation were supplied by the smectitization/zeolitization of rhyolitic mesostasis (for Al and part of K) and pyroxene microlites (for Fe and Mg) in the basaltic breccias during the associated oxidation of micro-nanoparticles by circulating fluids (for most of K). This mechanism is plausible, where both the magmatic fluid influx and the particle size reduction could be involved. This model suggests that any crustal rocks rich in ferromagnesian components could produce celadonite through the hydrothermal potassic alteration.
Gungerite, TlAs5Sb4S13, a new thallium sulfosalt with a complex structure containing covalent As-As bonds
https://doi.org/10.2138/am-2022-8003Kasatkin et al. describe a new thallium sulfosalt mineral, gungerite, discovered in Russia. It possesses an interesting structural architecture, quite unique among the known sulfosalt structures: the structure consists of doughnut-shaped (As,Sb)–S clusters, which have van der Waals contacts to most of the surroundings and are connected to them only by sparse cation–sulfur bonds.
Nitscheite, a new mineral with an unusual uranyl-sulfate sheet
https://doi.org/10.2138/am-2022-7994Inactive uranium mines have proven to be fruitful underground “natural laboratories”, providing insight into the factors driving the crystallization of uranium minerals. The great diversity observed for uranyl sulfate minerals stems primarily from the large number of stable linkages of uranyl pentagonal bipyramids and sulfate tetrahedra. The new uranyl sulfate nitscheite, described herein by Kampf et al., possesses a type of uranyl sulfate sheet not previously observed in nature.
Protocaseyite, a new decavanadate mineral containing a &91;Al4(OH)6(H2O)12&93;6+ linear tetramer, a novel isopolycation
https://doi.org/10.2138/am-2022-8059Kampf et al. describe the new mineral protocaseyite found in a low-temperature, post-mining, secondary mineral assemblage in the Burro mine in the Uravan Mineral Belt of western Colorado, U.S.A. The crystal structure of this mineral contains a decavanadate polyoxometalate anion together with a novel 4-member aluminate polyoxometalate cation referred to as an Al-flatimer. Polyoxometalate ions have been the subject of numerous synthesis studies in recent years, largely because of their potential technological uses. Polyoxometalate ions have also been proposed to exist in solution as precursors of more extended structural components (chains, sheets, and frameworks) in mineral structures, but only rarely are polyoxometalate ions found as isolated units in minerals.
Fission-track etching in apatite: A model and some practical implications
https://doi.org/10.2138/am-2022-8055Jonckheere et al. investigated the geometries of etched fission tracks in apatite based on a classic dissolution model. The results clarify long-standing misconceptions about estimates of apatite and track etch-rates inferred from the lengths and the cross-sections of etched tracks. It calculates surface track profiles and interprets the contours of confined tracks. The insight gained suggests some steps toward unifying etching protocols that might lead to greater agreement between track-based thermal histories.
Hydrothermal monazite trumps rutile: Applying U-Pb geochronology to evaluate complex mineralization ages of the Katbasu Au-Cu deposit, Western Tianshan, Northwest China
https://doi.org/10.2138/am-2022-8080The Tianshan orogenic belt hosts several world-class gold deposits and is one of the largest gold provinces on Earth. Zheng et al. report detailed mineralogical studies, combined with zircon U-Pb, in situ hydrothermal monazite as well as rutile U-Pb ages, to constrain the timing of Cu mineralization and its possible link to the Au mineralization. Mineralogical studies show that chalcopyrite is the main Cu-bearing mineral in the Cu-Au ores, and it is closely associated with some native gold, monazite, and rutile. SIMS U-Pb dating of zircon from the ore-hosting granite and mafic enclave yielded concordant ages of 354.1 and 355.8 Ma, respectively. The diorite that intruded granite has a zircon U-Pb age of 352.0 Ma. The trace element compositions of the monazite suggest they were formed by hydrothermal fluids rather than inherited from the ore-hosting granite. Hydrothermal monazite coexisting with chalcopyrite and native gold has a concordant age of 348.7 Ma, and the W-rich hydrothermal rutile associated with the chalcopyrite has a U-Pb age of 345 Ma, indicating an early Cu-Au mineralization event prior to the major Au mineralization. The formation time of early Cu-Au mineralization is consistent with the emplacement age of diorite and may be of magmatic-hydrothermal origin, whereas the main Au has no genetic associations with magmatic rocks in the ore district and may belong to the orogenic type.
May 2022
Interplay between fluid circulation and Alpine metamorphism in the Monte Rosa whiteschist from white mica and quartz in situ oxygen isotope analysis by SIMS
https://doi.org/10.2138/am-2020-7523Luisier et al. present a new set of white mica reference materials for in situ 18O/16O measurements by SIMS. Oxygen isotope compositions were measured in metagranite and whiteschists from the Monte Rosa nappe in the Western Alps. Several generations of white mica are characterized based on oxygen isotope composition, chemistry, and microtextural observation. Oxygen isotopes allow us to identify two fluid infiltration events: the first leading to pervasive alteration and a second late local fluid infiltration.
Halogen (F, Cl, Br, I) contents in silt and clay fractions of a Cambisol from a temperate forest
https://doi.org/10.2138/am-2021-7600Epp et al. investigated the proportions of F, Cl, Br, and I that are surface sorbed or incorporated in the structure of clay minerals and pedogenic oxides for different soil size fractions. Halogens can occur as radionuclides 36Cl and 129I with a long half-life or as organic and inorganic pollutants. A detailed understanding of halogen sorption in soil is crucial for potential pollutant retention processes in landfills or radioactive waste disposal to prevent harmful impacts on humans and nature.
Effects of arsenic on the distribution and mode of occurrence of gold during fluid-pyrite interaction: a case study of pyrite from the Qiucun gold deposit, China
https://doi.org/10.2138/am-2021-7675Zhang et al. focus on the distribution and modes of occurrence of gold and their link with arsenic and microstructures within arsenian pyrites from the epithermal Qiucun gold deposit (China). They found that arsenic-induced lattice defects are of significance for the incorporation of invisible gold as a homogeneous solid solution in pyrite lattice. Most visible auriferous inclusions are hosted at the fluid-pyrite reaction interface or fractures and grain boundaries within the As-deficient interiors of pyrite. A conceptual model is proposed to describe Au scavenging from the hydrothermal fluids and to illustrate the genetic relationship between visible- and invisible gold. These findings highlight the significance of systematic composition and textural observations in interpreting fluid-rock interactions; advance our understanding of the mechanisms of trace element incorporation in minerals, and provide guidance on the determination of mineral paragenetic sequences, further enabling us to understand the gold mineralization process.
Resolving sub-micrometer-scale zonation of trace elements in quartz using TOF-SIMS
https://doi.org/10.2138/am-2021-7896North et al. characterized quartz and silica at higher spatial resolution than previously possible by the novel application of time-of-flight secondary ion mass spectrometry (TOF-SIMS). Sub-micrometer resolution (65 nm) maps reveal complex trace element distributions (including the light element lithium) in silica minerals that could not be observed with lower resolution techniques. The use of TOF-SIMS could be advantageous in the Earth sciences for characterizing a range of other minerals. (See graphical abstract)
Specific roles of sodium for the formation process of manganese-substituted octacalcium phosphate
https://doi.org/10.2138/am-2021-7901Manganese (Mn) is a good candidate as a substitutional element for bone substitutes because of its regulation process of bone remodeling that could control osteo-cellular activities, but it strongly inhibits octacalcium phosphate (OCP) formation. Sugiura et al. investigated the specific effect of Na-to-Mn substitution in OCP. They demonstrate that Na-induced OCP formation enhances the HPO4-OH layer structure of OCP and can overcome the Mn inhibition effect. Consequently, the Mn-substituted OCP-induced Na (OCP-Mn,Na) shows a 4.7 ° peak in the X-ray diffraction pattern. Sub-peaks at 9.2 ° and 9.7 ° of OCP disappeared, but an extra peak at 9.3 ° was visible. The thermal stability of OCP-Mn,Na is significantly lower than that of conventional OCP because the layer structure of the former decomposes above ~70 °C. The results indicate that ionic conjugation to Mn is a unique phenomenon for Na.
Chukochenite (Li0.5Al0.5)Al2O4, a new lithium oxyspinel mineral from the Xianghualing skarn, Hunan Province, China
https://doi.org/10.2138/am-2021-7932Rao et al. report the occurrence of the new lithium oxyspinel mineral chukochenite. They reveal the structural relationships between the structures of chukochenite and other (Li0.5Al0.5)Al2O4 phases. The discovery of chukochenite will draw attention to the study of phase transitions between polymorphs of (Li0.5Al0.5)Al2O4. The structural features of polymorphs of (Li0.5Al0.5)Al2O4 may help to explain their luminescence and magnetism.
Ab initio study of the structure and relative stability of MgSiO4H2 polymorphs at high pressures and temperatures
https://doi.org/10.2138/am-2021-7937Solomatova et al. used computational methods to study the relative stability of hydrous magnesium silicate polymorphs with the MgSiO4H2 stoichiometry at pressure-temperature conditions relevant to the Earth’s mantle. Their simulations predict a new monoclinic phase that is preferred over all other polymorphs between 0 to 14 GPa at 1800 K, the approximate temperature of the deep mantle. At higher pressures, phase H is preferred over all other MgSiO4H2 polymorphs. They predict that phase H is the energetically most stable structure at pressures between 14 to 300 GPa at 1800 K. In this study, they also identified the positions of hydrogen in a hypothetical magnesium-endmember of phase Egg (“Phase Mg-Egg”), which was described in a previous study. They find that Phase Mg-Egg is never preferred over the other hydrated magnesium silicate phases; however, it is possible that with certain chemical substitutions the relative energies may change. These results have implications for the deep hydrogen cycle in that they identify two novel potential carrier phases for hydrogen in Earth’s deep interior and assess their stability relative to phase H.
Hexagonal magnetite in Algoma-type banded iron formations of the ca. 2.52 Ga Baizhiyan Formation (Wutai Group, North China): Evidence for a green rust precursor?
https://doi.org/10.2138/am-2022-7981Sun et al. studied hexagonal magnetites that occur in the 2.52 Ga Baizhiyan Formation, North China, and concluded that hexagonal magnetites were transformed from green rusts in ferruginous seawater. Green rust has a great impact on Precambrian biogeochemical cycles.
Magmatic volatiles and platinum-group element mineralization in the Stillwater layered intrusion, U.S.A.
https://doi.org/10.2138/am-2022-7982The activity of volatile-rich fluids may underpin the evolution of basaltic magmatic systems and associated precious metal ore formation. The platinum-group metal deposits of the Stillwater layered intrusion (U.S.A.) have long been held up as a classic example of a locality where the mineralization preserves evidence of a volatile control. The new halogen dataset in this paper by Parker et al. supports and extends the understanding of the role of volatiles in concentrating precious metals in fossil basaltic magmatic systems.
Thermal conductivity of single-crystal brucite at high pressures with implications for thermal anomaly in the shallow lower mantle
https://doi.org/10.2138/am-2022-7986Chien et al. reveal the critical roles that brucite, an important water-carrying mineral in subducting slabs, could play in affecting lower-mantle dynamics and seismic structures. They directly measured brucite’s thermal conductivity to lower mantle pressures and found that as brucite decomposes to periclase at ~800 km depth there would be a significant increase in the thermal conductivity by a factor of ~6-19. The large thermal conductivity discontinuity upon brucite’s decomposition to periclase will induce a local high-temperature anomaly within a subducting slab, enhancing the slab’s buoyancy and promoting its stagnation in the shallow lower mantle. The high-temperature anomaly also destabilizes surrounding hydrous minerals, which facilitates the release of water to the ambient mantle to form dehydration melts, resulting in seismic low-velocity zones at the top of the lower mantle.
Synthesis, structure refinement, and single-crystal elasticity of Al-bearing superhydrous phase B
https://doi.org/10.2138/am-2022-7989Dense hydrous magnesium silicates (DHMSs), such as superhydrous phase B (shy-B), with large water contents and wide stability fields are potential H2O reservoirs in the deep Earth. The incorporation of Al in shy-B will expand its phase stability to higher pressures and temperatures compared with Al-free shy-B. Li et al. synthesized shy-B with different Al contents. The crystal structure, substitution mechanisms, and elasticity were determined by various techniques. Their study helps to explain the low velocity zone and high anisotropy region in the subducted slabs located beneathTonga.
Oxygen isotope heterogeneity of olivine crystals in orogenic peridotites from Songshugou, North Qinling Orogen: Petrogenesis and geodynamic implications
https://doi.org/10.2138/am-2022-7990Yu et al.’s study of oxygen isotopes indicates that olivines in the Songshugou peridotites are heterogeneous in oxygen isotopes. They explain the origin of low-δ18O olivine due to seawater infiltration into the peridotites in the oceanic lithosphere mantle. The peridotites reside in the mantle only for a short time (<20 Ma) so that the low-δ18O olivine is preserved. The high-δ18O olivines found by Yu et al. are due to oxygen exchange with δ18O-shifted melts and/or fluids during exhumation at low temperatures.
Ferrous hydroxychlorides hibbingite (γ-Fe2(OH)3Cl) and parahibbingite (β-Fe2(OH)3Cl) as a concealed sink of Cl and H2O in ultrabasic and granitic systems
https://doi.org/10.2138/am-2022-8011Kodera et al. studied ferrous hydroxychlorides and concluded their paper with the description of the new mineral parahibbingite. They show that parahibbingite and hibbingite are likely to be frequently present in hydrothermally altered ultramafic rocks if they were in contact with chloride-rich anoxic brines. However, as these minerals are extremely susceptible to oxidation and hydration, they might have escaped the attention of researchers. Both minerals should be considered as storage sites of Cl and H2O in large volumes of altered basic and ultrabasic rocks; thus, they could play an important, but yet unrecognized, role in the remobilization of sulfidic ores (if these minerals are dissolved by later fluids) and as vehicles for H2O and Cl transport into the lower crust at shallow levels of subduction zones. Hibbingite was found to be a common daughter mineral in high salinity brines and salt melt inclusions worldwide, where it indicates cooling under reducing conditions. In liquid-free, salt melt inclusions, identification of this mineral enables an exact calculation of the amount of water dissolved in these extreme fluids. γ-Fe2(OH)3Cl (parahibbingite) is already known to occur in environments where low-carbon steel interacts with chloride-rich anoxic brines, however, the presence and the importance of the other polymorph β-Fe2(OH)3Cl (hibbingite) was not previously considered. Kodera et al. also show that Raman spectroscopy can be used as a simple and fast tool for quick and non-destructive recognition of both Fe2(OH)3Cl polymorphs in geological and archaeological materials.
Atomic-scale structure and non-stoichiometry of meteoritic hibonite: A transmission electron microscope study
https://doi.org/10.2138/am-2022-8014The mineralogy and petrography of primitive carbonaceous chondrite meteorites provide a window into events that occurred at the birth of this solar system ~4.567 billion years ago. Hibonite (CaAl12O19) is of particular interest because it is a common refractory mineral in Ca-Al-rich inclusions (CAIs) in primitive meteorites and is predicted to be one of the earliest phases to condense from a cooling gas of solar composition. Atomic resolution, high-angle, annular dark-field (HAADF) imaging, and energy-dispersive X-ray (EDX) analyses were used by Han et al. to determine the nature and origin of planar defects in hibonite from the Allende meteorite. Hibonite grains display lamellar intergrowths of common 1.6 nm spacing and less commonly 2.0 nm and 2.5 nm spacing, interspersed in stoichiometric hibonite showing 1.1 nm (002) spacing. Stoichiometric hibonite consists of alternating Ca-containing (“R”) and spinel-structured (“S”) blocks stacked in the sequence RS. In contrast, the 1.6 nm layers result from a doubled S block such that the stacking sequence is RSS, while in the widest defect observed, the stacking sequence is RSSSS. These intergrowths are epitaxial and have coherent, low-strain boundaries with the host hibonite. Atomic resolution EDX maps of hibonite grains in the Allende CAI confirm the preferred site occupancy of Mg on tetragonal M3 sites in S blocks and of Ti on trigonal bipyramidal M2 and octahedral M4 sites in R blocks. Mg is highly concentrated, but Ti is absent, in the planar defects where wider S blocks show Al-rich compositions compared to stoichiometric MgAl2O4 spinel. Therefore, Mg likely played a major role in the formation and metastability of planar defects in hibonite. The occurrence of non-stoichiometric hibonite in the Allende CAI deviates from the mineral formation sequence predicted from equilibrium condensation models. Our atomic resolution TEM observations signify non-equilibrium, kinetically controlled crystal growth during the high-temperature formation of refractory solids in the early solar nebula.
Xuite, Ca3Fe2[(Al,Fe)O3(OH)]3, a new mineral of the garnet group: Implications for the wide occurrence of nano-minerals
https://doi.org/10.2138/am-2022-8023Lee and Guo present the mineral Xuite, Ca3Fe2[(Al,Fe)O3(OH)]3, a new member of the garnet supergroup discovered in basaltic scoria from Menan Volcanic Complex, Idaho, U.S.A. The mineral has been approved by the Commission on New Minerals, Nomenclature and Classification (CNMNC) of the International Mineralogical Association (IMA 2018-135a).
Ground-truthing the pyrite trace element proxy in modern euxinic settings
https://doi.org/10.2138/am-2022-8024Gregory et al. provide the first pyrite trace element data from the Black Sea and additional stratigraphically resolved data from the Cariaco Basin. These data show variability in trace elements from different basins, which has important implications for interpretations made from pyrite trace element chemistry from ancient rocks. Furthermore, analyses from outside euxinic (sulfidic) water column deposition show potential errors that might be made if interpreting pyrite formed in non-euxinic basins.
Impact of fluorine on the thermal stability of phlogopite
https://doi.org/10.2138/am-2022-8051Sun et al. reveal the links between fluorine, the behavior of hydroxyl, and the lattice of phlogopite, which is important to constrain volatile cycling and fluorine’s role in the physical and chemical properties of the upper mantle.
Raman spectroscopy-based screening of zircon for reliable water content and oxygen isotope measurements
https://doi.org/10.2138/am-2022-8075Water content and the source region of melt have a pivotal influence on magmatic processes, such as magma origin, partial melting, and fractionation. Zircon is a ubiquitous, uranium-rich accessory mineral in magmatic rocks, characterized by high physical and chemical stability, and zircon oxygen isotopes are useful tracers of magma sources and processes. Meanwhile, the low diffusivity of water enables the zircon to be the ideal mineral to preserve its water. Study of zircon oxygen isotope and water content yields new insights into magmatic processes. Trace amounts of water that partitions into zircon from the melt during its crystallization are termed primary water, which can be used to infer the water content in the melt. The assignment of primary water is not straightforward because secondary water can enter zircon as OH or H2O molecules after crystallization. Metamictization caused by the accumulation of radiation damage markedly increases the water storage capacity of zircon and was considered the main cause for the absorption of secondary water. Such secondary water would interfere with the accurate analysis of oxygen isotope compositions. Using laser Raman spectroscopy, Yang et al. propose criteria to select the least metamict zircon for reliable primary water content and oxygen isotope composition analysis.
April 2022
Resolving the conundrum of equilibrium solubility of smectites—A Perspective
https://doi.org/10.2138/am-2022-8213Smectites are a group of clay minerals that have been recently discovered to be abundant on Mars. A better understanding of the behavior of this group of minerals in water will help us to understand how they form, and applying these to occurrences on Mars may enable a better understanding of aspects of the early history of the planet. However, some fundamental questions about these minerals have persisted for nearly 50 years. This perspective by Stephen U. Aja reviews the most recent studies that have a bearing on these questions and concludes that much of those questions may have been misguided.
Manjiroite or Hydrous Hollandite?
https://doi.org/10.2138/am-2021-7848This study by Post et al. raises questions about the material originally described as manjiroite by Nambu and Tanida (1967), suggesting that either there is considerable variation of the Na concentration in the type locality samples or an error in the original analyses. Although the low Na concentrations in the samples studied here ruled out a classification as manjiroite sensu stricto, they nevertheless represented a novel hydrous hollandite-like mineral. As such, they provided the opportunity for the first detailed characterization of the structure and dehydration behavior of a natural hollandite-like mineral with molecular water as the predominant tunnel species -- filling approximately three-quarters of the tunnel sites. This work focuses new attention on the role of molecular water in hollandite minerals, especially those associated with low-temperature aqueous systems. Studies of synthetic hydrous hollandites suggest that structural water affects their cation-exchange properties. Additionally, recent studies have shown that birnessite-like phyllomanganates, which are the major Mn oxide phases in soils and sediments, transform into hollandite-like phases and other tunnel structures under ambient environmental conditions. The structural water inherited from the interlayers of these phyllomanganates ends up in the tunnels as H2O and as OH-, and the hydrous components of the hollandite-like phases are important factors in determining their capacities to exchange or accommodate various cations, such as Li+, Na+, Ba2+, Pb2+, and K+, in natural systems.
Petrologic evolution of boninite lavas from the IBM fore-arc, IODP expedition 352: Evidence for open-system processes during early subduction zone magmatism
https://doi.org/10.2138/am-2021-7733Analysis of IODP Expedition 352 boninite lavas, using EPMA and LA-ICP-TOF, reveals anomalous pyroxene overgrowths, complex pyroxene zoning patterns, and oscillatory zoning in olivine. Pyroxene overgrowths likely formed during the rapid cooling and decompression in the eruptive process. Complex pyroxene zoning patterns suggest that crystals experience multiple undercooling and magma mixing events prior to eruption. Mg-Fe oscillatory zoning patterns in olivine support the hypothesis that magma mixing events occur prior to boninite eruptions. The presence of olivine xenocrysts in boninite lavas indicates that melts likely mix within an axial magma chamber. This study by Scholpp et al. highlights the importance of multi-element chemical maps and chemical transects in studies of zoned minerals in igneous systems. These techniques reveal chemical patterns within minerals, which would typically be overlooked, and can provide a better understanding of complex crystallization histories in igneous systems. This study also highlights the scientific benefits of an ostensibly education-focused investigation of a significant suite of rock samples.
Coupled hydrogen and fluorine incorporation in garnet: New constraints from FTIR, ERDA, SIMS, and EPMA
https://doi.org/10.2138/am-2021-7880Considerable progress has been made in the last three decades in understanding how trace amounts of hydrogen can be incorporated in nominally anhydrous minerals and how this H (most commonly incorporated as structurally bound OH- groups) affects geophysical and petrologic processes in the Earth and other planetary bodies. Recent work has also highlighted the potential importance of trace fluorine (substituting for O2-) in nominally anhydrous minerals. This paper by Mosenfelder et al. improves on previous methods for quantifying H and F in garnet and places new constraints on how these elements are incorporated. This work may be used in the future to place better constraints on the activities of H2O and F-species in fluids that were in equilibrium with garnet, as well as constraining the recycling of H and F into the mantle via study of xenoliths.
Incorporation mechanism of structurally bound gold in pyrite: Insights from an integrated chemical and atomic-scale microstructural study
https://doi.org/10.2138/am-2021-7812On the basis of Z-contrast HAADF-STEM imaging, Meng et al. document that Au atoms sit at the Fe atom sites in the lattice of arsenian pyrite. Their results, coupled with previous studies, suggest that Au and As occur dominantly as chemically bound Au+ and As-, respectively. It is thus proposed that both Au+ and As- are incorporated into the structure of pyrite through being bonded at the Fe and S atoms sites, respectively. Their results have significant implications for understanding the process by which Au was enriched from ore-forming fluids with low Au and As concentrations into arsenian pyrite and for exploring the role of As in Au mineralization of hydrothermal deposits.
The electrical conductivity of albite feldspar: Implications for oceanic lower crustal sequences and subduction zones
https://doi.org/10.2138/am-2021-7836Amulele et al. conducted high pressure-temperature (1 GPa & 573–1273 K) electrical conductivity measurements, via impedance spectroscopy, on single-crystal andesine (albite) feldspar from Nuevo Casas Grande, Chihuahua, Mexico. Comparison of the obtained data with previously published results for andesine and associated feldspar reveals the effect of oxygen fugacity on the conduction mechanism. Because of the diverse range of geological settings in which plagioclase feldspars such as labradorite occur, it is important to understand their conduction mechanism at a range of fO2 conditions for consistent and accurate interpretation of magnetotelluric datasets globally. Their measurements indicate that the electrical conductivity of a naturally dry plagioclase feldspar can be high and only drops below 10-4 S/m (the most resistive values for the crust on average) when it is hydrated with as little as 50 ppm wt. H2O. These results would imply a highly dry crust in certain regions of the Earth that may be dominated by plagioclase feldspars.
A high-pressure, clinopyroxene-structured polymorph of albite in highly shocked terrestrial and meteoritic rocks
https://doi.org/10.2138/am-2021-7925Ma et al. report a new vacancy-stabilized, high-pressure, and high-temperature clinopyroxene, with Si on the M1 site and a composition essentially equivalent to that of albitic plagioclase. This clinopyroxene was formed by shock metamorphism in terrestrial impactites and ordinary chondrites. It has not been previously observed or synthesized and, therefore, provides potential new insights into shock conditions and impact processes.
Water in the crystal structure of CaSiO3 perovskite
https://doi.org/10.2138/am-2022-8009Shim et al. conducted first-principles calculations for several hydrogen substitution mechanisms in CaSiO3 perovskite. They found that various hydrogen configurations can coexist in CaSiO3 perovskite at mantle-related pressure conditions. The calculated results also support the experimentally observed hydration of CaSiO3 perovskite. The result suggests that CaSiO3 perovskite may be an important mineral phase to consider for the deep storage of water in the mantle.
Release of chromite nanoparticles and their alteration in the presence of Mn-oxides
https://doi.org/10.2138/am-2021-7881McClenaghan and Schindler demonstrate for the first time that chromite nanoparticles persist through weathering of their host silicates and can be released into the environment. These observations change our understanding of the potential risks of Cr-bearing silicates in mine tailings and soils. The release of chromite nanoparticles, as opposed to Cr3+(aq) species, has a large impact on the fate of Cr in the environment as the behavior of nanoparticles is governed by surface reactions. This study also provides insights into mineralogical processes during the interaction of chromite and Mn-oxides nanoparticles.
The absorption indicatrix as an empirical model to describe anisotropy in X-ray absorption spectra of pyroxenes
https://doi.org/10.2138/am-2021-7950Anisotropic absorption is routinely observed in X-ray absorption spectroscopy. In many spectroscopic methods, absorption anisotropy complicates the relationship between absorption and absorber concentration. Steven et al. assess the efficacy of a triaxially anisotropic absorption indicatrix model to fit oriented X-ray absorption data.
Atomistic mechanism of cadmium incorporation into hydroxyapatite
https://doi.org/10.2138/am-2020-7560Hydroxyapatite has been widely used to remove cadmium (Cd) in contaminated water and soils via Cd-Ca substitution. Liu et al. synthesized and characterized a series of Cd-substituted hydroxyapatites. The obtained results indicate that (1) Cd incorporation causes a decrease in the a- and c-lattice parameters due to a slightly smaller radius of Cd2+ than Ca2+; (2) Cd prefers to occupy the Ca2 site, which is located at the hexagonal positions, and is surrounded by seven oxygen atoms with six from five phosphates and one from hydroxyl; and (3) Cd only occupies one Ca2 site at low Cd contents (~10 mol%), while both Ca1 and Ca2 sites are at higher contents.
Copper isotope evidence for a Cu-rich mantle source of the world-class Jinchuan magmatic Ni-Cu deposit
https://doi.org/10.2138/am-2021-7911A Cu-rich mantle source may play a key role in generating giant magmatic Ni-Cu deposits worldwide, but evidence for the source's Cu enrichment and its mechanism is still rare. Zhao et al. present a Cu isotopic study on the Jinchuan magmatic Ni-Cu deposit in China. The Cu isotopic composition of parental magmas for the Jinchuan Ni-Cu deposit was calculated to be 0.54 +/- 0.22 per mill, which is much higher than the mantle value of ~0.06 per mill. Copper transportation from oxidized subducted slabs to mantle peridotites or re-dissolution of pre-existing Cu-bearing sulfides in the mantle would cause oxidative breakdown and reprecipitation of sulfides and shift the mantle peridotites toward heavier Cu isotopic compositions. Metal enrichment in mantle source is a key step in generating giant magmatic Ni-Cu deposits worldwide, which significantly enhances our understanding of the Ni-Cu mineralization system and is vital for further mineral exploration.
Gamma radiation effects on quartz Al and Ti center electron spin resonance signal intensity: Implications for quartz provenance discrimination
https://doi.org/10.2138/am-2020-7565This diversity of quartz crystallization environments is reflected by trace-element compositional variations, which can be used to distinguish between different source sediments. Wei et al. conducted ESR measurements of quartz samples from five present-day fluvial sediments and eight ancient fluvial sand lenses from China. Results indicate that (1) different original quartz could be distinguished by element concentrations, (2) quartz Al and Ti center ESR-SI are closely related to the Al and Ti concentrations in quartz, and (3) the various provenance quartz could be separated by the ratio of quartz Al center and Ti center ESR-SI.
A new high-pressure experimental apparatus to study magmatic processes at precisely controlled redox conditions
https://doi.org/10.2138/am-2022-7977Oxygen fugacity (fO2) is typically controlled in high P-T experiments by using redox buffer assemblages. However, these do not allow fine enough resolution. Semi-permeable hydrogen membranes (Shaw 1963) are often used in internally heated pressure vessels for more flexible fO2 control in hydrous experiments; however, their implementation in more widely available externally-heated pressure vessels has not yet gained space. Alex and Zajacz present a prototype Molybdenum-Hafnium Carbide (MHC) pressure vessel apparatus that simultaneously allows flexible, precise, and accurate redox control via a custom-designed hydrogen membrane and rapid quenching. This new powerful yet cost-effective and low-maintenance apparatus may open up new pathways for studying redox reactions in hydrous magmas and magmatic fluids.
Effect of structural water on the elasticity of orthopyroxene
https://doi.org/10.2138/am-2021-7843Structural water embedded in nominally anhydrous minerals is a key factor in understanding the seismic signatures of the deep Earth. The effects of structural water on elastic properties of garnet, clinopyroxene, and olivine and its high-pressure polymorphs have been well studied. However, there is no study on the single-crystal elastic properties of hydrous orthopyroxene. Hou et al. report the effect of structural water on the elasticity of orthopyroxene at ambient conditions. Unlike the effects of structural water on elastic properties of olivine, garnet and clinopyroxene, 842-900 ppm structural water would increase C13 by 12.0(7)% and decrease C23 by 8.6(8)%. The effects on C11, C22, C33, C44, C66, KS0, and VP are subtle if not negligible when considering the uncertainties. The C55, C12, G0, and VS are not affected by the presence of structural water.
Cryogenic heat capacity measurements and thermodynamic analysis of lithium aluminum layered double hydroxides (LDHs) with intercalated chloride
https://doi.org/10.2138/am-2021-7935As lithium-ion batteries become increasingly important for new technology, researchers are finding new ways to extract lithium from different sources, including geothermal brines. Layered double hydroxides (LDHs) containing Li+ and Cl- have been shown to selectively absorb lithium from geothermal brine. Jayanthi et al. conducted heat capacity measurements to examine which factors influence LDH thermodynamic stability, including doping with iron and water/anion ratio.
A theoretical and experimental investigation of hetero- versus homo-connectivity in barium silicates
https://doi.org/10.2138/am-2021-7910Minerals are typically formed from a single type of structural unit, the SiO4 tetrahedron. Crystal structures with multiple tetrahedral types, such as in the amphiboles, are rare except when their chemistry is complex. The barium silicates form neso- to phyllo-silicate that contain structures with both singular and multiple structural units. Moulton et al. compare and contrast the signatures of mixed structural units in these phases using XRD, NMR, Raman, and ab initio calculations. The signatures of these phases are similar to those found in liquid and glassy silicates. Their simulations provide the exact structural origin of the vibrational modes, which permits unprecedented clarity of these interesting structures.
Radiation-induced changes in vanadium speciation in basaltic glasses: Implications for oxybarometry measurements using vanadium K-edge X-ray absorption spectroscopy
https://doi.org/10.2138/am-2021-7933Vanadium valence proxies for magmatic oxygen fugacity, measured using X-ray absorption near-edge spectroscopy (XANES), can provide highly sensitive determinations of the redox conditions in basaltic melts. However, X-ray beam-induced changes in V speciation will introduce uncertainty in the calculated vanadium valence that must be properly evaluated in order to make meaningful interpretations of the igneous evolution of the system. Lanzirotti et al. observed beam-induced modifications in V speciation in silicate glasses that are dependent on the radiation dose rate used during analysis. This study shows that, for most magmatic glasses, beam-induced changes in vanadium speciation can be effectively minimized, within the analytical uncertainty of the XAFS analysis, by limiting flux densities to be +/- 1 x 10^9 ph/s/μm^2.
The crystal structure of Fe2S at 90 GPa based on single-crystal X-ray diffraction techniques
https://doi.org/10.2138/am-2022-7973The Fe-S system was explored by Zurkowski et al. in a laser-heated diamond-anvil cell at 89(2) GPa, and 2380(120) K, and the structure of Fe2S was determined. The material properties of this Fe2S phase may play a role in the thermodynamics of Earth's core, adding new complexity to our knowledge of Earth's mysterious deep interior. The current results also highlight that single-crystal X-ray diffraction is an effective technique for probing the iron-sulfide phases that stabilize at these extreme conditions.
Hydration-driven stabilization and volume collapse of grain boundaries in Mg2SiO4 forsterite predicted by first-principles simulations
https://doi.org/10.2138/am-2021-7732Forsterite-water interaction plays a key role in several geochemical processes. However, the water incorporation mechanism at the interfaces and its consequences on stability is unclear. Ghosh et al. performed first-principles calculations and revealed dissociative adsorption (of water) driven volume collapse and stabilization of the material. The preferential incorporation of water at the grain boundary interfaces over the bulk implies greater importance of grain boundaries in mantle processes.
Kinetics of dehydrogenation of riebeckite: An HT-FTIR study
https://doi.org/10.2138/am-2022-8021Amphiboles are important water carriers at depth; their dehydroxylation contributes to the lithospheric electrical conductivity. The FTIR study by Della Ventura et al. addresses the kinetics of dehydration in Fe-rich sodium amphiboles and shows that: (1) the H+ diffusion through the crystal is mainly perpendicular to the silicate double chains; (2) external oxygen is necessary for H+ to leave the crystal; and (3) the H+ release occurs at low activation energy, but its diffusion requires extra energy to be effective.
Ferro-tschermakite with polysomatic chain-width disorder identified in silician magnetite from Wirrda Well, South Australia: A HAADF STEM study
https://doi.org/10.2138/am-2022-7975Scanning transmission electron microscopy using an HAADF detector enables unparalleled direct imaging of minerals and their intergrowths at the nanoscale, providing insights into petrological processes that cannot be otherwise constrained. Biopyriboles form a mineral group that combines T-sheet and -chain silicates and have been used to define polysomatism and derivation of modular series. No prior studies have addressed complex pyribole sequences, their degree of order and disorder, or the diverse range of defects present using HAADF STEM imaging. Ciobanu et al. identify ferro-tschermakite occurring as nanoinclusions in silician magnetite from Wirrda Well, South Australia, visualize the building blocks that define their crystal-structural modularity, characterize the polysomatic disorder, and discuss the petrogenetic significance of such nanoscale inclusions in the context of protracted igneous-metamorphic history.
New mineral names
https://doi.org/10.2138/am-2022-NMN107411March 2022
Structure of basaltic glass at pressures of up to 18 GPa
https://doi.org/10.2138/am-2021-7742In situ X-ray and neutron diffraction experiments were conducted by Ohashi et al. to determine the structures of basaltic glass up to 18 GPa. On compression, the mean O-O coordination number (CN_OO) starts to increase at 2-4 GPa. This rise reflects the elastic softening of fourfold-coordinated silicate glass, which may be the origin of anomalies of elastic moduli in basaltic glass at ~2 GPa (Liu and Lin 2014). The mean O-O distance and CN_OO change their compression behaviors along with an increase in the Si(Al)-O coordination number (CN_Si(Al)O) around 10 GPa.. Their results clarified that the oxygen packing fraction (eta_O) exceeds the value for the random close packing, suggesting that the eta_O cannot explain the pressure-induced structural changes of silica and silicate glasses. They also found As predicted by a recent ab initio simulation (Majumdar et al. 2020), the increase in CN_Si(Al)O can trigger the increase of viscosity, which can result in stabilizing basaltic magma in the deep Earth.
Synthesis of calcium orthocarbonate, Ca2CO4-Pnma at P-T conditions of Earth's transition zone and lower mantle
https://doi.org/10.2138/am-2021-7872Binck et al. report the crystal structure of calcium orthocarbonate, Ca2CO4-Pnma, synthesized in a diamond anvil cell at 20 and 89 GPa after laser heating to 1830 and 2500 K, respectively. The structure was solved and refined by synchrotron single crystal X-ray diffraction measurements. Raman spectroscopy and DFT calculations confirm the single-crystal results and provide further crystal chemical information, such as bulk and polyhedral compressibilities. The results provide an experimental basis for the recent discussion on the geophysical relevance of orthocarbonates and strengthen the argument that this orthocarbonate needs to be considered in studies of the carbon cycle by showing that at pressures of the transition zone and Earth's upper lower mantle, Ca2CO4 + 2SiO2 is more stable than 2CaSiO3 + CO2.
Melting phase relation of Fe-bearing Phase D up to the uppermost lower mantle
https://doi.org/10.2138/am-2021-7907Water (H+ or OH-) can be transported into the deep Earth by cold subducting slabs in the form of hydrous phases (DHMSs). Phase D is proposed to be one of the most important water holders in the uppermost lower mantle. Few experiments have been conducted so far to clarify the effect of Fe in DHMSs. Xu et al. investigated the stability of Fe-bearing phase D (PhD) in the AlOOH-FeOOH-PhD system at high temperature and high pressure. Although Fe decreases the stability region of PhD, aluminum- and iron-bearing PhD drastically shift PhD stability to higher temperatures in both MORB and pyrolite compositions compared to pure Mg-PhD. Therefore, Al-, Fe-bearing PhD could act as a water reservoir druing subduction to the deep lower mantle.
Evidence from HP/UHP metasediment for recycling of isotopically heterogeneous potassium into the mantle
https://doi.org/10.2138/am-2021-7923Stable potassium isotope systematics have great potential for tracing recycled sediments in the mantle due to the stark contrast in K abundance and isotopic ratios between subducting sediment and the mantle. However, whether the distinct K isotopic signatures of sediments could be transported to mantle depths is limited by the lack of knowledge on K isotope behavior during subduction zone metamorphism. Wang et al. investigate the K isotopic composition of a suite of well-characterized metasediments and their protoliths from the western Alps. Their results suggest that prograde metamorphism has limited influence on the K isotopic compositions of subducting sediments and the significant K isotopic variation in metasediments reflects K isotope heterogeneity produced in their protoliths during chemical weathering. These findings provide evidence for the transport of isotopically heterogeneous K from subducting sediments into the mantle.
Effect of sulfur on siderophile element partitioning between olivine and martian mantle melt
https://doi.org/10.2138/am-2021-7743Apparent incompatible behavior of Ni and Co in martian magmas cannot be explained by a chemical effect due to the presence of sulfur. Instead, as described by Usui et al., the anomalous behavior must be due to mixing of magmas, assimilation of Co-rich material into the magma, or a kinetic crystallization effect.
Gold speciation in hydrothermal fluids revealed by in situ high energy resolution X-ray absorption spectroscopy
https://doi.org/10.2138/am-2022-8008A combination of in situ spectroscopy and molecular modeling reveals the identity, structure, and stability of the major aqueous complexes transporting gold by hydrothermal fluids and highlights an important role of the trisulfur ion in gold transfer and concentration in hydrothermal-magmatic deposits of subduction zones. This approach by Pokrovski et al. opens new perspectives for studies of metals in geological fluids, and potentially silicate melts, inaccessible to direct observation in nature.
Characterization of carbon phases in Yamato 74123 ureilite to constrain the meteorite shock history
https://doi.org/10.2138/am-2021-7856Barbaro et al. investigated a carbon-bearing ureilitic fragment of Yamato 74123 by scanning electron microscopy, micro-Raman spectroscopy, and micro X-ray diffraction, to add further information on diamond and graphite formation in ureilites. Their results show the presence of both microdiamonds and nanodiamonds together with nanographite. The formation process of these phases can be ascribed to impact shock event(s), with the assistance of Fe-Ni melts catalysis during diamond growth.
Pressure-induced structural phase transitions in natural kaolinite investigated by Raman spectroscopy and electrical conductivity
https://doi.org/10.2138/am-2021-7863Raman scattering and electrical conductivity have been applied to investigate the pressure-induced structural transitions of natural kaolinite occurring at 2.9 and 6.5 GPa, which is characterized by an obvious inflection point in pressure-dependent Raman shifts and electrical conductivity. The influence of temperature on two phase transformations of natural kaolinite were studied by high-temperature and high-pressure electrical conductivity measurements and anticorrelations between the phase transition temperature and pressure found. Hong et al. obtained the stability field of a high-pressure polytype of kaolinite along the cold and hot subduction zones. Along a cold subduction path, the kaolinite I phase remains stable below ~60 km. At depths greater than ~60 km, the kaolinite II phase appears. However, under hot subduction conditions, kaolinite I phase occurs at shallower depth of around 40 km. Above ~40 km, kaolinite II phase forms. Their results indicate that the kaolinite II phase has the potential to enter the mantle via both cold and hot subduction zones and serves as a source of aluminum, silicon, and hydrogen to Earth's interior.
Magnetite-rutile symplectite in ilmenite records magma hydration in layered intrusions
https://doi.org/10.2138/am-2021-7777Tan et al. integrated electron backscatter diffraction (EBSD) and focused-ion beam - energy dispersive X-ray spectroscopy (FIB-EDS) tomography to investigate the crystallographic orientation and 3-D morphology of magnetite-rutile symplectite in ilmenite. Their results indicate that the symplectite is related to mineral assemblages crystallized from hydrated magmas. The magnetite-rutile symplectite can be considered as a textural indicator of magma hydration during the evolution of terrestrial, martian, and lunar magmas.
Ferromagnesian jeffbenite synthesized at 15 GPa and 1200°C
https://doi.org/10.2138/am-2021-7852Smyth et al. describe the first multi-anvil press synthesis and characterization of recently named mineral jeffbenite, which occurs as inclusions in diamonds thought to have formed in the transition zone or lower mantle of the Earth. Because the composition of natural samples appears to overlap that of garnet, previous studies have proposed that the mineral may result from a metastable inversion from bridgmanite, the major mineral of the lower mantle. The synthesis was accomplished with a composition rich in ferric iron rather than aluminum at a pressure of 15 GPa and a temperature of 1200°C. Experiments produced high-quality single crystals up to 200 micrometers in size that allowed characterization by multiple analytical methods, including single-crystal X-ray diffraction, electron microprobe, Raman and infrared spectroscopy, and synchrotron Mossbauer spectroscopy. While natural samples are rich in aluminum, experimental samples are rich in ferric iron that permitted high precision site occupancy refinement by X-ray diffraction, and analysis by synchrotron Mossbauer spectroscopy. Because of significant Fe occupancy in one of the tetrahedral sites, the stoichiometry is sufficiently distinct from garnet that the mineral may be a stable phase in the transition zone of the mantle and does not require (or preclude) an origin as a metastable inversion product from a higher-pressure phase such as bridgmanite.
Electrical conductivity of metasomatized lithology in subcontinental lithosphere
https://doi.org/10.2138/am-2021-7942Peng et al. explored the electrical conductivity (EC) of a natural metasomatized rock sample and compared those results with magnetotelluric (MT) observations to evaluate mantle metasomatism as a viable mechanism to explain the mid-lithospheric discontinuity (MLD). They found that Na concentrations in amphiboles will enhance the bulk EC of metasomatized rock significantly due to superionic conduction. High EC will cause a positive anomaly in EC profiles at MLD depths that is contrary to MT observations in MLD, so metasomatism is unlikely to explain the seismic and MT observations simultaneously.
Measurements of the Lamb-Mossbauer factor at simultaneous high-pressure-temperature conditions and estimates of the equilibrium isotopic fractionation of iron
https://doi.org/10.2138/am-2021-7884Zhang et al. present an original study of the Lamb-Mossbauer factor of γ- and ε-Fe at high pressures and high temperatures. The measurements of the Lamb-Mossbauer factor of Fe were carried out by combining laser-heated diamond anvil cells, synchrotron Mossbauer spectroscopy, and a recently developed unique fast temperature readout spectrometer. The vibrational behavior of Fe under these conditions is consistent with quasiharmonicity, therefore the quasiharmonic Debye model was used to calculate the stiffness of the γ- and ε-Fe at elevated pressure-temperature conditions from the Lamb-Mossbauer factor and the atomic mean-square displacement. From the stiffness data, the equilibrium isotopic fractionation β-factor of iron is calculated at high pressures and high temperatures. Calculations based on their experimental data demonstrate that the quasiharmonic correction would lower ln βFe57/54 by 0.1 per mil; compared to the typicallyused harmonic extrapolation of room temperature nuclear resonant inelastic X-ray scattering data. The quasiharmonic correction to the ln βFe57/54 for Fe determined by this method is five times the upper bound of previous estimates and implications of this result to iron isotope fractionation inside the earth is discussed.
Element mobility and oxygen isotope systematics during submarine alteration of basaltic glass
https://doi.org/10.2138/am-2021-7831He et al. demonstrate that elemental imaging of altered basaltic glass reveals major and trace elements fluxes when basaltic glass altered into palagonite. The palagonite is formed through congruent dissolution of glass and reprecipitation based on elemental distribution and oxygen isotopic investigations. The proposed alteration mechanism (selective leaching, congruent dissolution, initial precipitation, and post-polygonization provides insight into geochemical cycles and oceanic crust alteration.
Dissolved silica-catalyzed disordered dolomite precipitation
https://doi.org/10.2138/am-2021-7474Fang and Xu present an abiotic mechanism of dissolved-silica catalyzed dolomite precipitation that provides new insight into the long-lasting "dolomite problem". They demonstrate that the presence of 1-2 mM of aqueous Si(OH)4 in high Mg:Ca ratio solutions at room temperature will promote disordered dolomite (with up to 48.7 mol% MgCO3) precipitation and inhibit aragonite precipitation. Dissolved silica in solution also promotes Mg incorporation into Ca-Mg carbonates. Room temperature dolomite synthesis experiments all require the presence of dissolved molecules with low dipole moments such as hydrogen sulfide, dioxane, polysaccharide, and exopolymeric substances (EPS). The molecules with low dipole moment adsorbed on the dolomite surface can lower the dehydration energy of a surface Mg2+-water complex and promote dolomite nucleation and growth. This study provides a new model for abiotic sedimentary dolomite formation that is likely to be responsible for the significant amount of primary dolomite in the Earth history.
Elasticity and high pressure behavior of Mg2Cr2O5 and CaTi2O4-type phases of magnesiochromite and chromite
https://doi.org/10.2138/am-2021-7853Shieh et al. synthesized three high-pressure polymorphs of chromite spinels and report the equation of state of the CaTi2O4-type (CT) phase of magnesiochromite, natural chromite, and mLd-type Mg2Cr2O5. Among the three CT phases, FeCr2O4 CT has larger volumes, MgCr2O4 CT has moderate volumes, and natural chromite has the smallest volumes. The appearance of CT and mLd phases can be used as an indicator for shock pressures.
Significance of tridymite distribution during cooling and vapor-phase alteration of ignimbrites
https://doi.org/10.2138/am-2021-7814Heled et al. investigated the distribution of the silica polymorph tridymite in silicic ignimbrites. Their findings indicate tridymite is formed at the contact between ignimbrite flow packages where the overlying volcanic material seals in gases, allowing prolonged crystallization localized to flow package contacts. Implications of this mineralogical fingerprint of ignimbrite structure include insight to initial cooling and alteration history, and potential geothermal reservoir modeling.
Micropores and mass transfer in the formation of myrmekites
https://doi.org/10.2138/am-2021-7956This study by Yuguchi et al. expands the existing knowledge of myrmekitization within a granitic pluton and its underlying fluid chemistry. The core objectives of this work include: a quantitative estimate of mass transfer between the reactant and product minerals, and the inflow and outflow of components with consideration of the volume change due to micropore formation; the factors controlling the formation of micropores during myrmekitization; and the sequential variations in the hydrothermal fluid chemistry during sub-solidus conditions.
Mn3+ and the pink color of gem-quality euclase from northeast Brazil
https://doi.org/10.2138/am-2021-7838Graphical Abstract in place of words is provided here.
Geochemistry and boron isotope compositions of tourmaline from the granite-greisen-quartz vein system in Dayishan pluton, Southern China: Implications for the potential mineralization
https://doi.org/10.2138/am-2021-7591Chemical and boron isotopic compositions of tourmaline in granite, greisen, and quartz veins are reported by Zhao et al. The tourmalines formed from late-magmatic and hydrothermal stage. The slightly lighter B isotopic values of hydrothermal tourmalines are probably caused by fluid mixing. These results indicate that the Sn- and Zn-enriched fluids that created the Dayishan ore deposits may come from the host rock.
Lazaraskeite, Cu(C2H3O3)2, the first organic mineral containing glycolate, from the Santa Catalina Mountains, Tucson, Arizona, U.S.A.
https://doi.org/10.2138/am-2021-7895Yang et al. demonstrate that lazaraskeite is the first organic mineral found that contains glycolate. Not only does its discovery imply that more glycolate minerals may be found, but it also suggests that glycolate minerals may serve as a potential storage site for biologically fixed carbon.
Textural, fluid inclusion, and in-situ oxygen isotope studies of quartz: Constraints on vein formation, disequilibrium fractionation, and gold precipitation at the Bilihe gold deposit, Inner Mongolia, China
https://doi.org/10.2138/am-2021-7823By study of quartz CL textures, fluid inclusion microthermometry, and in situ O-isotope analysis, Qiao et al. show the complicated quartz textures and fluctuating δ18O variations in Bilihe deposit, the disequilibrium oxygen isotopic fractionation between quartz and ore-forming fluid, and the influence of meteoric water on the mineralizing processes of porphyry gold deposits.
Immiscible metallic melts in the upper mantle beneath Mount Carmel, Israel: Silicides, phosphides, and carbides
https://doi.org/10.2138/am-2021-7934Griffin et al. found aggregates of skeletal corundum crystals from pyroclastic volcanoes on Mt. Carmel, Israel, and trapped pockets of silicate melt with mineral assemblages that require a coexisting fluid dominated by methane and hydrogen. Balls of Fe-Ti-Zr silicides and phosphides represent immiscible melts that separated from the silicate melt. Microstructures in the silicide-melt inclusions show progressive further exsolution of more evolved melts, rich in Zr, P, and Ti, as temperature and oxygen fugacity decreased until crystallization was ended by explosive eruption.
February 2022
Alumino-oxy-rossmanite from pegmatites in Variscan metamorphic rocks from Eibenstein an der Thaya, Lower Austria, Austria: A new tourmaline that represents the most Al-rich end-member composition
https://doi.org/10.2138/am-2022-8047Ertl et al. investigated the new (pink) tourmaline alumino-oxy-rossmanite, which is the Al-richest known tourmaline. The significant content of tetrahedrally-coordinated Al is an indication for relatively high-temperature conditions (~700 °C) during formation of the pegmatite. Because of the low quantity of associated mica, the silica melt, which formed this pegmatite, may have crystallized under relatively dry conditions, in agreement with the observation that alumino-oxy-rossmanite contains a lower amount of OH than most other tourmalines. The geological unit, which contains the tourmaline-bearing pegmatites, consists of a thick crystalline complex and exposes medium- to high-grade metamorphic rocks with Paleoproterozoic to Devonian protolith ages. Such pegmatites could have formed from chemically simple metasediments during metamorphosis. Alumino-oxy-rossmanite was named because of its chemistry and was also named after Prof. George R. Rossman at the California Institute of Technology.
Fluorine partitioning between quadrilateral clinopyroxenes and melt
https://doi.org/10.2138/am-2021-7868Baker et al. measured the partitioning of F between clinopyroxene and silicate melt in 13 experiments at a variety of pressures, temperatures, and melt compositions ranging from basaltic to dacitic at hydrous and anhydrous conditions. Additionally, they determined the crystal-melt partitioning of F for 4 experiments with plagioclase, 2 with orthopyroxene, and 1 with olivine. The clinopyroxene/melt partition coefficients of F were linearly dependent upon the concentration of aluminum in the octahedral M1 site of clinopyroxene. Similar relationships were seen in previous measurements of the fluorine partition coefficient between clinopyroxene and melt, but each study's correlation between the F partition coefficient and pyroxene chemistry was unique, apparently due to differing analytical protocols. Nevertheless, the authors conclude that the self-consistency of each study indicates that F partition coefficients determined using one protocol can be applied to minerals or glasses analyzed using the same protocol to better understand the storage and transport of fluorine in magmatic systems.
Multi-stage magma evolution recorded by apatite and zircon of adakite-like rocks: A case study from the Shatanjiao intrusion, Tongling region, Eastern China
https://doi.org/10.2138/am-2021-7871Cao et al. report LA-ICP-MS zircon U-Pb ages as well as the trace element compositions and Sr isotopes of apatite from the Shatanjiao granites in the Tongling region, South China. Based on the obtained chemical compositions, they found that some specific elements in the apatite, such as Sr, Th, U, and REE, might provide a new insight on the magma evolutionary process from the perspective of apatite.
The physical and chemical evolution of magmatic fluids in near-solidus silicic magma reservoirs: Implications for the formation of pegmatites
https://doi.org/10.2138/am-2021-7915Over the past decades, magmas are increasingly recognized as being stored over long timescales in near-solidus crystal-rich "mush" systems. This concept provides a physical and mechanical framework for the chemical processes recorded in volcanic and plutonic rocks around the world. Troch et al. assess the implications of this model for the formation of pegmatites. Combining thermomechanical and trace element models suggests that pegmatites may be precipitated from a magmatic volatile phase and mark the magmatic-hydrothermal transition in a continuous crustal distillation column.
Texture, geochemistry and geochronology of titanite and pyrite: Fingerprint of magmatic-hydrothermal fertile fluids in the Jiaodong Au province
https://doi.org/10.2138/am-2021-7889Li et al. conducted textural, geochemical (LA-(MC)-ICPMS trace element, Nd and S isotopes) and geochronological (LA-ICPMS U-Pb dating) analyses of titanite and pyrite from monzonite in the Jiaodong Au province, China. Their main results are: (1) Different types of titanite (magmatic Ttn1 and hydrothermal Ttn2, Ttn3) and pyrite (magmatic Py1, Py4 and hydrothermal Py2, Py3) were distinguished; (2) Titanite and monazite U-Pb dating constrains the monzonitic magma and hydrothermal events to ca. 119 Ma; and (3) Fertile hydrothermal fluids were exsolved from the monzonitic magma at ca. 119 Ma, contributing to the gold mineralization.
Polytypism in semi-disordered lizardite and amesite by low-dose HAADF-STEM
https://doi.org/10.2138/am-2021-7867Zang et al. demonstrated the powerful imaging technique, low-dose annular dark-field scanning transmission electron microscopy, with 1-Å resolution of highly beam sensitive clay materials, and unraveled the short-range ordering in semi-disordered serpentine. Using this method, they visualized oxygen atomic columns, revealed within-sheet shifts, determined the prevalence of regular alternating octahedral tilts in lizardite and amesite, and resolved the ordered structure with long-period complex octahedral tilts in lizardite. This research lays the foundation for atomic and nano-scale characterization of the structures and microstructures of a wide variety of clays and other beam sensitive materials.
Local peralkalinity in peraluminous granitic pegmatites. I. Evidence from whewellite and hydrogen carbonate in fluid inclusions
https://doi.org/10.2138/am-2021-7790Carbonates and hydrogen carbonates have fairly often been reported to occur in spodumene-bearing and other peraluminous granitic pegmatites, mostly in fluid inclusions. Their origin is still unclear as these minerals do not form in granites. Liu et al. studied fluid inclusions in several pegmatite minerals (mostly from the Alto Ligonha Province, Mozambique) using Raman spectroscopy to determine the carbon species. Besides the ubiquitous CO2, they detected CH4 in the vapor phase, hydrogen carbonate in the aqueous liquid, a calcite-group mineral and, to their surprise, whewellite. The latter occurs in fluid inclusions in topaz from Khoroshiv (Volodarsk-Volynskii), Ukraine, and is the first report of an oxalate from a peraluminous granitic pegmatite. Whewellite forms in peralkaline pegmatites via reaction of reduced carbonaceus material with an alkaline fluid. Based on the detection of whewellite and hydrogen carbonate, they conclude that peralkaline fluid or melt can locally be present during the evolution of a peraluminous granitic pegmatite.
Peralkalinity in peraluminous granitic pegmatites. II. Evidence from experiments on carbonate formation in spodumene-bearing assemblages
https://doi.org/10.2138/am-2021-7909Spodumene-hosted aqueous fluid inclusions can contain zabuyelite [Li2(CO3)], and additionally CO2, cookeite, quartz or cristobalite, albite, pollucite or analcime, and calcite. However, their origin is still unclear. Liu et al. used Raman spectroscopy combined with a hydrothermal diamond-anvil cell to study at which conditions carbonate and hydrogen carbonate can be generated from spodumene, CO2, and H2O. The obtained results indicate that the formation of carbonate (as zabuyelite and/or dissolved in the aqueous fluid) in spodumene+H2O+CO2-bearing assemblages requires a peralkaline fluid. They conclude that hydrogen carbonate-rich peralkaline fluid or melt may be involved in the evolution of a peraluminous granitic pegmatite. Note: Supplement added post-publication of pictures of spodumene.
Click here for Online Material added post publication
Ab initio study of structural, elastic and thermodynamic properties of Fe3S at high pressure: implications for planetary cores
https://doi.org/10.2138/am-2021-7268Sulfur is potentially one of the most favored light elements to be alloyed with iron in terrestrial planetary cores. It is therefore of primary importance to constrain the physical properties of Fe-S alloys under extreme conditions. Valencia et al. present a detail study of several physical properties of the tetragonal phase of Fe3S at high pressure. They discuss variations of the thermodynamic, elastic and magnetic properties of this phase as a function of pressure as well as the behavior of sound velocities with direction of propagation and magnetic effects. They use these results to discuss the possibility of Fe3S being a crystalline phase within Earth's inner core and Mars core. They estimate the amount of sulfur that could be present in the core if this phase is present together with pure Fe.
Removal of barite from zircon using an aqueous solution of diethylenetriaminepentaacetic acid and potassium carbonate
https://doi.org/10.2138/am-2021-7906In most geologic applications, if barite is present, it must be separated from zircon to enable analysis of the zircon. Current methods of barite removal include mechanical comminution in a ball mill or conversion to barium carbonate by boiling in an aqueous solution of sodium carbonate. Martin and Rocha-Estopier optimized an alternative technique for barite removal. In repeated experiments, boiling for one hour in an aqueous solution of DTPA and potassium carbonate dissolved about 90% of sand-size barite grains. This solution attacked the surfaces of barite crystals and it did not attack the surfaces of zircon crystals at the microscopic level.
Improving grain size analysis using computer vision techniques and the implications for grain growth kinetics
https://doi.org/10.2138/am-2021-7797Ezad et al. has investigated the reliability of grain size analysis from 2D photomicrograph images and find researcher bias in oversampling large grains and entirely missing small ones. This leads to discrepancies in the average grain size of greater than 5%, for multiple expert researchers analyzing the same photomicrograph. To reduce human bias and speed up the laborious process of image analysis, they developed an alternative automated user-friendly workflow to analyze and report grain size, at a fraction of the time required through manual image processing. Their new automated workflow increases the number of grains identified and indexed by 68%, compared to the same images analyzed through old manual techniques. Additionally, they report smaller errors on the estimated average grain size and provide a consistent and transparent method of grain size analysis. Their workflow is available via GitHub for automated, reproduceable grain size analysis from 2D images.
Crystal chemistry of arsenian pyrites: A Raman spectroscopic study
https://doi.org/10.2138/am-2021-7806Zhang et al. investigated the occurring modes of As within natural arsenian pyrite and presented direct Raman evidence for the As-induced structural variations in pyrite. The key findings are: (1) The wavenumbers of Raman vibrational modes vary approximately linearly with the As content in pyrite, correlating with the change in bond constants with increasing substitution of As for S; and (2) The linewidth of the Raman vibrational mode increases with increasing As content in pyrite, which is attributed to the increase in lattice strain associated with the substitution of As for S. These findings (1) highlight the significance of systematic Raman spectroscopic investigation in interpreting the effects of elemental substitution on the structure; (2) advance the understanding of the enrichment mechanism of valuable metals in sulfides; and (3) provide guidance on controlling the release of toxic heavy ions into the environment.
Formation of the Maoniuping giant REE deposit: Constraints from mineralogy and in situ bastnasite U-Pb geochronology
https://doi.org/10.2138/am-2021-7778Weng et al. investigated the time and processes of hydrothermal mineralization in the Maoniuping giant rare earth elements (REE) deposit, China. Major results include: (1) Magmatic and hydrothermal bastnasite from the Maoniuping giant REE deposit has been recognized, with U-Pb ages ranging from 28.2 Ma to 25.8 Ma; (2) Clinopyroxene and apatite compositions provide supporting for the separation of pegmatite dikes from the carbonatite rather than the syenite; (3) A continuous magmatic-hydrothermal mineralization model during the evolution of alkaline-carbonatite complex from the Maoniuping giant REE deposit was proposed; and (4) Bastnasite can be used as a powerful mineral proxy for magmatic-hydrothermal evolution and associated REE mineralization in alkaline-carbonatite complexes.
Amphibole as a witness of chromitite formation and fluid metasomatism in ophiolites
https://doi.org/10.2138/am-2021-7593Pan et al. present new occurrences of amphibole in a suite of chromitites, dunites and harzburgites from the mantle sequence of the Lycian ophiolite in the Tauride Belt, Turkey. The amphibole occurs both as interstitial grains among the major constituent minerals and as inclusions in chromite grains. A comparison of amphibole inclusions in chromite with interstitial grains provides direct evidence for the involvement of water in chromitite formation and the presence of hydrous melt/fluid metasomatism in the peridotites during initial subduction of Neo-Tethyan oceanic lithosphere. The hydrous melts/fluids were released from the chromitites after being collected on chromite surfaces during crystallization. Considering the wide distribution of podiform chromitites in this ophiolite, the link between chromitite formation and melt/fluid metasomatism may be applicable to other ophiolites worldwide.
Ferro-papikeite, ideally NaFe2+2(Fe2+3Al2)(Si5Al3)O22(OH)2, a new orthorhombic amphibole from Nordmark (Western Bergslagen), Sweden: Description and crystal structure
https://doi.org/10.2138/am-2021-7877The orthorhombic Mg-Fe-Mn amphiboles are very common rock-forming minerals in a variety of metamorphic and igneous rocks. Low-Na compositions are extremely common, but high-Na compositions are less common, and have a rather grey and hidden existence in the scientific literature as they have no mineral names. With the characterization of ferro-papikeite as an IMA-approved mineral species by Hawthorne et al., amphiboles of these compositions will become much more visible in the literature and their significance can begin to be understood. In particular, the authors show that the parallels between monoclinic and orthorhombic amphiboles are even stronger than previously realized and provide an understanding of why compositions in the fields of edenite and rootname 1 are of considerable interest, both with regard to the stability of the amphibole structure as a function of chemical composition and the possible relations to the carcinogenic properties of fibrous fluoro-edenite.
HP-PbF2-type FeCl2 as a potential Cl-carrier in the deep Earth
https://doi.org/10.2138/am-2022-8283Hydrogen and chlorine cycles are closely related via subduction into the Earth's mantle. The actual budget and distribution of H and Cl in the deep Earth remains an open question, partly due to the lack of knowledge on the deep geological processes. Yuan et al. report the discovery of a dense cubic HP-PbF2-type FeCl2 phase, through reactions between Fe-bearing silicate/hydroxide and NaCl in a diamond anvil cell under hydrous deep lower mantle conditions. They present evidence of HP-PbF2-type FeCl2 phase coexisting with a major mineral, Na,Fe-bearing MgSiO3 post-perovskite, in the lowermost mantle. Combining with previous observation of a highly stable hydroxide FeO2Hx with the identical symmetry of Pa-3, they suggest that this topology is an important hosting candidate for storage of both H and Cl. The possible presence of volatiles such as H and Cl could likely change the composition and iron valence state in post-perovskite in the lowermost mantle.
January 2022
MSA at 100 and why optical mineralogy still matters: The optical properties of talc
https://doi.org/10.2138/am-2022-8066This open access paper by Mickey E. Gunter memorializes MSA’s 100th anniversary, reminds us why optical mineralogy still matters and provides original data for the optical properties of talc.
Boron isotope compositions establish the origin of marble from metamorphic complexes: Quebec, New York, and Sri Lanka
https://doi.org/10.2138/am-2021-7811Boron abundances and isotopic signatures, along with other geochemical data, are used in this study by Kuebler et al. to determine the origin of multi-colored marble units found in both the Grenville Province and southwestern Sri Lanka. Samples from the Grenville Province represent marble formed during high-temperature metamorphism of limestone units. Sri Lankan samples were formed from carbonate-rich and B-11-poor fluids derived from a crustal source. This work establishes three isotopically distinct fields for carbonate: mantle-derived, sedimentary, and metasedimentary.
Celleriite, a new mineral species of the tourmaline supergroup
https://doi.org/10.2138/am-2021-7818Bosi et al. describe the structure and chemical characteristics of new tourmaline mineral (celleriite) recently approved by the IMA-CNMNC, and its genesis is in the pegmatite environment. Celleriite appears to be valuable in understanding Earth as a complex, evolving system in which metasomatic fluid-rock interactions may lead to new mineral-forming environments, in particular for B minerals. Moreover, the crystal overgrowth of celleriite implies that while the dark termination (the so-called Moor’s head textural type) is characteristic for the Elba Island pegmatites, it is not a feature exclusively produced by Fe enrichment but may also imply the presence of a Mn-dominant component.
Jingsuiite, TiB2, a new mineral from the Cr-11 podiform chromitite orebody, Luobusa ophiolite, Tibet, China: Implications for recycling of boron
https://doi.org/10.2138/am-2021-7647The lithophile element boron is generally taken to be quintessentially crustal since it is greatly enriched in the upper continental crust relative to primitive mantle by processes such as weathering, adsorption onto clay minerals in marine sediments, partial melting of sedimentary rocks, and differentiation of the resulting granitic melts. However, under highly reducing conditions, boron becomes siderophile, much preferring intermetallic melts to silicate melts (Griffin et al. 2020). This switch in behavior has resulted in the crystallization of a new B mineral found by Xiong et al. in association with Ti intermetallics such as osbornite, khamrabaevite, Ti10(Si,P)6-7, and Ti-Fe silicides during rapid exhumation from the upper mantle. The presence of qingsongite (cubic BN) in a fragment of crustal rock that had been buried at 400 km depth implies B can be recycled back to the mantle, yet how B is retained in the subducting slab is an open question. Dehydration and melting, the processes receiving the most attention for their impact on the subducted rocks, lead to extraction of B from the subducting slab. Could the siderophile behavior of B under highly reducing conditions play a major role in retaining B in the subducted slab as it does in rapidly exhumed rocks at Mount Carmel and Cr-11? Although the minerals associated with qingsongite and N isotopes in osbornite indicate crystallization at 12 GPa at 400 km depth, there are several striking similarities between the qingsongite-bearing fragment and jingsuiite-bearing assemblages included in corundum.
Incorporation of incompatible trace elements into molybdenite: Layered PbS precipitates within molybdenite
https://doi.org/10.2138/am-2021-7609Yang et al. found that several percentages of Pb atoms can incorporate into the structure of molybdenite at high temperature. The incompatible Pb atoms tend to migrate along the (001) plane forming nano-scale Pb heterogeneity within the structure of molybdenite. The incorporation of Pb atoms can lead to stacking disorder of S-Mo-S “sandwich” layers in molybdenite, which transforms into a Pb-poor ordered 2H1 polytype with the exsolution of Pb. The migration and storage of incompatible Pb in molybdenite may shed new light on the understanding of heterogeneity of incompatible trace elements in molybdenite and may better constrain geochemical data obtained by in-situ analysis techniques.
Experimental melt inclusion homogenization in a hydrothermal diamond-anvil cell: A comparison with homogenization at one atmosphere
https://doi.org/10.2138/am-2021-7781Li et al. compared measured phase transition temperatures of melt inclusions (MIs) using HDAC and Linkam stages. Phase transition temperatures in HDAC group were as much as 374 °C lower than that of the Linkam stage. They assessed the reliability of HDAC in the MIs actual phase transition temperatures measurement. They concluded that HDAC is the preferred platform for MIs homogenization with advantages of in situ observation of phase changes during heating while elevating external pressure on MIs
Thermoelastic Properties of Zircon: Implications for Geothermobarometry
https://doi.org/10.2138/am-2021-7731Ehlers et al. report new data and combine it with previously published data to calculate an EoS for non-metamict zircon that is as accurate and precise as current experimental capabilities allow. The new EoS reported here opens up the opportunity for the conditions of zircon entrapment during metamorphism to be combined with dating on the zircons themselves to provide P-T-time points in metamorphic histories.
A Rayleigh Model of Cesium Fractionation in Granite-Pegmatite Systems
https://doi.org/10.2138/am-2021-7855David London models the fractionation of Cs (and Rb) in granite-pegmatite systems and verifies that model in the field, demonstrating the model’s validity. Primary, igneous feldspars, and micas record chemical fractionation of Cs that results from the crystallization of these phases from silicate liquid without the involvement of an aqueous solution. The model demonstrates that the fractionation of Cs between feldspars and aqueous solution would yield low or decreasing Cs contents in the feldspars that are not those of the principal chemical trends that are found in pegmatites. Loss of Cs is observed only in the latest stages of pegmatite consolidation, when miarolitic cavities form, and when primary K-feldspar, whether perthitic or not, experiences hydrothermal alteration in an open system. The conclusion of the manuscript is that the patterns of K/Rb or K/Cs in K-feldspar and micas in pegmatites conform to crystal-melt fractionation in which an aqueous solution played no part.
The atomic arrangement and electronic interactions in vonsenite at 295, 100, and 90 K
https://doi.org/10.2138/am-2021-7851Maderazzo et al. present structure refinements of a natural vonsenite at 295, 100, and 90 K. They observe and characterize a structural phase transition in the temperature range covered. They consider the electronic distribution observed, introduce the Peierls instability and its consequences, and provide context for the electronic interactions and correlations observed in the phase.
Oxalate formation by Aspergillus niger on manganese ore minerals
https://doi.org/10.2138/am-2021-7651Microscopic fungi play an important role in rock and minerals alteration, often leading to formation of insoluble biogenic oxalates (oxalic acid salts) on their surface. In recent years, the world scientific community has shown significant interest in the mechanisms of biomineralization with the participation of microorganisms, which is associated with the study of modern mineral formation at the nano and micro levels, as well as processes and phenomena occurring at the border of living and nonliving. These fundamental results, in this paper by Frank-Kamenetskaya et al., create the scientific basis for the development of modern nature-like biotechnologies in various fields of science.
High-Pressure Experimental Study of Tetragonal CaSiO3-Perovskite to 200 GPa
https://doi.org/10.2138/am-2021-7913Because the addition of Ti could elevate the phase transition temperature of CaSiO3-perovskite from the tetragonal to cubic phase studies on the physical properties of tetragonal CaSiO3-perovskite are important to better understand the structure of the Earth’s lower mantle. Here structure and equation of state of tetragonal CaSiO3-perovskite were determined up to 200 GPa. Sun et al. further compare the density and sound velocity of tetragonal CaSiO3-perovskite to the cubic phase to discuss the importance of this phase transition in the lower mantle.
Mesoproterozoic seafloor authigenic glauconite-berthierine: Indicator of enhanced reverse weathering on early Earth
https://doi.org/10.2138/am-2021-7904Ma et al. report several results. (1) Seafloor authigenic glauconite and berthierine occur in the 1.4 Ga basal Xiamaling Formation, NC. (2) Reverse weathering (authigenic formation of clay minerals) is enhanced by Fe- and Si-rich seawater. (3) Mid-Proterozoic enhanced reverse weathering maintained high CO2 levels and a warm climate.
Chemical variability in vyacheslavite, U(PO4)(OH): Crystal-chemical implications for hydrous and hydroxylated U4+, Ca, and REE phosphates
https://doi.org/10.2138/am-2021-7875Steciuk et al. show that vyacheslavite, ideally U(PO4)(OH), has a greater chemical variability than originally thought. This variability is manifested namely by the presence of Ca and REEs and consequently by the increased content of H2O (despite pure vyacheslavite being considered as anhydrous phase ). Vyacheslavite, along with ningyoite and rhabdophane, is an important source of U in the sedimentary U-deposits all around the world.
Bennesherite — a new melilite group mineral from the Hatrurim Basin, Negev Desert, Israel.
https://doi.org/10.2138/am-2021-7747Krzatala et al. report a discovery of a new mineral, bennesherite—the first barium member of the melilite group. This mineral was found in a rankinite paralava from the Hatrurim Basin, Negev Desert, Israel. Bennesherite is the only Ba- and Fe-mineral in the melilite group, although iron is a usual minor element in other melilite-group minerals. The presence of Fe2+ in bennesherite sheds light on the problem of rankinite paralavas genesis.
Single-crystal elasticity of phase Egg by Brillouin spectroscopy
https://doi.org/10.2138/am-2022-8056Wang et al. report the full elastic constants of phase Egg and δ-AlOOH determined by Brillouin scattering measurements at ambient conditions. Their results indicate that the hydrogen-bond configurations in the crystal structures of the two phases have a profound effect on their principal elastic constants. The anisotropic factors and aggregated elastic properties are determined from obtained elastic constants and compared with other typical mantle minerals. The results indicate phase Egg is likely a candidate mineral causing seismic anisotropy in subducting slabs and may result in a high-velocity anomaly at a depth of the base of the upper mantle, while δ-AlOOH may result in a high-velocity anomaly at a depth of mantle transition zone.
On the Origin of Martian Blueberries
https://doi.org/10.2138/am-2022-8167The initial discovery, made by Opportunity rover, of iron-rich Martian blueberries was exciting because it suggested that water was needed for their formation, and, therefore, water was present on Mars. Other workers contended that water was not needed because the blueberries could have formed by aerial condensation of mineral clouds related to bolide impact. There is ample evidence for the presence of water on Mars, even without the blueberry evidence. However, D.D. Eberl shows in this contribution that the size distributions of the blueberries have a more detailed story to tell. Not only do the shapes of their PSDs indicate the presence of water, but they also indicate initial relative solution concentrations, and they may reveal relict concentration gradients in regional groundwater systems. More measurements of blueberry PSDs from existing rover photos may yield further information about hydrologic systems in this remote and exotic environment.
American Mineralogist Volume 106
December 2021
Contrasting magma compositions between Cu and Au mineralized granodiorite intrusions in the Tongling ore district in South China using apatite chemical composition and Sr-Nd isotopes
https://doi.org/10.2138/am-2021-7497Pan et al. demonstrated that apatite is a good petrogenetic and metallogenic indicator. The compositions of apatite from Tongling region suggest that the Au-mineralized magma is less oxidized and has higher S contents and Cl/F ratios than the Cu-mineralized magma due to the more mantle-derived components in the former. Dramatic variation of apatite Cu content indicates that extremely high Cu concentration in magma may not be an essential requirement to form a porphyry and skarn Cu deposit.
Halogen heterogeneity in the subcontinental lithospheric mantle revealed by I/Br ratios in kimberlites and their mantle xenoliths from South Africa, Greenland, China, Siberia, Canada, and Brazil
https://doi.org/10.2138/am-2021-7332Toyama et al. report new halogen data of rocks derived from the subcontinental lithospheric mantle (SCLM) of distinct locations worldwide. This work enables tracing compositional variabilities and similarities on a global scale. The authors distinguish two groups of compositions, defined by high and low I/Br ratios. This observation is interpreted to mirror halogen heterogeneities within the SCLM derived from subducted serpentinites and altered oceanic crust after discarding several competing arguments, e.g., contributions of crustal xenoliths to the host magma, dissolution of major halogen carrier minerals during alteration, degassing of halogens during magma emplacement.
Mineralogy of the 2019 Aguas Zarcas (CM2) carbonaceous chondrite meteorite fall
https://doi.org/10.2138/am-2021-7815The 2019 Aguas Zarcas CM2 meteorite fall is the most significant carbonaceous chondrite CM2 fall since Murchison in 1969. Samples collected immediately following the fall and studied here by Garvie provide the rare opportunity to analyze the bulk mineralogy of a CM2 meteorite largely free of terrestrial contamination. Analysis of this meteorite provides the basis for understanding the mineralogy of samples returned from hydrated, carbonaceous asteroids, including Bennu and Ryugu.
Keplerite, Ca9(Ca0.5□0.5)Mg(PO4)7, a new meteoritic and terrestrial phosphate isomorphous with merrillite, Ca9NaMg(PO4)7
https://doi.org/10.2138/am-2021-7834Keplerite is a Ca-dominant counterpart of the most abundant meteoritic phosphate, merrillite. The solid solutions merrillite-keplerite is a main reservoir of phosphate phosphorus in the solar system. The Na-number measure, 100xNa/(Na+Ca), is herein proposed by Britvin et al. for the characterization of the minerals intermediate between merrillite and keplerite.
Thermodynamic, elastic and vibrational (IR/Raman) behavior of mixed type-AB carbonated hydroxylapatite by density functional theory
https://doi.org/10.2138/am-2021-7826The work by Ulian et al. extends the overall knowledge of thermodynamic, elastic, and vibrational properties of type-AB carbonated apatite. Carbonated hydroxylapatite is the main mineral phase in bone and dental tissues and in most phosphate-based biomaterials. The knowledge of its thermodynamic, elastic, and vibrational properties is important to understand the basics of this mineral and to develop new synthetic analogs for a wide range of biomedical applications.
Internal stress-induced recrystallization and diffusive transport in CaTiO3-PbTiO3 solid solutions: A new transport mechanism in geomaterials and its implications for thermobarometry, geochronology and geospeedometry
https://doi.org/10.2138/am-2021-7588Beyer and Chakroborty conducted a series of high-temperature experiments to study the incorporation of Pb in natural CaTiO3 single crystals. They observed a partial replacement of the original CaTiO3 crystal by a polycrystalline rim composed of (Ca,Pb)TiO3 microcrystals. The shape and chemical zonation of the rim suggest that diffusion and dissolution-precipitation processes work in parallel. The newly discovered process has a distinctly smaller activation energy than the diffusion alone. This has consequences for geothermobarometry, geochronology, and geospeedometry.
Experimental determination of carbon diffusion in liquid iron at high pressure
https://doi.org/10.2138/am-2021-7644Carbon dissolves readily in liquid iron, and most of Earth's carbon is expected to be in the core. The behavior of carbon in liquid iron at high pressure (P) and temperature (T) is therefore essential for understanding the formation and evolution of planetary cores. Here, Rebaza et al. measured the diffusion rates of carbon in liquid iron at high P-T in the laboratory. Their data show that carbon transport is insensitive to P over the conditions studied but may be affected by the presence of nickel.
Reduction of structural Fe(III) in nontronite by humic substances in the absence and presence of Shewanella putrefaciens and accompanying secondary mineralization
https://doi.org/10.2138/am-2021-7828Zuo et al. demonstrated that humic substances, a common type of soil organic matter, can serve as electron donor, shuttle, and even acceptor to mediate abiotic and biotic reduction of structural Fe(III) in a clay mineral nontronite. Different types of humic substances show dramatically different effects in Fe(III) reduction and mineral transformations due to their intrinsic differences in electrochemical and molecular compositions. Mineralogical transformations accompany such redox reactions, forming high-temperature minerals under ambient low-temperature conditions. This study highlights a strong need for understanding the interaction between soil organic matter and clay minerals under environmental relevant conditions.
Trace element segregation to dislocation loops in experimentally heated zircon
https://doi.org/10.2138/am-2021-7654Peterman et al. conducted nanoscale analysis of an Archean zircon that was experimentally heated to 1450 C for 24 h and found trace-element-enriched crystal defects (5 to 25 nm) that replicate features observed in natural zircon. The formation and distribution of these features are controlled by crystallography, effective radiation dose, and the rate of heating and structural recovery. The entrapment of trace elements in nanoscale defects may have practical minerals engineering applications, such as storage of significant nuclear elements.
Tin isotopes via fs-LA-MC-ICP-MS analysis record complex fluid evolution in single cassiterite crystals
https://doi.org/10.2138/am-2021-7558Liu et al. present cathodoluminescence imaging, trace element, and in-situ Sn isotope compositions of two cassiterite crystals from an early and a relatively late stage of ore formation of the Xiling vein-style Sn deposit, China. Their results indicate compositional heterogeneity at the sub-millimeter scale in terms of trace elements and tin isotope composition, which reveal a more complex fluid evolution than expected by analytical bulk mineral techniques. The features of the tin isotope fractionation can be attributed to fractional crystallization of cassiterite, both in relatively closed-system and open-system environments.
Tracking dynamic hydrothermal process: Textures, in-situ Sr-Nd isotopes and trace element analysis of scheelite from the Yangjiashan vein-type W deposit, South China
https://doi.org/10.2138/am-2021-7677Li et al. present characterization of textures, in-situ Sr-Nd isotopes, and trace element analysis of scheelite from the Yangjiashan vein-type W deposit, China. The results suggest that fluid compositions, different substitution mechanisms, and primary-secondary processes all contribute to the variation in REE fractionation patterns. These in-situ data emphasize that Sr and Nd are both mobile during hydrothermal process, indicating that conventional bulk powder analysis of Sr and Nd isotope compositions may be problematic for texturally complicated scheelite. Careful analysis enables the source of ore-forming metals to be identified and the dynamics of the hydrothermal process constrained.
Oxygen isotope evidence for input of magmatic fluids and precipitation of Au-Ag-tellurides in an otherwise ordinary adularia-sericite epithermal system in NE China
https://doi.org/10.2138/am-2021-7825Gao et al. present evidence for the involvement of magmatic fluids in the formation of Te-rich epithermal Au-Ag deposits using mineral textures and CL images to guide high spatial resolution SIMS (10-micrometer scale) oxygen isotope analysis and fluid inclusion microthermometric measurements on the same generation of banded quartz. The results show that magmatic fluid inputs are critical to formation of Te-rich adularia-sericite epithermal Au-Ag deposits. The correlation of fluid inclusion temperatures with in situ analysis of oxygen isotope ratios by SIMS is novel and has important advantages.
Thalliomelane, TlMn4+7.5Cu2+0.5O16, a new member of the coronadite group from the preglacial oxidation zone at Zalas, southern Poland
https://doi.org/10.2138/am-2021-7577Gołębiowska et al. present data on a new mineral thalliomelane, TlMn4+7.5Cu2+0.5O16, a member of the coronadite group, which is interesting not only due to exotic composition, but also probable conditions of its formation. It is the first Mn oxide containing abundant Tl, and along with recent studies of Wick et al. (2019) on sorption of Tl onto manganese oxides, may document the replacement of other large tunnel cations (K, perhaps Ba) in minerals of the coronadite group by Tl+ in supergene geological environments.
New Mineral Names
https://doi.org/10.2138/am-2021-NMN1061225November 2021
First Principles Molecular Dynamics maps out complete mineral surface acidity landscape
https://doi.org/10.2138/am-2022-7991Highlights and Breakthroughs
Sulfur solubility in the Earth magma ocean—testing the hypothesis of the "Hadean matte"
https://doi.org/10.2138/am-2022-8281Highlights and Breakthroughs
Spectroscopic evidence for the Fe3+ spin transition in iron-bearing δ-AlOOH at high pressure
https://doi.org/10.2138/am-2021-7541Su et al. performed high-pressure X-ray emission (XES) experiments on δ-(Al0.85Fe0.15) up to 53 GPa using silicone oil as a PTM in a DAC. Laser Raman spectroscopy experiments were conducted on the δ-(Al0.85Fe0.15) OOH and δ-(Al0.52Fe0.48) OOHsamples up to 57 and 62 GPa, respectively, using neon as a PTM. Our XES spectra of δ-(Al0.85Fe0.15)OOH showed a broadening spin crossover approximately from 30 to 42 GPa. By contrast, our laser Raman spectra of δ-(Al0.85Fe0.15)OOH and δ-(Al0.52Fe0.48)OOH illustrated a relatively sharp change at 32−37 and 41−45 GPa, respectively, across the spin transition. Together with literature data, we found that the spin transition pressure of δ-(Al,Fe)OOH slightly increases with increasing iron content. These results shed new insights into the knowledge of iron effects on the spin transition pressure and vibration properties of δ-(Al,Fe)OOH. The presence of iron in δ-AlOOH can substantially influence its high-pressure behavior and stability at the deep mantle conditions and play an important role in the deep-water cycle.
Quantitative WDS compositional mapping using the electron microprobe
https://doi.org/10.2138/am-2021-7739Elemental zoning in minerals contains critical information for the interpretation of rocks and is commonly investigated by EPMA X-ray mapping. However, raw X-ray intensity maps can contain analytical artifacts from the effects of matrix effects, interferences, beam damage, and continuum production. Donovan et al. present a rigorous quantification protocol for the treatment of WDS element X-ray maps to produce accurate results and discusses the impact of these corrections relative to the raw data.
Interfacial structures and acidity constants of goethite from first principles Molecular Dynamics simulations
https://doi.org/10.2138/am-2021-7835Goethite plays important roles in numerous geochemical processes, and its interface chemistry has attracted significant attention. pKa values are central to surface reactivity of goethite, but they are still lacking up to now. To fill this gap the advanced FPMD technique was applied by Zhang et al. to study the major surfaces. The derived interfacial structures and pKa values can be directly used to investigate a wide range of processes including surface complexation reactions. The findings form a microscopic basis for understanding the processes on goethite interfaces.
Sound velocities of iron-nickel (Fe90Ni10) alloy up to 8 GPa and 773 K: The effect of nickel on the elastic properties of bcc-iron at high P-T
https://doi.org/10.2138/am-2021-7716In this paper, Wang et al. report the acoustic velocity measurements of bcc-Fe90Ni10 alloy up to 8 GPa and 773 K in a multi-anvil apparatus. Fe-Ni alloys are considered as the major constituent of Earth's and other planetary cores. Understanding the elastic properties of Fe-Ni alloys at high pressure and high-temperature conditions is essential to compare with seismological observations of the core and then place constraints on its composition. The study reports the elastic moduli (KS and G) and their pressure derivatives and, for the first time, provides the temperature dependences of the elastic bulk and shear moduli of bcc-Fe90Ni10. These findings should motivate future acoustic measurements of various Fe-Ni-light elements alloys and compounds at simultaneous high P-T conditions to provide a more comprehensive understanding of the composition and thermal structure of Earth's and planetary cores.
Formation of metallic-Cu-bearing mineral assemblages in type-3 ordinary and CO chondrites
https://doi.org/10.2138/am-2021-7689Both thermal and shock metamorphism could be responsible for the formation of metallic Cu in chondrites. Li et al. found that Ni-rich metal is the main Cu-carrier for CO3.2-3.7 chondrites, and most metallic Cu in CO3 chondrites were formed during thermal metamorphism. The growth of metallic Cu in ordinary chondrites seems to be related to shock heating and post-shock metamorphism. We also predict that some other groups of carbonaceous chondrites (e.g., CM and CI) are less likely to produce metallic Cu.
Behavior and origin of hydrogen defects in natural orthopyroxene during high-temperature processes
https://doi.org/10.2138/am-2021-7741To understand complex features of hydrogen defects in mantle orthopyroxene, Yang et al. carried out in situ high-temperature FTIR spectroscopic investigations on hydrogen defects in three natural orthopyroxenes. The study revealed what happens to hydrogen defects during high-temperature processes. These new data are enlightening for correctly relating features of hydrogen defects to geological processes.
Phase transitions in CaCO3 under hydrous and anhydrous conditions: Implications for the structural transformation of CaCO3 during subduction processes
https://doi.org/10.2138/am-2021-7575The detailed phase transition processes among CaCO3-I (calcite), CaCO3-II, III/IIIb, and aragonite at P-T conditions up to 2.5 GPa and 600 °C, in hydrous and anhydrous environments, were investigated by Yuan et al. Our results show that in contrast to the displacive CaCO3-I ↔ II transformation and the reconstructive CaCO3-II → III transformation which is divided into two sub-stages by the occurrence of intermediate CaCO3-IIIb, the reconstructive phase transition between CaCO3-I or II and aragonite requires greater activation energy and can only be achieved by progressive solid recrystallization under high P-T conditions (e.g., above 450 °C and 1.5 GPa), or alternatively via fluid-assisted dissolution-precipitation under relatively lower temperatures (e.g., 100-300 °C), depending upon the presence of aqueous fluids and the heating rate of the system. Considering the high P-T condition, low heating rate, and considerable amounts of aqueous fluids being generated during the subduction of hydrous oceanic crust, the CaCO3-I/II →aragonite transformation via the dissolution-precipitation process should commonly occur, and that the calcite aragonite →CaCO3-VII →post aragonite should be the predominant phase transition sequence during transport of CaCO3 into the deep earth through subduction processes.
Strain-induced partial serpentinization of germanate olivine with a small amount of water
https://doi.org/10.2138/am-2021-7735Sawa et al. demonstrate that strain-induced hydration of germanate olivine results in antigorite formation even in the presence of only small amounts of water. Thus, partly hydrated peridotite in the oceanic lithosphere can be formed under slight water infiltration due to high strain accumulated by the subduction.
The origin of trapiche-like inclusion patterns in quartz from Inner Mongolia, China
https://doi.org/10.2138/am-2021-7454Farfan et al. found that split crystal and two-dimensional nucleation-growth morphology preferentially trap clay inclusions to form trapiche-like patterns within quartz crystals from Inner Mongolia, China. This natural example of crystal growth hosts abundant inclusions complements materials science studies of synthetic split crystals on the nano-scale and suggests that split growth may be an indicator for highly supersaturated and/or turbid formation fluids.
Upper temperature limits of orogenic gold deposit formation: constraints from TiO2 polymorphs in the Dongyuan Au deposit, Jiangnan Orogen, China
https://doi.org/10.2138/am-2021-7754TiO2 minerals are commonly formed during the early stage of the Au mineralization and may therefore be used to fingerprint and investigate the formation of orogenic Au deposits. Raman spectroscopy, EPMA, and TEM analyses by Wu et al. show that orogenic Au deposits, such as the Dongyuan deposit, contain TiO2 polymorphs of anatase, anatase-rutile intergrowths, and rutile, which formed in the early stage of auriferous vein formation. The TiO2 polymorphs constrain the upper temperature limits of the Au vein formation to 450-550 °C, based on the thermodynamics of TiO2 polymorphs. Compared with TiO2 mineral compositions from magmatic-hydrothermal deposits (e.g., porphyry Cu/W deposits), they have distinct features in orogenic Au deposits, reflecting that ore-forming fluids stemmed from, or flowed through, metamorphic rocks. Furthermore, orogenic Au deposits widely distributed in the low-intermediate-grade metamorphic rocks stem from the deeply sourced fluids (e.g., from granulite-facies rocks of the middle to lower crust).
Formation of clinohumite spinel in dolomitic marbles from the Makrohar Granulite Belt, Central India: Evidence for Ti mobility during regional metamorphism
https://doi.org/10.2138/am-2021-7755Karmakar documents an occurrence of the rare mineral clinohumite in a suite of forsterite marbles from a part of the Central Indian Tectonic Zone. The humite group of minerals has a limited paragenesis wherein Ti- and F-bearing clinohumites have been reported from very few contact or regionally metamorphosed limestones, dolomites, and skarns, with only 4 reported occurrences in India. The clinohumite developed due to infiltration of T-i and F-bearing H2O-rich fluids, which in turn indicates that the presence of F in H2O-rich fluids must enhance Ti mobility. Ti is generally considered immobile during regional metamorphism and the constant-Ti frame of reference is widely used in most mass-flux calculations in fluid transport, to evaluate open-system behavior during metasomatism. As a result, documenting natural examples of mobility of Ti in metamorphic fluids is crucial to understanding the recycling of such commonly perceived nominally soluble elements and to understand HFSE recycling during metamorphic processes.
Carletonmooreite, Ni3Si, a new silicide from the Norton County, aubrite meteorite
https://doi.org/10.2138/am-2021-7645A new mineral named carletonmooreite, Ni3Si is described from the Norton County aubrite meteorite by Garvie et al.. This new mineral is named in honor of Carleton B. Moore, chemist and geologist, and founding director of the Center for Meteorite Studies, Arizona State University, for his many contributions to cosmochemistry and meteoritics.
Sulfur content at sulfide saturation of peridotitic melt at upper mantle conditions
https://doi.org/10.2138/am-2021-7649Blanchard et al. present new high-pressure and high-temperature experimental data that are used to understand the evolution of the solubility of sulfur in peridotitic melts. They show that pressure and temperature have an opposite effect: the higher the temperature, the higher the solubility of sulfur, and the higher the pressure, the lower the solubility of sulfur. This is coherent with previous studies using extrapolations from lower pressure and temperature experiments. We extrapolated our results to pressure and temperature conditions of the Earth's primitive magma ocean and show that FeS will exsolve from the molten silicate and can effectively be extracted to the core by a process that has been termed the "Hadean Matte".
Johnkoivulaite, a new mineral of the beryl group from the gem deposits of Mogok, Myanmar
https://doi.org/10.2138/am-2021-7785Palke et al. describe the new mineral johnkoivulaite from the legendary gem deposits of Mogok, Myanmar. Johnkoivulaite, Cs(Be2B)Mg2Si6O18, is the 7th member of the beryl group, a collection of silicate minerals in which the main identifying structural feature are Si6O18 rings which stack along the crystallographic c-axis. Johnkoivulaite is unusual in that magnesium and cesium are not typically enriched through the same geochemical processes. The chemistry of this mineral suggests potential interaction between pegmatites intruding gneisses and/or limestones.
Age determination of oriented rutile inclusions in sapphire and of moonstone from the Mogok metamorphic belt, Myanmar
https://doi.org/10.2138/am-2021-7487Shi et al. found that dating of acicular rutile inclusions in sapphire and furnace step-heating 40Ar/39Ar dating of moonstone (antiperthite) in syenites from the Mogok metamorphic Belt (MMB) yield ages of 13.43 0.92 and 13.55 0.08 Ma. Myanmar sapphire and moonstone formed at the same time, and the ages are the youngest published in the region. Origin of the oriented rutile inclusions in the sapphire prefers coprecipitation instead of exsolution. The ages provide insight into the complex histories and processes of magmatism and metamorphism of the MMB, the formation of gemstone species in this belt, and the collision between India and Asia.
X-ray computed microtomography of diamondiferous impact suevitic breccia and clast-poor melt rock from the Kara astrobleme (Pay-Khoy, Russia)
https://doi.org/10.2138/am-2021-7578Zubov et al. describe three types of suevitic breccias, clast-poor melt rock and a melt clast from suevite by X-ray computed microtomography. The effect of indirect data with "underestimation" or "overestimation" of the natural attenuation coefficients is described. The features of low contrast in X-rays of impact mineral products are discussed. The perspectives of X-ray computed microtomography for 3D petrography of impactites and contrast mineral phases distribution are proposed.
October 2021
Fingerprinting REE mineralization and hydrothermal remobilization history of the Carbonatite-Alkaline complexes, Central China: Constraints from in situ elemental and isotopic analyses of phosphate minerals
https://doi.org/10.2138/am-2021-7746Carbonatites and related alkaline rocks host most REE resources around the world. However, it is still controversial whether late-stage tectonothermal events are crucial for REE enrichment in carbonatite. Su et al. report in situ elemental and isotopic analyses on phosphates (monazite and apatite) from two mineralized carbonatite complexes, Central China. They suggest that most REE minerals were crystallized from primary carbonatite during the Silurian and only locally remobilized by Triassic regional metamorphism.
Formation of miarolitic-class, segregation-type pegmatites in the Taishanmiao batholith, China: The role of pressure fluctuations and volatile exsolution during pegmatite formation in a closed, isochoric system
https://doi.org/10.2138/am-2021-7637The processes that lead to the formation of pegmatitic textures (large crystals) are poorly understood. Most models require a rapid shift away from equilibrium conditions driven by either volatile-exsolution from the melt or significant undercooling of the melt. The resulting liquidus deficit leads to rapid crystal growth to produce a pegmatitic texture. Here, Yuan et al. describe an alternative method to drive the system from equilibrium during pegmatite formation in a closed, constant volume system. Rapid and significant pressure decrease leads to a liquidus deficit that drives crystallization as the system attempts to return to an equilibrium state. They propose that this pressure-driven mechanism is appropriate for miarolitic class, segregation style pegmatites that form in the waning stages of crystallization of moderate to deep magmas.
Fractal distribution of mineral species among the crystallographic point groups
https://doi.org/10.2138/am-2021-7698Hummer performed an analysis of the symmetry of thousands of mineral species and found that symmetry is distributed among minerals in a fractal pattern. Within each crystal system, the number of minerals belonging to each symmetry group scales as a power law function with respect to the order of the symmetry group, but the comparison between crystal systems follows the opposite trend. These results indicate that the incorporation of symmetry itself into crystalline materials follows a universal scaling law of the same sort as objects with fractal geometry.
The suppression of lone-pair stereoactivity in [Cu+(As3+O3)4] clusters in dixenite: A tribute to Paul B. Moore
https://doi.org/10.2138/am-2021-7719Hathorne and Hughes describe the crystal structure of dixenite, which is one of two minerals known to contain a metallic cluster in an oxide matrix. The mineral contains [Cu+(As3+O3)4] clusters in which four (As3+O3) groups link to a central Cu+ that occupies the positions normally taken by the stereoactive lone-pairs of electrons.
A Gruneisen tensor for rutile and its application to host-inclusion systems
https://doi.org/10.2138/am-2021-7618Musiyachenko et al. report the full phonon-mode Gruneisen tensors for the Raman active modes of rutile. They show that stiff inclusions in a softer host (e.g., rutile inclusions in garnet) exhibiting residual pressures are, in fact, multiphase inclusions that include a softer phase. They describe how to model the variation of elastic properties of such mixed-phase inclusions in order to predict their residual pressures.
Various antiphase domains in garnet-hosted omphacite in low-temperature eclogite: A FIB-TEM study on heterogeneous ordering processes
https://doi.org/10.2138/am-2021-7784The omphacite antiphase domains (APDs) are keys to understanding the mineral growth kinetics during eclogitization. In this open access paper, Fukushima et al. conducted a transmission electron microscopic (TEM) study combined with a focused ion beam technique (FIB) on omphacite inclusions within a prograde-zoned garnet. The FIB-TEM observation confirms two types of APD morphology: (1) equiaxed and (2) columnar. Size distributions of the equiaxed APDs indicate multiple nucleation events of the ordered phase. The columnar APDs aligned perpendicularly to garnet-omphacite interfaces suggest heterogeneous spatial distribution of syngenetic omphacite. These features demonstrate a potential to understand the kinetics of the cation ordering and mineral inclusion processes.
Characterization and assessment of the potential toxicity/pathogenicity of Russian commercial chrysotile
https://doi.org/10.2138/am-2021-7710Di Giuseppe et al. report the first complete characterization of a representative commercial chrysotile from Orenburg region, Russia. The results demonstrate that Russian chrysotile can be considered a potential health hazard. The toxicity/pathogenicity index of commercial Russian chrysotile is lower than that of amphibole asbestos but above the threshold limit for "safe" mineral fibers. It was found that the investigated representative sample is similar to the Italian Balangero chrysotile and does not contain any amphibole asbestos.
Graphite paradox in Baikal geyserite paleovalley, Russia
https://doi.org/10.2138/am-2021-7711Shumilova et al. describe unusual graphite mineralization in hydrothermalites of the Baikal geyserite paleovalley (Russia). They found that the co-existence of relict and newly formed graphite presents the observed "high temperature" paradox in Baikal travertines and geyserites "in situ" being the result of redeposition of host rocks metamorphic graphite within H2O-rich low-pressure low-temperature C-O-H fluid.
Establishing the P-T path of UHT granulites by geochemically distinguishing peritectic from retrograde garnet
https://doi.org/10.2138/am-2021-7681The P-T evolution (and particularly the prograde path segment) of ultrahigh-temperature (UHT) granulites is commonly ambiguous, hampering our understanding of deep crustal processes. Jiao et al. establish the P-T path by distinguishing garnet origin (peritectic or retrograde) based on the combined Ca, Ti, Zr, and Y+REE chemical signatures, coupled with the application of the Ti-in-Grt geothermometer and the Ca-in-Grt geobarometer and phase equilibria modeling, using the residual UHT granulites of the Khondalite Belt, North China Craton, as a test case. The integrated results allow formulation of an earlier, high-pressure, high-temperature garnet formation and a later abundant retrograde garnet formation during post-peak cooling. The authors conclude that change of Zr and other elements (e.g., Ti, Ca, Y, and Eu/Eu*) can well distinguish different garnet formation events in UHT granulites, which is critical for the P-T path establishment, and further sheds light on the cause of UHT metamorphism and the geodynamic evolution.
Dissolution-reprecipitation vs. solid-state diffusion in electrum: Examples from metamorphosed Au-bearing, volcanogenic massive sulfide (VMS) deposits
https://doi.org/10.2138/am-2021-7674Electrum is the predominant precious metal host in Au-bearing volcanogenic massive sulfide (VMS) deposits. Liu and Beaudoin present detailed textural and compositional data of electrum from the Ming (Canada) and Boliden (Sweden) Au-bearing VMS deposits. The analyses by EPMA and LA-ICP-MS confirm the chemical zoning in heterogeneous electrum grains. Electrum zoning formation is attributed to a complex interaction between fluid facilitated solid-state diffusion (SSD) within the grain and coupled dissolution and reprecipitation (CDR) reactions at the grain interface in response to changing fluid composition and extrinsic parameters. The authors suggest that metamorphic events probably provide the elevated temperature and deformation to allow pervasive fluids to remobilize trace metals in electrum in metamorphosed VMS deposits.
High-temperature and high-pressure Raman spectra of Fo89Fa11 and Fo58Fa42 olivines: Iron effect on thermodynamic properties
https://doi.org/10.2138/am-2021-7686The harmonic approximation traditionally plays a central role in modeling the thermodynamic properties of minerals, such as the heat capacities, equations of state, and equilibrium isotopic fractionation factors. Nevertheless, such approximation deviates from reality, especially at high temperatures. Then the intrinsic anharmonic correction becomes necessary and crucial to restore agreement between theoretical models and experiments at high temperatures. Liu et al. evaluate the Fe effect on the harmonic contributions to the thermodynamic properties of olivine, the most abundant mineral in the upper mantle, by Raman spectroscopy at high pressures and temperatures.
Mixing of cogenetic magmas in the Cretaceous Zhangzhou calc-alkaline granite from SE China recorded by in-situ apatite geochemistry
https://doi.org/10.2138/am-2021-7786Mixing of cogenetic magmas represents an important process in granite petrogenesis but is difficult to identify and is consequently often overlooked due to the absence of obvious isotopic distinctions between the mixed melts. Zhang et al. conducted in situ elemental and O isotope analyses on apatite from Cretaceous Zhangzhou calc-alkaline granite in southeast China. Integration of these data with micro-analyses on other minerals (plagioclase, zircon, and titanite) as well as whole-rock geochemistry allows the authors to decipher the mixing history of this granitic complex, which involved mixing between two cogenetic magma batches, with variable degrees of subsequent differentiation. This study demonstrates that apatite geochemistry is potentially a suitable monitor of complex magmatic evolution, including devolatilization and mixing of isotopically indistinguishable magmas.
Hydrothermal upgrading as an important tool for the REE mineralization in the Miaoya carbonatite-syenite complex, Central China
https://doi.org/10.2138/am-2021-7779Ma et al. conducted a combined textural and in situ chemical investigation on the REE mineralization in the ~430 Ma Miaoya carbonatite-syenite complex, central China. The results indicate that (1) REE-bearing fluorapatite and calcite were altered through a coupled dissolution-reprecipitation mechanism; (2) Fluids responsible for the REE remobilization were CO2-rich with medium temperature (227 to 340 C) and low salinity (1.42 to 8.82 wt%); and (3) Hydrothermal upgrading is an important tool for REE mineralization..
September 2021
Stable and transient isotopic trends in the crustal evolution of Zealandia Cordillera
https://doi.org/10.2138/am-2021-7626Schwartz et al. use zircon δ18O and Lu-Hf isotope analyses to explore the interplay between spatial and temporal magmatic signals in the Mesozoic Zealandia Cordillera. They find that Mesozoic pluton chemistry was controlled by both crustal/upper mantle architecture and a transient process involving a mantle-controlled flare-up event that involved mantle melts contaminated with recycled sediments. Their data demonstrate that spatial and temporal signals are intimately linked, and when evaluated together they provide important insights into the role of stable and transient arc magmatic trends in Cordilleran batholiths.
An evolutionary system of mineralogy, Part V: Aqueous and thermal alteration of planetesimals (~4565 to 4550 Ma)
https://doi.org/10.2138/am-2021-7760The evolutionary system of mineralogy classifies minerals based on their roles in planetary evolution. The first five parts of the system summarize all known minerals that emerged prior to the formation of planet Earth. Most of those minerals are preserved in the meteorites that still fall to Earth today. In Part V, by Hazen and Morrison, the focus is on the varied secondary minerals that formed in planetesimals more than 4.5 billion years ago through the often linked processes of aqueous alteration and thermal metamorphism. In this contribution Hazen and Morrison catalog 166 secondary meteorite minerals, including the earliest known appearances of species with the chemical elements Co, Ge, As, Nb, Ag, Sn, Te, Au, Hg, Pb, and Bi. Among the varied secondary meteorite minerals are the earliest known examples of halides, arsenides, tellurides, sulfates, carbonates, hydroxides, and a wide range of micas and clay minerals.
Cr2O3 in Corundum: Ultra-high contents under reducing conditions
https://doi.org/10.2138/am-2021-7680Griffin et al. describe a suite of corundum xenocrysts in Cretaceous, mafic, pyroclastic rocks on Mt Carmel, Israel, which includes rubies with the highest recorded contents of Cr2O3. Many coexist with K- and Na-beta alumina phases and native chromium, requiring extremely reducing conditions, indicating oxygen fugacities circa 5 log units below the Iron-Wustite buffer. Zoning patterns indicate a progressive decrease in oxygen fugacity during crystallization (Fig. 1), which is interpreted as tracking the interaction of oxide melts with mantle-derived methane and hydrogen near the crust-mantle boundary.
Plagioclase population dynamics and zoning in response to changes in temperature and pressure
https://doi.org/10.2138/am-2021-7491In this paper by Andrews, the numerical model SNGPlag tracks the numbers, sizes, morphologies, and compositional zoning of plagioclase crystals through time in response to changes in magmatic conditions. Low growth rates and dissolution result in significant fractions of time (>>50%) missing from the final crystal record. Growth of complexly zoned phenocrysts requires the addition of new magma. Crystal records are biased towards more recent intervals and periods of decreasing temperature. Crystallization (or dissolution) acts to return magmas to near-equilibrium crystal fractions within hundreds of days.
Limited channelized fluid infiltration in the Torres del Paine contact aureole
https://doi.org/10.2138/am-2021-7622This paper by Siron et al. describes the metamorphic evolution of the Torres del Paine contact aureole that experienced a limited amount of fluid infiltration from the crystallizing granite. This evolution was tracked using hydrogen and oxygen isotopes and Cl concentrations in biotite. The potential of these different tracers to track fluid-rock interactions is evaluated and highlights the sensitivity of hydrogen vs. oxygen isotopes for such use. Cl content in biotite is also a promising tool for such work.
Quantitative determination of the shock stage of L6 ordinary chondrites using X-ray diffraction
https://doi.org/10.2138/am-2021-7554The mean lattice strain and grain size factor for olivine and orthopyroxene of 14 L6 chondrites were determined by Imae and Kimura from analyses based on Williamson-Hall plots via X-ray diffraction. The combination of mean lattice strain with grain size factor enables the construction of a shock barometer.
A new method to rapidly and accurately assess the mechanical properties of geologically relevant materials
https://doi.org/10.2138/am-2021-7455This new method by Deng et al. will enable geological scientists and engineers to rapidly determine the elastic behavior of any rock or mineral in a simple manner with a robust statistical response. It will also facilitate the determination of elastic properties of rocks and minerals as a function of composition, structure, hydration, or other physical variables. This will undoubtedly impact the development of geologic-based deformation models through knowledge of the constituent material properties for validation and prediction accuracy.
Two-stage magmatism and tungsten mineralization in the Nanling Range, South China: Evidence from the Jurassic Helukou deposit
https://doi.org/10.2138/am-2021-7678In this study, Cao et al. report LA-ICP-MS zircon U-Pb and molybdenite Re-Os ages of the Helukou W deposit, which is a representative W deposit located in the Guposhan district, SW of Nanling Range, South China. This geochronological data aims to reveal persistent (Early to Jurassic) granitic magmatism and related W-Sn mineralization events in the Nanling region. In addition, the in-situ trace element and Sr isotopes compositions of scheelite from the skarn- and altered granite-type ores of the Helukou W deposit are reported, with the purpose of outlining the nature of the ore-forming fluids in the long-lived magmatic-hydrothermal system. The data reveals two stages of magmatism and tungsten mineralization in the Guposhan ore field during Early Jurassic (~180 Ma) and Middle Jurassic (~165 Ma). This 20 m.y. long-lived successive magmatism and mineralization in the Nanling Range will provide new insights into the magmatism, mineralization, and metallogenesis in this region.
Constraints on scheelite genesis at the Dabaoshan stratabound polymetallic deposit, South China
https://doi.org/10.2138/am-2021-7499Stratabound polymetallic deposits serve as one of the most important reserves of base metals in South China, but the genetic relationship between stratabound base metal mineralization and porphyry mineralization remains in dispute. Scheelite occurs as a characteristic mineral in many stratabound polymetallic deposits and in the adjacent porphyry system. In this paper, Su et al. use scheelite geochemistry, zircon, and hydrothermal apatite U-Pb dating to constrain the genesis of scheelite in stratabound orebodies at Dabaoshan. By integrating scheelite CL textures, trace element characteristics, and the new dating results, they demonstrate that ore-forming fluids responsible for scheelite formation in stratabound orebodies were derived from the Dabaoshan porphyry system. Furthermore, Jurassic porphyry mineralization at least partially contributed to Cu mineralization in the stratabound orebodies. This work suggests that scheelite geochemistry, combined with geochronology studies, can contribute to our understanding of stratabound orebody genesis.
Crystal chemistry of schreibersite, (Fe,Ni)3P
https://doi.org/10.2138/am-2021-7766Schreibersite is the most common phosphide mineral and the main carrier of reduced phosphorus in the celestial bodies. The present paper by Britvin et al. reports on previously obscured trends in Fe and Ni distribution across schreibersite structures, which are directly related to crystallization histories of metal-rich planetary interiors.
Elastic geobarometry: how to work with residual inclusion strains and pressures
https://doi.org/10.2138/am-2021-7928Gilio et al. discuss the difference between various approaches to retrieve the residual pressures and residual strains from Raman measurements of inclusions. They provide general guidelines on how to deal with inclusion strain, measurements, inclusion pressure, and their uncertainties.
Controls on tetrahedral Fe(III) abundance in 2:1 phyllosilicates - - Discussion
https://doi.org/10.2138/am-2021-7865Controls on tetrahedral Fe(III) abundance in 2:1 phyllosilicates - Reply
https://doi.org/10.2138/am-2021-8107New Mineral Names
https://doi.org/10.2138/am-2021-NMN106921Book Review: Geochronology and Thermochronology
https://doi.org/10.2138/am-2021-B106922August 2021
Highlights and Breakthroughs: Crustal melting: deep, hot, and salty
https://doi.org/10.2138/am-2022-8108Lamadrid and Steele-MacInnis provide perspective on the study by Ferrero et al.: High pressure, halogen-bearing melt preserved in ultra-high temperature felsic granulites of the Central Maine Terrane, Connecticut (U.S.A.).
Petrogenetic and tectonic interpretation of strongly peraluminous granitic rocks and their significance in the Archean rock record
https://doi.org/10.2138/am-2022-8001Strongly peraluminous granitic rocks first become abundant in the rock record in the Neoarchean. In this study by Frost and Da Prat (MSA Presidential Address by Carol Frost), six suites of Neoarchean strongly peraluminous granitic rocks are described from the Wyoming province. These are shown to form in both subduction and collisional environments from a variety of sources, including hornblende-plagioclase rocks, biotite-bearing gneisses, and two-mica metapelitic rocks. The appearance of strongly peraluminous granitic rocks in the rock record signals the development of strong, thick felsic continental crust and the formation of the first supercontinents.
Partial melting and P-T evolution of eclogite-facies metapelitic migmatites from the Egere Terrane (Central Hoggar, South Algeria)
https://doi.org/10.2138/am-2021-7342This contribution by Arab et al. improves our understanding of high-pressure metapelites from Central Hoggar, provides very advanced P-T modeling, and shows the first description of evidence of partial melting phenomena from Hoggar. This study resolves numerous problems regarding the metamorphic evolution of the Egere area and their geodynamic implication within the Tuareg belt.
High pressure, halogen-bearing melt preserved in ultra-high temperature felsic granulites of the Central Maine Terrane, Connecticut (US)
https://doi.org/10.2138/am-2021-7690Ferrero et al. performed micro-petrology experiments on preserved droplets of melt in garnet from the ultrahigh temperature felsic granulites of the Central Maine Terrain (Connecticut, U.S.A.). The results reveal a history of metamorphism and melting at a depth consistent with the orogenic roots, ~70 km, twice the previous estimates for these rocks. The high-pressure melt here preserved is peculiar: it has an uncommonly high mafic component, coupled with significant amounts of carbon dioxides and halogens (chlorine and fluorine). Whereas the first phenomenon results from the extreme melting temperatures, the halogens abundance likely relates to the presence of brines (highly saline liquids) at depth during melting. The latter is a direct evidence that brines may play a role in promoting melt production when the crust attains temperature in excess of 1000 °C, where melt production is actually expected to be hindered by the extremely dehydrated character of the rocks.
Targeting Jarosite -- Clay-mineral mixtures for Mars exploration
https://doi.org/10.2138/am-2021-7415Hinman et al. compared, with multiple techniques, the detectability and abundance of organic matter associated with clay minerals and the sulfate salts, alunite, and jarosite, from a hydrothermal area in Yellowstone National Park, Wyoming, U.S.A. Spectroscopy and diffractometry produced comparable mineral identifications in sample matrices. The matrix material (clay minerals or sulfate salts) had no effect on the detection of organic matter by Raman spectroscopy. However, mineral matrix composition had a significant effect on the detectability of organic matter by laser-desorption, Fourier transform mass spectrometry (LD FTMS). Indeed, LD FTMS detected diverse organic compounds in samples containing jarosite, more so than in samples dominated by aluminosilicate minerals, despite higher organic content in the later matrix. Further, organic compounds were detected more frequently in jarosite samples than in aluminosilicate samples. Consequently, jarosite-bearing sites on Mars could be potential targets for life detection, especially if associated with clay minerals.
Zirconolite from Larvik Plutonic Complex, Norway, its relationship to stefanweissite and nöggerathite, and contribution to the improvement of zirconolite end-member systematics
https://doi.org/10.2138/am-2021-7510Haifler et al. performed crystal-chemical characterization of a complex zirconolite from Larvik Plutonic Complex, Norway. A concept called "edgemembers" was introduced to quantitatively express compositional variation of a complex solid solution. Moreover, a modified scheme of zirconolite end-member set and redefinition of a composition space was suggested.
Nanomineralogy of hydrothermal magnetite from Acropolis, South Australia: Genetic implications for iron-oxide copper gold mineralization
https://doi.org/10.2138/am-2021-7557The Acropolis prospect is a magnetite dominant iron-oxide copper gold (IOCG) prospect in the Olympic Dam district (South Australia). Verdugo-Ihl et al. used complementary microbeam techniques (HAADF STEM, XMg-in-magnetite thermometry, and nanothermobarometry using ilmenite-magnetite pairs) to characterize titanomagnetite from veins hosted in volcanic rocks and Ti-poor magnetite from a granite body stratigraphically higher in the sequence. Hydrothermal titanomagnetite from Acropolis is comparable with magmatic magnetite in granites across the district and should typify early alkali-calcic alteration. Open-fracture circulation, inhibiting additional supply of Si, Ca, K, etc., during magnetite precipitation, prohibits formation of silician magnetite hosting calc-silicate NPs, as known from IOCG systems characterized by rock-buffered alteration of host lithologies. Obliteration of trellis textures during subsequent overprinting could explain the scarcity of this type of hydrothermal magnetite in other IOCG systems.
Effect of magnesium on monohydrocalcite formation and unit cell parameters
https://doi.org/10.2138/am-2021-7673Vereshchagin et al. report the phase diagrams for monohydrocalcite (MHC), calcite, aragonite, and dypingite precipitation, as a function of pH and Mg/Ca ratio in solution. They demonstrate that elevated magnesium content and low temperature are favorable for MHC formation. On the basis of changes in the unit cell parameters, the possibility of the significant incorporation of magnesium into the crystal structure of MHC has been unequivocally proven. The increase in Mg in MHC is accompanied by an increase in water content, which leads to multidirectional changes in a and c cell parameters.
Formation pathway of norsethite dominated by solution chemistry under ambient conditions
https://doi.org/10.2138/am-2021-7348"Dolomite problem" is a long-standing puzzle in geology. Zhang et al. investigated low-temperature formation of norsethite &91;BaMg(CO3)2&93; as a dolomite-analog under different solution chemistry conditions. The results reveal that the initial Mg/Ba ratio in solution is a crucial factor controlling the formation pathway of norsethite. At low Mg/Ba ratios, a multistep pathway occurs, i.e., a precursor witherite first forms, followed by norsethite precipitation and transformation from witherite to norsethite. In contrast, at Mg/Ba ratio > 20, norsethite can be directly precipitated from aqueous solution. This is the first report on direct precipitation of dolomite analogs under ambient conditions, and the findings provide new insights into the formation pathways of dolomite analogs and thus low-temperature dolomite.
A model for the kinetics of high-temperature reactions between polydisperse volcanic ash and SO2 gas
https://doi.org/10.2138/am-2021-7691Scientists have collected volcanic ash from eruptions and noticed that tiny salt crystals exist on their surfaces. Wadsworth et al. use experiments and mathematics to work out how quickly those salt crystals grow. Because the salts contain sulfur, they propose that some of the sulfur that would have ended up in the atmosphere actually ends up on ash surfaces. They further propose that these equations could tell us just how much sulfur is removed from the atmosphere in this process.
Redox control and measurement in low-temperature hydrothermal experiments
https://doi.org/10.2138/am-2021-7687Fang and Chou propose a new oxygen buffer technique for hydrothermal experiments carried out at temperatures below 450 °C and demonstrate its success in redox control and monitoring. Their work extends the low-temperature limit of previously well-developed redox control techniques to, at least, 200 °C, and it has promising prospects of application in low-temperature hydrothermal experiments.
Heat capacity and thermodynamic functions of partially dehydrated sodium and zinc zeolite A (LTA)
https://doi.org/10.2138/am-2021-7726Dickson et al. offer a thermodynamic perspective into cation exchange, water sorption, and sorbate-induced structural transformations in zeolites. Heat capacities and thermodynamic functions are reported for industrially important zeolite A and for the ion-exchanged version zinc zeolite A. In addition, zeolitic water in sodium zeolite A is compared with that in other zeolites. Inflections common to all the zeolitic water heat capacities suggest that the hydration-influenced framework flexibility that has been found in zeolite A may be present in other zeolites as well.
P-V-T measurements of Fe3C to 117 GPa and 2100 K
https://doi.org/10.2138/am-2021-7581McGuire et al. measured the unit-cell volume of an iron carbide, Fe3C, by X-ray diffraction at simultaneous extreme pressure and temperature conditions relevant to the deep interior of the Earth. These measurements are used to construct a thermodynamic model of Fe3C for the Earth's core. They find that existing measurements support two possible scenarios for the Earth's core in the Fe-C system: (1) Fe3C is the stable end-member at inner core conditions but cannot explain the inner core density; and (2) Fe7C3 is stable and may explain the inner core density at relatively low temperatures.
New Mineral Names
https://doi.org/10.2138/am-2021-NMN106818Erratum
https://doi.org/10.2138/am-2021-E106817Book Review
https://doi.org/10.2138/am-2021-B106819July 2021
Tourmaline composition and boron isotope signature as a tracer of magmatic-hydrothermal processes
https://doi.org/10.2138/am-2021-7495Qiu et al. in their paper make three key points. They found that tourmalines crystallized from magmatic-hydrothermal fluids with different oxygen fugacities and salinities; that degassing during fluid evolution leads to significant B isotope fractionation and increases oxygen fugacity; and that tourmaline composition and the boron isotope signature can be tracers of magmatic and hydrothermal processes, as well as used to distinguish mineralized from barren host-granites.
Deformation and strength of mantle relevant garnets: Implications for the subduction of basaltic-rich crust
https://doi.org/10.2138/am-2021-7587Garnet is an abundant mineral in the upper mantle, and there have been no previous measurements of both its room-temperature strength and deformation mechanism. Vennari et al. deformed three compositions of garnet between two diamonds and probed its deformation with X-rays perpendicular to the compression axis. These measurements allowed them to determine garnet's resistance to plastic deformation and show that its strength is similar to other mantle phases. Vennari et al. also provide a modeling analysis to explore the deformation mechanism on an atomistic scale when axial stress is applied.
Ultra-reduced phases in ophiolites cannot come from Earth's mantle
https://doi.org/10.2138/am-2021-7612It is claimed that ophiolites carry highly reduced minerals and that the source regions of ophiolites in the Earth's mantle are located in the transition zone or lower mantle. By showing experimentally that highly reduced phases such as moissanite (SiC) are unstable in a FeO-bearing mantle, Ballhaus et al. refute this hypothesis. They caution that accessory diamond and ultra-reduced phases may not provide sufficient evidence to question existing models of ophiolite genesis.
Olivine from aillikites in the Tarim large igneous province as a window into mantle metasomatism and multi-stage magma evolution
https://doi.org/10.2138/am-2021-7521Wang et al. report the first aillikite in China. Phlogopite- and carbonate-rich metasomatic veins are involved in the source of the aillikite magma. Phenocrystic olivines in the aillikite are cognate cumulates formed by multi-stage crystallization at temperatures of 906-1136 degrees C.
Precise determination of the effect of temperature on the density of solid and liquid iron, nickel, and tin
https://doi.org/10.2138/am-2021-7509Kamiya et al. precisely measured the density of solid Sn, Ni, Fe, and liquid Fe using a high-temperature furnace. The density of solid Sn shows a drastic decrease near melting temperature (Tm). By contrast, the densities of solid Ni and Fe decrease linearly with increasing temperature up to Tm without any drastic decrease near Tm. The thermal expansion of liquid Fe was also determined to depend on temperature. The results are important for understanding the elastic behavior of solid and liquid metals at high temperatures.
Timescales of crystal mush mobilization in a volcanic system based on olivine diffusion chronometry
https://doi.org/10.2138/am-2021-7670Caracciolo et al. applied diffusion chronometry to olivine crystals erupted at different ages from a single volcanic system. They carried out a chemical characterization of olivine crystals coupled with a detailed Fe-Mg, Mn, and Ni diffusion study from temporally diverse (sub-glacial to historical time) eruptive units to retrieve timescales of crystal mush disaggregation in the volcanic system in Iceland. One important implication of their study is that magma replenishment and mush disaggregation events, possibly triggered by rifting-assisted processes, do not result in imminent eruption. In addition, only a few data exist on timescales of magmatic processes in mid-Ocean Ridge settings, and this is the first detailed study of the temporal variation of crystal-mush-to-eruption timescales in any volcanic system in any magmatic setting.
Chemical reactions in the Fe2SiO4-D2 system with a variable deuterium content under a pressure of 7.5 GPa
https://doi.org/10.2138/am-2021-7697The reactions examined in the paper by Efimchenko et al. are expected to occur under natural conditions in the interiors of the icy satellites and lead to the removal of fayalite from rocks and minerals due to its dissolution in hydrogen fluid or replacement by iron, ferrosilite, or silica depending on the molar ratio H2/Fa.
High-pressure syntheses and crystal structure analyses of a new low-density CaFe2O4-related and CaTi2O4-type MgAl2O4
https://doi.org/10.2138/am-2021-7619The high-pressure polymorphs of MgAl2O4 are considered important and abundant components that are stable under lower-mantle conditions in alumina-rich rocks, such as basalt, upper continental crust, and sediment. In this study, Ishii et al. synthesized single crystals of CaTi2O4 (CT)-type, CaFe2O4 CF-type, and a novel MgAl2O4 phases at 27 GPa and 2500 °C by conventional multi-anvil technique. They also synthesized CT-type MgAl2O4 at 45 GPa and 1727 °C using an advanced multi-anvil technique that they developed. Their single-crystal X-ray diffraction measurements showed that the novel phase has a completely new structure with a smaller density than the ambient pressure phase of spinel, despite its high-pressure synthesis, indicating that this phase is a back-transformed phase from a high-pressure phase during the recovery. This finding solves the crystal structure of a previously unknown phase. CT-type MgAl2O4 has been considered to be stable at more than 40 GPa. Ishii et al.’s finding that CT-type phase is synthesized even at 27 GPa indicates that the CT phase in natural-rock compositions may be stable in wide lower mantle conditions, especially upwelling hot plume conditions. They also suggest that the novel phase can be found in shocked meteorites and can be used as a shock indicator.
Phase diagram and thermal expansion of orthopyroxene-, clinopyroxene-, and ilmenite-structured MgGeO3
https://doi.org/10.2138/am-2021-7685CCBYDespite the ubiquity of studies of low-pressure analogs for Earth-forming phases, these systems are often incompletely characterized. Frequently, parts of the system remain unmeasured, but an analogue is only as good as its characterization. Here, Hunt et al., in their open access paper, complete the characterization of the lowest pressure phases of MgGeO3 and reconcile the disagreement between previous measurements. The reconciliation points to important differences between MgSiO3 and its low-pressure analog MgGeO3.
Mass transfer associated with chloritization in the hydrothermal alteration process of granitic pluton
https://doi.org/10.2138/am-2020-7353This study by Yuguchi et al. focuses on the petrography and mineral chemistry of hornblende chloritization, K-feldspar chloritization, and fracture-filling chlorite, in order to better understand mass transfer among the reactants, products, and hydrothermal fluids during chloritization. A combination of this study and Yuguchi et al. (2015) dealing with biotite chloritization reveals the characteristics of mass transfer in the overall chloritization process.
Non-linear effects of hydration on high-pressure sound velocities of rhyolitic glasses
https://doi.org/10.2138/am-2021-7597Gu et al. measured sound velocities in hydrous rhyolitic glasses containing up to ~6 wt% H2Ot at pressures to 3 GPa. The results show that hydration has a non-linear effect on sound velocities at high pressure, in contrast to the linear effects of hydration previously observed at ambient pressure. These results imply that the different water species, OH- and H2Om, play unique roles in altering sound velocities in rhyolitic glasses. The non-linear effects of hydration found here provide some insight into the behavior of hydrous silicate melts in felsic magma chambers at depth.
Crystal chemistry and high-temperature vibrational spectra of humite and norbergite: fluorine and titanium in humite-group minerals
https://doi.org/10.2138/am-2021-7538The humite-group minerals on the brucite-olivine join may be important carriers of H2O into the Earth’s deep interior, as well as water reservoirs in subduction zones and the upper mantle in extremely hydrous compositions. In this study, Liu et al. carried out single-crystal X-ray diffraction and used in-situ high-temperature Raman and Fourier transform infrared (FTIR) measurements on natural humite and norbergite samples to study the effects of F and Ti incorporation on the hydrogen behavior in these hydrous minerals. The OH-stretching modes above 3450 cm-1 are affected by the neighboring H-H repulsion, while the ones below 3450 cm-1 could be explained by F/Ti substitutions, both of which could relieve the H-H repulsion effect. The Raman-active OH bands below 3450 cm-1 are determined by Ti-substitution, while the IR-active ones are contributed by both F and Ti substitutions. Both the structure refinements and high-temperature vibrational spectra on natural humite samples could provide microscopic insight for understanding F and Ti effects on the stability field of the hydrous minerals (such as humite-group minerals) in subducting slabs.
Exomorphism of Jacobsite Precipitates in Bixbyite Single Crystals from the Thomas Range in Utah
https://doi.org/10.2138/am-2021-7715CCBYNCNDPeter et al. investigated single crystals of bixbyite via transmission electron microscopy (TEM), revealing a lamellar 3D network consisting of the tetragonal manganese-silicate braunite. Additionally, nanometer-sized precipitates were found and identified as jacobsite, a cubic iron-manganese-spinel, not previously known to occur in the Thomas Range. The presence of spinel implies higher formation temperatures than described in the literature, possibly acting as a novel geo-thermometer. Interestingly, TEM observations indicate that the shape of the crystallites is exomorphously determined by the braunite lamellae, leading to their unusual cuboid morphology, which is, to the best of our knowledge, the first report on exomorphism of a naturally formed spinel.
Ferropyrosmalite-bearing fluid inclusions in the North Patagonian Andes metasedimentary basement, Argentina: A record of regional metasomatism
https://doi.org/10.2138/am-2021-7525Sosa et al. present new cathodoluminescence, Raman spectroscopy, and microthermometric data of fluid inclusions in metamorphic quartz segregations from metasedimentary rocks of northwestern Patagonia (Argentina). This information is combined with heating experiments of fluid inclusions, which allowed studying equilibrium conditions of ferropyrosmalite with clino-amphibole, providing novel insights into their genetic conditions and mineral equilibria. Implications for fluid evolution during regional metasomatic processes are included in this work.
Memorial of Alden Bliss Carpenter (1936-2019)
https://doi.org/10.2138/am-2021-1009New Mineral Names
https://doi.org/10.2138/am-2021-NMN106714June 2021
Cation partitioning among crystallographic sites based on bond-length constraints in tourmaline-supergroup minerals
https://doi.org/10.2138/am-2021-7804Bacik and Fridrichova determined the positions of major, minor, and trace elements in the tourmaline structure based on the calculated bond lengths. Major elements can be divided into the structural sites based on the chemical and structural data, but it is far more complicated for minor and trace elements due to their low contents. Predictions can be made based on bond-length constraints for each site. Theoretical bond-length calculation from ideal bond valences for each ion and coordination can predict ion site preference in the structure.
Magnesio-lucchesiite, CaMg3Al6(Si6O18)(BO3)3(OH)3O, a new species of the tourmaline supergroup
https://doi.org/10.2138/am-2021-7496Scribner et al. describe magnesio-lucchesiite, a new species of the tourmaline supergroup. The mineral originates from the replacement of mafic minerals by boron-rich metasomatic fluids; it may also form in metacarbonate rocks during crystallization of common uvitic tourmaline. High miscibility with other tourmaline end-members indicates the large petrogenetic potential of magnesio-lucchesiite in Mg,Al-rich calcsilicate rocks, as well as contact-metamorphic, and metasomatic rocks.
Raman spectroscopic quantification of tetrahedral boron in synthetic aluminum-rich tourmaline
https://doi.org/10.2138/am-2021-7758Kutzchbach et al. describe the correlation of the tetrahedral B content in synthetic Al-rich tourmaline determined by EMPA analyses with the summed relative intensity of all OH-stretching bands between3300–3430 cm–1. Applying the regression equation to natural 4B-bearing tourmaline from the Koralpe (Austria) reproduces the EMPA-derived value. This demonstrates that Raman spectroscopy provides a fast and easy-to-use tool for the quantification of tetrahedral boron in tourmaline. The knowledge of the amount of tetrahedral boron in tourmaline has important implications for the better understanding and modeling of B-isotope fractionation between tourmaline and fluid/melt, widely used as a tracer of mass transfer processes.
Thermal expansion of minerals in the pyroxene system and examination of various thermal expansion models
https://doi.org/10.2138/am-2021-7650Based on the new data for thirteen chemically distinct pyroxenes, Hovis et al. provide a review of thermal expansion data and models for clino- and orthopyroxenes and model changes in thermal expansion with temperature according to the Fei empirical and Kroll physical models of thermal expansion. Conclusions are drawn about the effects of Fe2+- Mg substitution, Ca-Na concentration, and other chemical substitutions on the magnitudes of volume and axial thermal expansion. In part, differences in thermal expansion can be related to the concentration of M1 polyhedra along the b axis and relative charges of the substituted cations in that site. These results have implications for the modeling of geophysical and thermodynamic behavior of high-temperature pyroxene-bearing assemblages.
Incorporation of tetrahedral ferric iron in hydrous ringwoodite
https://doi.org/10.2138/am-2021-7539Thomson et al. synthesized hydrous Fo91 ringwoodite crystals at 20 GPa and high-temperature conditions using a multi-anvil press. Recovered crystals were analyzed using electron microprobe analysis, Raman spectroscopy, infrared spectroscopy, synchrotron Mossbauer spectroscopy, single-crystal X-ray diffraction, and single-crystal Laue neutron diffraction to characterize the chemistry and crystallography of the samples. Analysis of the combined datasets provides evidence for the presence of tetrahedrally coordinated ferric iron and multiple hydrogen incorporation mechanisms within ringwoodite. Tetrahedral ferric iron is coupled with cation disorder of silicon onto the octahedrally coordinated site. Cation disorder in mantle ringwoodite minerals may be promoted in the presence of water, which could have implications for current models of seismic velocities within the transition zone.
The evolution of saponite: An experimental study based on crystal chemistry and crystal growth
https://doi.org/10.2138/am-2021-7625Zhang et al.’s synthesis experiments on saponite show that element distributions within clay minerals are related to the synthesis temperature gap between starting materials and resultant products. Larger Mg- or Ni- clusters can form when the respective synthesis temperature of the starting materials and of the resultant products are close (i.e., low T). Thus, the clustering is not only related to given temperature conditions but also to the characteristics of the starting materials in terms of crystallinity degree and particle size. Crystal-chemistry and crystal growth could be used to predict the environmental transition and element migration. Similar experiments with more complexities could offer potentials to predict the mineral geochemistry and better understand and parametrize natural processes.
Hydroxylpyromorphite, modern description and characterization of a mineral important to lead-remediation
https://doi.org/10.2138/am-2021-7516Olds et al. describe the structure and properties of a new mineral, hydroxylpyromorphite. Hydroxylpyromorphite is a key phase used for remediation of Pb-polluted aquifers and its ability to remove Pb from drinking water has been heavily studied. Their description provides a deeper understanding to the anion column arrangements in pyromorphites and other Pb-bearing apatites, which may help guide and optimize Pb sequestration, or reveal how anion and cation substitution affects important properties such as solubility, reactivity, and mobility of Pb in natural systems.
Experiments on two techniques for the removal of barite from detrital zircon
https://doi.org/10.2138/am-2021-7436Martin et al. performed experiments on two techniques for the removal of barite from detrital zircon: conventional milling method and a new chemical technique. Milling in a ball mill fractures and removes zircon along with barite. Milling thus can introduce bias into a detrital zircon sample. Boiling in aqueous Na2CO3 solution converts barite to barium carbonate, which can be separated from zircon by hand or by hot nitric acid bath. Boiling in concentrated HNO3 did not affect the U-Pb or Lu-Hf isotopic systems in zircon and boiling in Na2CO3 solution followed by HNO3 did not disturb the Lu-Hf system. Boiling in concentrated Na2CO3 solution followed by HNO3 did affect the U-Pb isotopic system in zircon. These results highlight the importance of proper technique during zircon isolation to minimize the introduction of bias into the sample.
Discovery of terrestrial allabogdanite (Fe,Ni)2P, and the effect of Ni and Mo substitution on the barringerite-allabogdanite high-pressure transition
https://doi.org/10.2138/am-2021-7621Britvin et al. report the discovery of a high-pressure phosphide, allabogdanite, (Fe,Ni)2P, in phosphide assemblages confined to the surficial metamorphic rocks at the Dead Sea basin in Israel. High-pressure high-temperature experiments show that the low- to high-pressure phase transition for natural (Fe,Ni)2P from the Dead Sea occurs at a pressure of 25 GPa (~250 kbar) and a temperature of ~1400 °C.
Pseudomorphic 9-line silician ferrihydrite and Fe-rich serpentine-group minerals in FeTi-oxide rich ferroan peridotite, Laramie anorthosite complex, Wyoming, U.S.A.
https://doi.org/10.2138/am-2021-7768Evans et al. describe a unique occurrence of the mineral ferrihydrite as a pseudomorph after a sulfide mineral in a Larimie Complex, Wyoming, peridotie. Ordinarily, ferrihydrite is a supergene mineral occurring as mine waste. In this new occurrence, ferrihydrite displays elemental covariations never before described in this mineral. Co-variations of Si, Mg, and total Fe represent a crystal chemical response to subsurface altering solutions and its superior (9-line) crystallinity.
Synthesis and characterization of Fe(III)-Fe(II)-Mg-Al smectite solid solutions and implications for planetary science
https://doi.org/10.2138/am-2020-7419CCBYNCNDFox et al.’s detailed characterization of the full compositional range of smectite minerals using techniques analogous to those employed in planetary exploration will help improve these capabilities for identification and compositional quantification, and in turn better the ability to interpret alteration environments throughout the solar system. They synthesize and report on the spectroscopic properties of a suite of smectite samples that span the intermediate compositional range between Fe, Mg, and Al end-member species, including both Fe(III)- and Fe(II)-dominated samples.
Evidence for a two-stage particle attachment mechanism for phyllosilicate crystallization in geological processes
https://doi.org/10.2138/am-2021-7529He et al. describe a nonclassical crystallization process for phyllosilicates forming at elevated temperatures in magmatic and metamorphic environments whereby oriented attachment of building blocks occurs along the (001) plane and/or the &91;001&93; direction simultaneously. Nanoparticles are basic building blocks for coarsening of phyllosilicate crystals. The results suggest that the microstructure of magmatic and high-grade metamorphic minerals may have the potential to reveal the evolution history of melts and high-temperature metamorphic processes.
Multiple generations of tourmaline from Yushishanxi leucogranite in South Qilian of western China record a complex formation history from B-rich melt to hydrothermal fluid
https://doi.org/10.2138/am-2021-7473Liu and Jiang studied elemental and boron isotopic compositions of tourmaline in leucogranite dike in the Yushishanxi Nb-Ta mining area in the Yushishan district of the South Qilian orogenic belt in western China. They identified five distinct types of tourmaline that show significant chemical variations and core-rim zoning trends. Detailed petrological, geochemical, and isotopic studies reveal four major generations of tourmaline growth from magmatic to hydrothermal, all of which belong to the alkali group and schorl-dravite solid solution series. The authors propose that the isotopically light fluids may have derived from B release during mica breakdown within the surrounding metamorphic rocks.
Zhanghuifenite, Na3Mn2+4Mg2Al(PO4)6, a new mineral isostructural with bobfergusonite, from the Santa Ana mine, San Luis province, Argentina
https://doi.org/10.2138/am-2021-7598Yang et al. describe the physical and chemical properties of a new mineral, zhanghuifenite, and its crystal structure determined from single-crystal X-ray diffraction data, illustrating its structural relationships to bobfergusonite in particular and alluaudite-type minerals in general. Furthermore, they predict the existence of three more new minerals in the bobfergusonite group based on the data.
Re-examined heterotype solid solution between calcite and strontianite and Ca-Sr fluid-carbonate distribution: an experimental study in the system CaCO3-SrCO3-H2O at 0.5-5 kbar and 600 degrees Celcius
https://doi.org/10.2138/am-2021-7783Schiperski et al. present a revised phase diagram in the system CaCO3-SrCO3 at 600 °C and 0.5-5.0 kbar based on new experimental data and thermodynamic calculations. In addition, for the first time Kd-values of Ca/Sr carbonate-fluid are presented at this condition. Results show that Sr has a high tendency for being accumulated into carbonates rather than in the fluid, which is a substantial difference from silicate-fluid systems.
On the formation of arrays of micro-tunnels in pyrope and almandine garnets
https://doi.org/10.2138/am-2021-7939Rabier et al. show that the intricate tunnels imaged in pyrope and almandine garnets found in soils and river sediments can be fully explained by abiogenic etching of dislocation microstructures contained within the minerals. There are striking geometric similarities between these tunnels and dislocation networks that have been documented in numerous natural and synthetic garnets.
Book review
https://doi.org/10.2138/am-2021-B106613May 2021
How to apply elastic geobarometry in geology
https://doi.org/10.2138/am-2021-7845This paper by by Fabrizio Nestola highlights the new "EntraPT" web application, published by Mazzucchelli et al. (2021) in American Mineralogist, which provides all recent advances of elastic geobarometry within a single platform. This application allows the user to interpret the residual strain of anisotropic inclusions in an intuitive and consistent manner. At the same time, the platform provides the tools needednto perform calculations of the residual pressure and of the entrapment pressure and temperature of isotropic and anisotropic systems using a self-consistent set of thermoelastic properties.
Early Archean alteration minerals in mafic-ultramafic rocks of the Barberton greenstone belt as petrological analogs for clay mineralogy on Mars
https://doi.org/10.2138/am-2021-7656The upcoming Mars2020 rover analyses of Jezero crater will investigate phyllosilicate and carbonate-bearing outcrops in delta sediments, as well as basaltic bedrock containing olivine, chlorite, and serpentine (e.g., Goudge et al. 2017, Bramble et al. 2017; Amador et al., Brown et al. 2020). If chlorite- and serpentine-bearing rocks are encountered by Mars2020, the rock textures and spectral analyses in this study by Grosch et al. will provide ground truth to understand potential formation conditions for those rocks.
Characterization of the metasomatizing agent in the upper mantle beneath the northern Pannonian Basin based on Raman imaging, FIB-SEM, and LA-ICP-MS analysis of silicate melt inclusions in spinel peridotite
https://doi.org/10.2138/am-2021-7292This study by Liptai et al. presents a combination of analytical methods applicable to silicate melt inclusions as an alternative to microthermometry. The combined analyses allow the acquisition of both geochemical properties and 3D structure of the daughter phases. With the use of these methods, composition, origin and crystallization history of a trapped metasomatizing melt was revealed in the upper mantle of the northern part of the Pannonian Basin (Central Europe).
The potential for aqueous fluid-rock and silicate melt-rock interactions to re-equilibrate hydrogen in peridotite nominally anhydrous minerals
https://doi.org/10.2138/am-2021-7435Hydrogen diffusion chronometry provides first-order constraints on how quickly mantle mineral water contents can be overprinted by hydrothermal alteration and melt-rock interactions. Peridotite xenoliths likely completely lose their mantle-inherited water contents prior to eruption due to fast H re-equilibration timescales. Re-equilibration occurs in as little as 10 minutes at 1000 °C to several of years at 600 °C, suggesting that both melt- and/or aqueous fluid-rock interactions can explain why many peridotites have water contents decoupled from trace elements and that are too high to reflect residues of melting. Lynn and Warren also posit in this contribution that hydrogen has the unique potential to be used at low-T (e.g., 300 °C) to investigate the timescales of serpentinization.
Oxygen isotope ratios in zircon and garnet: A record assimilation and fractional crystallization in the Dinkey Dome peraluminous granite, Sierra Nevada, California
https://doi.org/10.2138/am-2021-7472Quintero et al. demonstrate that δ18O variations in zircon and garnet from the Dinkey Dome peraluminous granite in the Sierra Nevada, California, show how a peraluminous pluton is constructed from multiple batches of variably contaminated melts, suggesting that early-stage arc magmas sample crustal heterogeneities in small melt batches. Further progressive invigoration of the arc more effectively blends with mantle melts in source regions. Peraluminous magmas provide key details of the nascent arc and pre-batholithic crustal structure.
An evolutionary system of mineralogy, Part IV: Planetesimal differentiation and impact mineralization (4566 to 4560 Ma)
https://doi.org/10.2138/am-2021-7632The evolutionary system of mineralogy attempts to classify minerals based on their historical context, considering when and how the minerals formed, in addition to the traditional approach of documenting major element chemistry and atomic structure. In Part IV of their series, Morrison and Hazen consider minerals formed in the partially molten interiors of asteroids that began to separate metal-rich cores from silicate-rich mantles, as well as minerals transformed by high-pressure impacts. The 130 new natural kinds of minerals considered here all formed within the solar system's first 10 million years -- a time when high pressure first became an important factor in the formation of minerals.
Constraints on deep, CO2-rich degassing at arc volcanoes from solubility experiments on hydrous basaltic andesite of Pavlof Volcano, Alaskan Peninsula, at 300 to 1200 MPa
https://doi.org/10.2138/am-2021-7531Mafic magmas that enter Earth's crust from the mantle are commonly estimated to have appreciable CO2 concentrations, but direct experimental determinations are few of CO2 solubility at pressures appropriate for the continental lower crust and underlying uppermost mantle. This study by Mangan et al. determines CO2 solubility in that pressure range for basaltic andesite, a widespread subduction zone magma type, across a range of dissolved H2O concentrations suitable for arc magmas. Results show that basaltic andesite can retain substantial dissolved CO2 in the deep arc crust, consistent with geochemical estimates, but that published models of magmatic CO2 solubility are inaccurate for mid-crustal to uppermost mantle pressures, some overestimating and others underestimating the measured concentrations. Systematic studies varying individual melt compositional components may be required to improve understanding of controls on solubility and to develop robust models of H2O-CO2 degassing in the deep arc crust.
Electrical conductivity of diaspore, δ-AlOOH and ε-FeOOH
https://doi.org/10.2138/am-2021-7605This study by Wang and Yoshino addresses the measurement of the electrical conductivity of three hydrous minerals: diaspore, δ-AlOOH, and ε-FeOOH. Investigations on such phases became hot topics because of their wide pressure-temperature stability field (Sano et al. 2008; Nishi et al. 2017; Duan et al. 2018; Hu et al. 2016, 2017). Electrical conductivity measurement of these minerals is also useful to understand deep water cycle. This study could provide important insights on the electrical behaviors of hydrous minerals and water cycle in the Earth's interior. Three significant outcomes were obtained from this study. (1) Diaspore shows a negative pressure dependence on the conductivity because of the ineffectual change of the length of O1H bond with increasing pressure. (2) δ-AlOOH and ε-FeOOH have shorter H...O2 bonds and longer O1H bonds which can account for the relatively higher conductivity than diaspore. (3) ε-FeOOH indicates higher conductivity than δ-AlOOH, which might be caused by the contribution of a small polaron conduction. The result of this study could be applied to the trends of the conductivity-depth relation for various lithologies including hydrous minerals in subduction zones such as altered oceanic crust, hydrous peridotites, and subducted sediments.
A reassessment of the amphibole-plagioclase NaSi-CaAl exchange thermometer with applications to igneous and high-grade metamorphic rocks
https://doi.org/10.2138/am-2021-7400The existing calibrations of amphibole-plagioclase and amphibole-only thermometers yield inaccurate temperature estimates. Three new expressions of the amphibole-plagioclase NaSi-CaAl exchange thermometer are calibrated in this work by Molina et al. that yield an overall similar precision (plus or minus 50°C), but are significantly more accurate. The new expressions can be used in a wide range of igneous and high-grade metamorphic rocks that bear subcalcic to calcic amphibole and oligoclase or more calcic plagioclase
Structural variations across the nepheline (NaAlSiO4)-kalsilite (KAlSiO4) series
https://doi.org/10.2138/am-2021-7484Antao and Hovis obtained the crystal structure of 19 samples within the (Na,K)AlSiO4 series on a fine compositional scale. Well-defined structural variations occur across the Ne-Ks series. Natural samples are not yet available to give a complete understanding of Ne-Ks solid series, which is overcome by using "synthetic" ion-exchanged, powder samples instead of single crystals. Natural samples with limited solid solutions plot close to their "synthetic" samples, so their results apply to natural systems. However, it would be interesting to see how the results for other natural samples with different chemistries, if discovered, compare to the present results. Phase transitions in minerals and their reversibility continue to intrigue researchers. Synchrotron high-resolution powder X-ray diffraction uses a highly intense beam that is sensitive to fine-scale intergrowths or overlapping multi-phases with subtle change in symmetry that cannot be resolved with conventional powder X-ray diffraction. The P31c to P63 transition in kalsilite may be monitored using the 111 reflection that is clearly observable with HRPXRD. K-rich nepheline and kalsilite occur in volcanic rocks as high temperatures expand the aluminosilicate framework to accommodate large K atoms. Particularly, P63 Ks occurs in volcanic rocks, and low temperature P31c Ks occurs in metamorphic rocks. The high to low transition is not completely reversible.
The (chemical) potential for understanding overstepped garnet nucleation and growth
https://doi.org/10.2138/am-2021-7354This paper by Nagurney et al. utilizes phase equilibria modeling to understand the energetic consequences of metamorphic minerals, specifically garnet, not forming at thermodynamic equilibrium. They compare the chemical potentials of garnet-forming oxide components (MnO, CaO, FeO, MgO, Al2O3) between two calculations: one in which Gibbs free energy is minimized and one in which the minimization proceeds under identical conditions but in the absence of garnet. This allows them to study the energetic differences between a garnet-bearing and a garnet-absent calculation. Their results highlight that immediately up temperature of garnet-in, differences in the chemical potentials between the two calculations are commonly minimal for some components. In all twelve examples used in this study the chemical potential of Al2O3 (mu-Al2O3) diverges between garnet-bearing and garnet-absent calculations at greater P-T conditions than that of MnO, CaO, FeO and MgO. This P-T point at which mu-Al2O3 diverges is a function of bulk-rock MnO content. Further, results emphasize that immediately up temperature of garnet-in the total volume of garnet in a rock is commonly calculated to be minimal. These results highlight the necessity for petrologists to interrogate phase equilibria modeling data when interpreting the amount of overstepping of the garnet-in reaction.
EntraPT: an online platform for elastic geothermobarometry
https://doi.org/10.2138/am-2021-7693CCBYNCNDDuring geodynamic processes minerals in rocks can recrystallize at depth in the Earth at high pressures and temperatures and new minerals form. During crystallization, growing minerals can trap other minerals within them as inclusions. After millions of years, some of these rocks are exposed on the Earth's surface and become available to researchers for direct observation. The inclusions deform during exhumation because of the removal of external pressure and temperature. By measuring the residual deformation in the inclusion and applying specific calculations it is possible to estimate the conditions (pressure and temperatures) of the recrystallization; the time when the inclusion was entrapped in its host. This in turn gives us a wealth of information to understand large scale geological processes such as the subduction and the collision of tectonic plates. In this Open Access paper, Mazzucchelli et al. present EntraPT, a new freely accessible online application that eases the interpretation of the residual deformation of inclusions and performs the calculations to estimate the entrapment conditions. EntraPT gives a standardized procedure to make it easier for researchers to check, store, and share their data.
Sodium nanoparticles in alkali halide minerals: Why is villiaumite red and halite blue?
https://doi.org/10.2138/am-2021-7917Radiation damage in natural alkali halides causes alkali ions to form metallic nanoparticles that produce vivid bright colors. In this paper by Calas et al., the Lorentzian lineshape of the main absorption band characterizes a Surface Plasmon Resonance effect due to Na colloids, about 2.5-3 nm large. As the resonance wavelength depends on the refractive index of the mineral, the position of this band shifts between the two minerals. The resulting change of the transmission window causes a dramatic change of color from red villiaumite to blue halite. This approach may also help understand the purple color of irradiated fluorite, which also arises from the presence of metal colloids.
April 2021
Gamma-enhancement of reflected light images: A rapid, effective tool for assessment of compositional heterogeneity in pyrite
https://doi.org/10.2138/am-2020-7606Zhu et al. developed a gamma correction method to display impurity-induced reflectance variation of pyrite at grain scale. This method enhances optical digital signal differences obtained under reflected light using an optical microscope. Incorporation of As in pyrite reduces the total number of electrons that could be excited to produce reflected light, resulting in decrease of reflectance. Gamma correction is a rapid, effective tool for the assessment of compositional heterogeneity in pyrite and other ore minerals with negligible bireflectance. Gamma-enhanced images may help constrain links between textures and compositions of minerals prior to subsequent quantitative analyses.
Thermal metamorphic history of Antarctic CV3 and CO3 chondrites inferred from the first- and second-order Raman peaks of polyaromatic organic carbon.
https://doi.org/10.2138/am-2021-7507Yesiltas et al. determined the first- and second-order carbon Raman peak parameters in an effort to infer asteroidal thermal metamorphic history. The second-order carbon peaks are quantitatively studied for the first time, which contain information that the first-order peaks don't. Peak metamorphic temperatures of the investigated meteorites have been estimated based on the width of the D band as well as the calculated Raman spectral curvature.
A quantitative description of fission-track etching in apatite
https://doi.org/10.2138/am-2021-7614Aslanian et al. propose a quantitative model of fission-track etching in apatite and use it for calculating the shapes and dimensions of etched tracks in different faces. Their model replaces an earlier model and invalidates certain restrictive dating practices based on it. This can have a profound effect on fission-track dating. Their measurements show that the length of confined tracks increases with etch time at a decreasing average rate that differs from the track-etch rate as well as from that of undamaged apatite. Step-etch experiments are shown to be useful for reducing or eliminating procedure-related effects from the track-length data and so for accessing more fundamental track properties resulting from formation and annealing.
Spectroscopic analysis of allophane and imogolite samples with variable Fe abundance for characterizing the poorly crystalline components on Mars
https://doi.org/10.2138/am-2020-7329Poorly crystalline nanophase minerals that occur as weathering products on Mars contain critical information about the evolution of the early martian climate. Jeute et al. present compositional, structural, and spectroscopic data on a set of synthetic Mars analog nanophase aluminosilicates, including imogolite and allophanes with varying Al-Si ratios. They show that changes in the Al-Si ratio can be remotely detected, and that this measurement will help evaluate current models for the climatic evolution of Mars.
The relationship between 207Pb NMR chemical shift and the morphology and crystal structure for the apatites Pb5(AO4)3Cl, vanadinite (A = V), pyromorphite (A = P), and mimetite (A = As)
https://doi.org/10.2138/am-2021-7368NMR spectroscopy of single crystals is the method of choice to precisely determine the full tensors of NMR interaction parameters via orientation-dependent measurements. Zeman et al. conducted a series of single-crystal NMR experiments on natural minerals, in particular on lead-bearing compounds, with the aim to extract the chemical shift tensor of 207Pb in high precision. Going beyond mere tensor determination, they noted that within the mineral family of vanadinite, pyromorphite, and mimetite, the NMR chemical shift may be related to some structural parameters such as unit-cell volume. From evaluating the number of the NMR resonances and their respective line widths, information about the mosaicity of these minerals could be derived.
Effect of cationic substitution on the pressure-induced phase transitions in calcium carbonate
https://doi.org/10.2138/am-2021-7547Martirosyan et al. studied a member of the (Ca,Sr)CO3 solid solution series using in situ Raman spectroscopy at pressures up to 55 GPa and temperatures up to 1273 K. They observed crystallization of the CaCO3-II-type structure at 1273 K and 2 GPa. A new high-pressure modification, Sr-calcite-IIIc, was detected at 7-14 GPa. Thus, substitution of Ca2+ with Sr2+ promotes the formation of structures with larger cation coordination numbers such as aragonite, CaCO3-VII, and post-aragonite at lower P-T conditions compared to pure CaCO3.
Immiscible-melt inclusions in corundum megacrysts: Microanalyses and geological implications
https://doi.org/10.2138/am-2020-7347Xu et al. identified two types of inclusions in the corundum megacrysts from Changle, China. Type I inclusion consists of a dark part (DP) and a bright part (BP), which were formed due to liquid immiscibility at ~1200 C; the former is composed of quartz, corundum, and amorphous substance-1, and the latter is composed of baddeleyite and amorphous substance-2. Type II inclusion is composed of zircon, quartz, and amorphous substance-3. The novel inclusions, together with other previously found mineral inclusions in Changle corundum, demonstrate that both the alkaline felsic melt and carbonatitic melt existed, and they derived from metasomatized mantle. The ages of zircon inclusions show that the corundum megacrysts crystallized from syenitic-type differentiated of earlier underplated basalts at the crust-mantle boundary and were brought up by later episodic basalt eruptions.
Water quantification in olivine and wadsleyite by Raman spectroscopy and study of errors and uncertainties
https://doi.org/10.2138/am-2021-7264Martinek and Balfan-Casanova demonstrate that Raman spectroscopy allows the study of water quantification in different phases of fine polymineralic samples of complex composition, with a wide range of measurable water contents. The water contents of olivine and wadsleyite can be measured using this method with a simple sample preparation. Despite being around 25%, the uncertainties on water concentration are sufficiently low to infer the presence of dehydration that was induced by melting or other geologic processes.
High-pressure and high-temperature vibrational properties and anharmonicity of carbonate minerals up to 6 GPa and 500 C by Raman spectroscopy
https://doi.org/10.2138/am-2020-7404Carbonate minerals play a dominant role in the deep carbon cycle. Farsang et al. measured the high-pressure and high-temperature vibrational properties of all aragonite-group and calcite-group carbonate minerals up to 6 GPa and 500 C by Raman spectroscopy in order to understand their anharmonicity under crustal and upper mantle conditions.
Vanadium-induced coloration in grossite (CaAl4O7) and hibonite (CaAl12O19)
https://doi.org/10.2138/am-2020-7544Ardit et al. studied the unusual coloration in hibonite (purple) and grossite (light violet) crystals, caused by high concentrations of vanadium. Characterization of these specimens by means of single-crystal X-ray diffraction and absorption spectroscopy (aided by EMPA chemical analyses) provides information of both long- and short-range characteristics of their crystal structures.
Incorporation mechanism of tungsten in W-Fe-Cr-V-bearing rutile
https://doi.org/10.2138/am-2021-7653Majzlan et al. examined the position of tungsten in the structures of two common minerals, rutile and hematite. It is of interest because these and related minerals serve as vehicles that enrich the sediments in the element tungsten. These sediments can be later melted and transformed into fertile magmas that form ore deposits. Tungsten was found to enter the structure of rutile, but in hematite, there exist nanoinclusions of iron-tungsten oxide intergrown with the host mineral. Rutile is a better vehicle for tungsten; hematite can serve as such under specific moderately reducing conditions.
Titanium diffusion profiles and melt inclusion chemistry and morphology in quartz from the Tshirege Member of the Bandelier Tuff
https://doi.org/10.2138/am-2021-7395Boro et al. describe that melt Inclusions in quartz from the chemically zoned Tshirege Member of the Bandelier Tuff record the compositional evolution of a caldera-forming magma chamber. Variable melt inclusion faceting suggests timescales of millennia for crystal mush generation and crystal storage. Titanium concentration zoning in quartz suggests shorter timescales of decades to centuries for recharge and mobilization of said mush to produce an eruptible, zoned magma body.
Vasilseverginite, Cu9O4(AsO4)2(SO4)2, a new fumarolic mineral with a hybrid structure containing novel anion-centered tetrahedral structural units
https://doi.org/10.2138/am-2020-7611Pekov et al. describe the unique structure of a new mineral, vasilseverginite Cu9O4(AsO4)2(SO4)2, which can be considered as a hybrid of the structures of popovite Cu5O2(AsO4)2 and dolerophanite Cu2O(SO4). The concept of hybridization of mineral species developed in this study may give new ideas for the preparation of novel structural architectures on the border of stability fields of chemically and structurally simpler compounds. The discovery of vasilseverginite indicates the existence of polynuclear oxocentered copper clusters in a gaseous phase, which may be a form of transport of Cu by volcanic gases.
Priscillagrewite-(Y), (Ca2Y)Zr2Al3O12: A new garnet of the bitikleite group from the Daba-Siwaqa area, the Hatrurim Complex, Jordan
https://doi.org/10.2138/am-2021-7692Galuskina et al. report the discovery of a new garnet, priscillagreowte-(Y), belonging to the bitikleite group of the garnet supergroup. The mineral was found in apatite-bearing varicolored spurrite marble in the Daba-Siwaqa area, central Jordan. Priscillagrewite-(Y) is interpreted to be a relic of the high-temperature association formed in the progressive stage of the peak pyrometamorphism conditions, when the temperature could have reached close to 1000 C. The authors suggest that there is a reasonable chance of finding priscillagrewite-(Y) in ultrarefractory calcium-aluminum-rich inclusions from chondrites, then it can be the third garnet species originating in the solar nebula.
Stoefflerite, (Ca,Na)(Si,Al)4O8 in the hollandite structure: A new high-pressure polymorph of anorthite from martian meteorite NWA 856
https://doi.org/10.2138/am-2021-7563Stoefflerite is the high-pressure polymorph of anorthite. Tschauner et al. show that in Earth's transition zone, it is a component of the important carrier of K, liebermannite. In shock-metamorphic environments, it marks high-pressures at temperatures above the Hugoniot but below formation of stable phases.
Recycled volatiles determine fertility of porphyry deposits in collisional settings
https://doi.org/10.2138/am-2021-7714There have long been debates about whether copper and gold come from the mantle or crust. Previous hypotheses dominantly relied on the metal concentration, which is lower in both Earth's mantle and crust. The volatile elements Cl and S are the most important factors controlling the transport of ore metals in magma; therefore, their source and evolution can be a key to understanding the genesis of Cu-Au deposits. Xu et al. show that geochemical and Rb-Sr isotopic data on apatite from 12 porphyry systems across Iran, Tibet, and western China, can distinguish fertile magmas from barren magmatic suites and indicate the importance of volatiles, recycled from previous oceanic subduction, in collisional settings. This study is of importance to the Earth's volatile evolution and economic geologists.
Nitrogen diffusion in silicate melts under reducing conditions
https://doi.org/10.2138/am-2021-7799CCBYNCNDNitrogen and argon are the two most abundant elements in the current atmosphere, but the elemental composition of Earth's interior and surficial reservoirs may have evolved over time. In this study, Boulliung et al. provide the first constraints on nitrogen diffusion in natural-like silicate melt with implications on N/Ar fractionation during reducing magmatic events, such as Earth's magma ocean stage. The result implies a N/Ar fractionation during reducing magmatic events, such as Earth's magma ocean stage.
March 2021
An evolutionary system of mineralogy, Part III: Primary chondrule mineralogy (4566 to 4561 Ma)
https://doi.org/10.2138/am-2020-7564The evolutionary system of mineralogy relies on varied physical and chemical attributes, including trace elements, isotopes, solid and fluid inclusions, and other information-rich characteristics. To understand processes of mineral formation and to place natural condensed phases in the deep-time context of planetary evolution, Hazen et al. add Part III of their evolutionary system that considers the formation of 43 different primary mineral phases in chondrules, which are igneous droplets that formed early in the history of the solar system, more than 4.56 billion years ago.
Raman Spectroscopy Study of Manganese Oxides - Layer Structures
https://doi.org/10.2138/am-2021-7666Micro-Raman is a powerful tool for identification and characterization of biotic and abiotic Mn oxide phases from diverse natural settings (including on other planets) and thereby can provide new insights into the roles of these phases in our environment. Post et al. provide results from what they believe is the most comprehensive analysis of the Raman spectra for layer-structure Mn oxide phases to date, collected from a large number and variety of natural and synthetic samples, drawing from the Smithsonian Institution's extensive collection of Mn oxide specimens, and elsewhere. In many cases, the specimens have been characterized in detail using supplementary techniques. Additionally, Post et al. present representative spectra from different specimens, localities, and crystal orientations. A major goal of this study is to provide a comprehensive base of information (a spectral database is provided as supplementary data) that can be used for identifying the various Mn oxide mineral phases, with an emphasis on natural samples. Finally, Post et al. explore spectral trends for some specific phases that provide insights about composition, crystal structure, symmetry, and in some circumstances, Mn oxidation states.
Raman signatures of the distortion and stability of MgCO3 to 75 GPa
https://doi.org/10.2138/am-2020-7490Zhao et al. report the Raman modes of natural magnesite, MgCO3 , up to 75 GPa at room temperature. They detected abnormal behavior in MgCO3, including the splitting of Raman peaks of T and v4 modes at approximately 30 and 50 GPa, respectively. The phenomena are assigned as MgCO3-Ib and MgCO3-Ic produced by the rotation of MgO6 octahedra. The distorted environment of the chemical bond would greatly improve the stability of magnesite over a large pressure and temperature range in relation to its melting or decomposition. Both experimental and theoretical evidence indicates that the diversity of distorted structural environments, including corner-sharing CO4 tetrahedra forming C3O9 three-membered rings. Compared to the low-pressure threefold coordinated carbonates (CO3)2-triangles in the structure, the tetrahedrally coordinated carbonates are expected to exhibit substantially different reactivity and different chemical properties in the liquid state. These crystallographic characteristics in carbonates may play an important role in deep carbon reservoirs and fluxes in the deep Earth. Furthermore, the bonding strength in MgCO3 changes through lattice distortion and structural transition, likely impacting the distribution of carbon and magnesium isotopes in the deep mantle.
Competitive adsorption geometries for the arsenate As(V) and phosphate P(V) oxyanions on magnetite surfaces: Experiments and theory
https://doi.org/10.2138/am-2020-7350Adsorption of arsenate and phosphate on magnetite was studied by Liang et al. using in situ ATR and 2D-COS. Monodentate mononuclear and bidentate binuclear complexes dominate in phosphate adsorption. Arsenate forms bidentate binuclear complexes with fewer outer-sphere species. Arsenate displays a higher competitive ability than phosphate. The competitive ability is related to adsorption geometry and the heterogeneity of surface active sites.
Probing transformation path from aluminum (oxy)hydroxides (boehmite, bayerite, and gibbsite) to metastable alumina: A view from high-resolution 27Al MAS NMR
https://doi.org/10.2138/am-2020-7481Kim and Lee investigated the dehydration paths to metastable alumina from various aluminum (oxy)hydroxide precursors (i.e., boehmite, bayerite, and gibbsite) in the low-temperature range (~300 °C) using high-resolution 27Al NMR. The results confirm that the phase transformation paths depend on the type of precursor minerals. The precursor-dependent structural evolution in the low-temperature range helps to understand the geological processes involving metastable phases and their dehydration in the Earth's surface environments.
Crystal structure of K-cymrite and kokchetavite from single-crystal X-ray diffraction
https://doi.org/10.2138/am-2020-7407Romanenko et al. report their investigation of K-cymrite (KAlSi3O8+H2O) and kokchetavite (KAlSi3O8, IMA-2004-011), which were earlier identified as mineral inclusions in ultra-high pressure metamorphic crustal rocks. However, their crystal structures previously were only guessed on the basis of powder X-ray diffraction patterns. Romanenko et al. present the crystal structures of K-cymrite and kokchetavite by single-crystal X-ray diffraction. For kokchetavite a new space group and unit cell were identified. In addition, the spectroscopic and Thermogravitational data provide important information for the identification and interpretation of these phases in mineral inclusions.
Fluid source and metal precipitation mechanism of sediment-hosted Chang'an orogenic gold deposit, SW China: constraints from sulfide texture, trace element, S, Pb and He-Ar isotopes, and calcite C-O isotopes
https://doi.org/10.2138/am-2020-7508Yang et al. highlight that ore metals in sediment-hosted disseminated orogenic gold deposits can be sourced from both deep fluids and local wallrock and that fluid-rock interaction behaved as a key control on ore precipitation.
Iron isotope fractionation in reduced hydrothermal gold deposits: A case study from the Wulong gold deposit, Liaodong Peninsula, East China
https://doi.org/10.2138/am-2020-7534Pyrite and pyrrhotite are the major Fe-bearing minerals of the quartz-sulfide veins in the Wulong reduced gold deposit. Iron isotope fractionation modeling by Zheng et al. shows that under relatively low oxygen fugacity conditions, pyrrhotite with light δ56Fe crystallized first from the initial ore-forming fluids, resulting in ore-forming fluids with elevated δ56Fe values. Due to an increase of oxygen fugacity, pyrite with heavy δ56Fe started to precipitate later. The iron isotopic compositions provide a new perspective for the initial redox conditions and evolution of the Wulong gold deposit, which are important to trace the source of ore-forming materials and further exploration.
Tungsten mineralization during the evolution of a magmatic-hydrothermal system: mineralogical evidence from the Xihuashan rare-metal granite in South China
https://doi.org/10.2138/am-2020-7514Micas can record the magmatic-hydrothermal evolution of tungsten granite. Li et al. demonstrate that the geochemical variations and textures of zoned micas indicate magmatic fluids, rather than external fluids, were involved in greisenization. The siderite present is related to a Fe, Mn, and CO2-rich fluid under reducing conditions. The greisenization process plays a critical role in tungsten mineralization. The reducing environment and the mixture of a W-rich solution and a Fe-, Mn-rich external fluid facilitated tungsten mineralization.
Crystallization and melt extraction of a garnet-bearing charnockite from South China: Constraints from petrography, geochemistry, mineral thermometer and rhyolite-MELTS modeling
https://doi.org/10.2138/am-2020-7335Zhang et al. investigated the Yunlu garnet-bearing charnockite as an example of the very few peraluminous magmatic charnockites around the world. The magmatic pressure-temperature-melt H2O content and associated crystallization of the charnockite was constrained quantitatively by the integration of petrography, geochemistry, fluid inclusion investigations, mineral thermo-barometry, and thermodynamic modeling. The Yunlu magma solidified at "wet" (H2O-saturated) and "cold" (~630 °C) conditions, which is different from metaluminous charnockites that solidified at "dry" (H2O-unsaturated) and "hot" (>800 °C) conditions. This study indicates that the peraluminous charnockites may experience a distinct crystallization process compared to metaluminous charnockites. Meanwhile, the temperature discrepancies between mineral thermometer results and the magmatic solidus were interpreted by the "melt extraction" model. The study sheds new light on the interpretations of granite thermometry.
Reducing epistemic and model uncertainty in ionic inter-diffusion chronology: A 3D observation and dynamic modeling approach using olivine from Piton de la Fournaise, La Réunion
https://doi.org/10.2138/am-2021-7296CCBYModeling of Mg-Fe zonation in olivine crystals from mafic ejecta and deposits from volcanic eruptions is an often-used tool for calculating magmatic timescales, but sub-perfect diffusion profiles are often rejected. This is a bias that Couperthwaite et al. suggest should, and can, be addressed as a community. The results in this open access paper by Couperthwaite et al. unlock the majority of these profiles and, in doing so, reveal a richer view of magmatic processes than previously could be seen.
Erratum
Memorial of James J. Papike .
February 2021
Effect of water on carbonate-silicate liquid immiscibility in the system KAlSi3O8-CaMgSi2O6-NaAlSi2O6-CaMg(CO3)2 at 6 GPa: Implications for diamond-forming melts
https://doi.org/10.2138/am-2020-7551Partial melting of recycled sediments (metapelites) subducted to mantle depths is essentially controlled by the phengite/K-feldspar + clinopyroxene + carbonate assemblage. Here Shatskiy et al. show that at 6 GPa and nominally dry conditions incipient melting of this assembly occurs at 1050-1100 °C and yields a K-dolomitic melt. By contrast, in the presence of H2O two immiscible phonolite-like and K-dolomitic melts appear. The established melts resemble the carbonatitic and silicic HDFs established in diamonds worldwide.
Jasonsmithite, a new phosphate mineral with a complex microporous framework, from the Foote mine, North Carolina, U.S.A.
https://doi.org/10.2138/am-2020-7582Kampf et al. describe a new mineral, jasonsmithite, from the Foote mine in North Carolina. It has a complex framework structure containing large channels. Its 70% void space makes it one of the most porous mineral structures known and suggests that it may have technological applications.
Titanium in calcium amphibole: Behavior and thermometry
https://doi.org/10.2138/am-2020-7409Thermometry of high-grade metamorphic rocks is difficult due to fast cationic diffusion during slow cooling. Liao et al. propose to use Ti content of amphibole (Ti-Amp) as thermometric tools for igneous and high-grade metamorphic rocks.
Phase relationships in the system ZnS-CuInS2: Insights from a nanoscale study of indium-bearing sphalerite
https://doi.org/10.2138/am-2020-7488Xu et al. conducted micrometer- to nano-scale characterization on sphalerite containing 17-49 mol% CuInS2 that occurs in a pyrrhotite-dominant matrix. Results provide new insights into phase relationships in the system ZnS-CuInS2. Metal ordering is modeled as mixed sites in a sphalerite-type structure, [(Cu,In,Zn)3(Zn0.5Fe0.5)]4S4, with P4-3m symmetry. This modification is distinct from the reported cubic-tetragonal phase transition. Different degrees of fluid percolation explain the textures shown.
Major and trace element composition of olivine from magnesian skarns and silicate marbles
https://doi.org/10.2138/am-2020-7566Nekrylov et al. analyzed olivine from magnesian skarns and silicate marbles, which could be easily distinguished from olivine formed in other processes by unusually low contents of Ni, Co, and Cr and high content of B. These features are linked to the composition of its formation environment -- dolomites and their contacts with intermediate-acidic magmatic rocks.
Decompression experiments for sulfur-bearing hydrous rhyolite magma: Redox evolution during magma decompression
https://doi.org/10.2138/am-2020-7535Understanding redox evolution during magma ascent is important for exploring oxygen fugacity of magma and mantle and for modeling and predicting the chemical species of volcanic gases emitted to the surface. Okumura et al. experimentally investigated redox evolution during magma ascent to the surface. Results showed that sulfur-bearing hydrous rhyolite magma is slightly reduced during the ascent with decompression rates corresponding to explosive and effusive volcanic eruptions.
On the crystal chemistry of sulfur-rich lazurite
https://doi.org/10.2138/am-2020-7317Sapozhnikov et al. report that dark-blue lazurite from Malo-Bystrinskoe deposit (Russia) contains high sulfur. In addition to SO42-, the S3- radical-ion occupies beta-cages within the structure. The idealized formula of the mineral is Na7Ca[Al6Si6O24](SO4)2–(S3)–·H2O. The structure of studied lazurite contains incommensurate modulations.
Experimental evaluation of a new H2O-independent thermometer based on olivine-melt Ni partitioning at crustal pressure
https://doi.org/10.2138/am-2020-7014Pu et al. demonstrate that the partitioning of Ni between olivine and basaltic melt (DNi) is independent of dissolved water (up to 4.3 wt%), unlike the partitioning of Mg (DMg). Olivine-melt equilibrium experiments were performed at 1 bar (anhydrous) and 0.5 GPa (anhydrous and hydrous). The results confirm that an olivine-melt thermometer based on DNi can be applied to hydrous natural basalts from subduction zones without any correction for H2O content.
Contrasting compositions between phenocrystic and xenocrystic olivines in the Cenozoic basalts from central Mongolia: Constraints on source lithology and regional uplift
https://doi.org/10.2138/am-2020-7431Two Cenozoic prominent features are spatio-temporally associated in central Mongolia, i.e., the continental basalts and regional uplift, but their causes and relationship remain unclear. To solve these issues, Zhang et al. conducted analyses of major and trace element compositions for olivine phenocrysts and xenocrysts in the Cenozoic basalts. They conclude that mass deficit in the lithosphere could have caused isostatic uplift of central Mongolia in the Cenozoic.
The composition of garnet in granite and pegmatite from the Gangdese orogen in southeastern Tibet: constraints on pegmatite petrogenesis
https://doi.org/10.2138/am-2020-7388Yu et al. report two stages of garnet growth in granite and pegmatite from the Gandese orogen in Tibet. The first generation of garnet (Grt-I) grew in the pegmatite from early evolved magmatic-hydrothermal fluids, and the second generation (Grt-II) crystallized after dissolution of the preexisting pegmatite garnet in the presence of a granitic magma. Both granite and pegmatite originate from partial melting of the same juvenile crust in the Mesozoic continental arc prior to the Cenozoic continental collision forming the Himalayan orogen.
Formation of metasomatic tourmalinites in reduced schists during the Black Hills Orogeny, South Dakota
https://doi.org/10.2138/am-2020-7405Boron is an important component of granites, pegmatites, and metamorphic rocks in many geologic settings, including collisional orogens. Boron is a highly fluid-mobile element and thus it exchanges easily between these geologic materials. Nabelek describes mineral chemistry of tourmalinite in the Black Hills orogen and proposes ion-exchange reactions that led to its formation. The tourmalinite places constraints on the chemistry of the fluid that caused replacement of a schist by tourmaline and graphite.
New insights into the crystal chemistry of sauconite (Zn-smectite) from the Skorpion zinc deposit (Namibia) via a multi-methodological approach
https://doi.org/10.2138/am-2020-7460Schingaro et al. conducted a multi-methodical characterization of a sauconite (Zn-bearing trioctahedral smectite) specimen from the Skorpion ore deposit (Namibia) using X-ray diffraction, cation exchange capacity analysis, differential thermal analysis, thermogravimetry, infrared spectroscopy and transmission electron microscopy. The results have implications not only for economic geology/recovery of critical metals but also, more generally, in the field of environmental sciences.
The new mineral crowningshieldite: A high-temperature NiS polymorph found in a type IIa diamond from the Letseng mine, Lesotho
https://doi.org/10.2138/am-2020-7567Smith et al. describes crowningshieldite, a new nickel monosulfide mineral that is the high-temperature polymorph of millerite. It was discovered within a fine-grained multi-phase inclusion in a gem quality diamond from the Letseng Mine, Lesotho. The mineral name recognizes G.R. Crowningshield, who was a central figure in research at the Gemological Institute of America for over fifty years.
Elucidating the natural-synthetic mismatch of Pb2+Te4+O3: The redefinition of fairbankite
https://doi.org/10.2138/am-2020-7536The determination of fairbankite's structure by Missen et al. has solved the long-standing uncertainty around the natural Pb2+Te4+O3 formula. Additionally, the fairbankite structure is unique and contains a trimeric tellurite anion new to both synthetic and natural structures.
Are the Thermodynamic Properties of Natural and Synthetic Mg2SiO4-Fe2SiO4 Olivines the Same?
https://doi.org/10.2138/am-2021-7764CCBYIt is unclear whether the thermodynamic properties of some rock-forming minerals and their synthetic analogues are quantitatively the same. Olivine is an important substitutional solid-solution consisting of the two end-members forsterite, Mg2SiO4, and fayalite, Fe2SiO4. Gieger et al. undertook first low-temperature CP measurements on two natural olivines between 2 and 300 K; nearly end-member fayalite and a forsterite-rich crystal Fo0.904Fa0.096. They show that the CP behavior of the natural and synthetic crystals is similar.
January 2021
P-V-T equation of state of hydrous phase A up to 10.5 GPa
https://doi.org/10.2138/am-2020-7132Yang et al. report the precise equation of state of Mg7Si2O8(OH)6 phase A by using the pressure-volume-temperature (P-V-T) data of synthetic pure phase A up to ~10.5 GPa and ~900 K by in situ X-ray diffraction study at the Photon Factory-Advanced Ring (PF-AR) in Japan. Because phase A is one of the important hydrous phases and the water carriers into the Earth's mantle, the precise equation of state of phase A is very important and valuable to investigate the precise thermoelastic properties (e.g., temperature dependence of bulk modulus, thermal expansion, etc.), for discussing the state of the Earth interiors.
Elastic properties and structures of pyrope glass under high pressures
https://doi.org/10.2138/am-2020-7410Hisano et al. indicate that the structural densification of pyrope glass is closely correlated to the change in elastic properties. Compared to other silicate glasses, the variation of chemical composition has a significant effect on the properties and their pressure dependences. Especially, the influence of the magnesium cation on the aluminosilicate glass is larger than that of other cations. Considering the melting of Earth's mantle, the magnesium component is abundant in the melt at the deeper region. Therefore, the knowledge of magnesium-bearing aluminosilicate glass can help us to understand the behavior of deep magmas.
Effects of pH and Ca exchange on the structure and redox state of synthetic Na-birnessite
https://doi.org/10.2138/am-2020-7112Elmi et al. investigate the structures of a series of synthetic Ca-birnessite analogs prepared by cation-exchange with synthetic Na-birnessite at pH concentrations from 2 to 7.5. The exchange experiments described in this paper, performed over a range of pH values (3 to 7.5) and Ca2+ concentrations that are relevant to natural environments, yielded Ca-birnessite products that all have the same triclinic structure with nearly identical unit-cell parameters. Their results reveal that the transformation of Na- to Ca-birnessite is more than a simple replacement of Na by Ca.
A systematic assessment of the diamond trap method for measuring fluid compositions in high-pressure experiments
https://doi.org/10.2138/am-2020-7453Rustioni et al. provide tests of the diamond trap method to study fluid compositions in experimental charges in order to evaluate the reliability of this method. They also describe several experimental improvements of the method that will be useful for further studies. This paper will be quite interesting for a wide audience of experimental petrologists and geochemists.
Origin, properties, and structure of breyite: the second most abundant mineral inclusion in super-deep diamonds
https://doi.org/10.2138/am-2020-7513Breyite is the second most abundant mineral inclusion after ferropericlase in diamonds of super-deep origin. The occurrence of breyite is widely used as a strong indication of a lower mantle (>670 km depth) or at least lower transition zone (>520 km depth) origin of both the host diamond and the inclusion suite. Brenker et al. demonstrate through different formation options that the finding of breyite alone in a diamond is not a reliable indicator for the formation depth, accompanying paragenetic phases such as ferropericlase and/or MgSiO3 are needed. As a large set of papers were published in recent years based on findings on breyite in super-deep diamonds and used to determine the chemical composition of the deep Earth and to decipher deep Earth processes, the new work presented here question some of the conclusions drawn if based on the sole finding of breyite.
Why Tolbachik diamonds cannot be natural
https://doi.org/10.2138/am-2020-7562Litasov et al. provide comprehensive evidence that type Ib cuboctahedral diamonds and microcrystalline diamonds from Kamchatka volcanic rocks and alluvial placers cannot be natural and undoubtedly represent synthetic materials, which appear in the natural rocks by anthropogenic contamination. The major arguments provided in favor of the natural origin of those diamonds can be easily disproved. The cavitation model proposed for the origin of Tolbachik diamonds is also unreliable, since cavitation can cause the formation of nanosized diamonds only.
Deciphering the enigmatic origin of Guyana's diamonds
https://doi.org/10.2138/am-2020-7486Bassoo et al. present a comprehensive study of Guyana's diamonds, using cathodoluminescence, UV fluorescence, Raman spectroscopy, FTIR and δ13C analyses. Inclusion suites and δ13C values indicate diamonds are derived from peridotitic lithospheric mantle at ~1120 °C. Dissolution textures suggest diamonds were hosted in a H2O rich magma. Detrital zircon geochronology and accessory mineral chemistry suggest most diamonds are likely sourced from middle to late Trans-Amazonian rocks which have since weathered into the ~1.98 Ga Roraima Supergroup conglomerates and volcaniclastics. However, provenance of a population of pristine, non-abraded diamonds is enigmatic, because cathodoluminescence and dissolution textures suggest a lesser metamorphic overprint and different source melt than abraded diamonds. This study characterizes and places Guyana's diamonds into the wider context of Guiana Shield and West African Craton evolution.
Precipitation of low-temperature disordered dolomite induced by extracellular polymeric substances of methanogenic Archaea Methanosarcina barkeri: Implications for sedimentary dolomite formation
https://doi.org/10.2138/am-2020-7381Dolomite [CaMg(CO3)2] is a common mineral in the rock record. However, the rarity of modern dolomite and the notorious difficulty in synthesizing dolomite abiotically under Earth-surface conditions result in the long-standing enigma in sedimentary geology, known as the "dolomite problem." Anaerobic microorganisms, such as sulfate-reducing bacteria (SRB) and methanogens have been recognized to mediate dolomite precipitation. However, the mechanistic link between anaerobic bacteria and dolomite formation is still not revealed. In the present study, Zhang et al. report precipitation of disordered dolomite in Ca-Mg carbonate solutions containing purified inactive biomass of a natural consortium of the methanogen Methanosarcina barkeri. Carbonate precipitation experiments in solutions containing capsular extracellular polymeric substances (EPS) extracted from the biomass showed that EPS was the active component that triggered dolomite crystallization. The present study clearly demonstrates the catalytic role of EPS in dolomite formation. It also proves the possible involvement of fermentative bacteria in dolomite formation, which to their best knowledge has not been reported previously. This study provides significant insight into the formation mechanism of microbial-induced dolomite with heavy δ13Cvalues during diagenesis.
Atomic-scale Characterization of Commensurate and Incommensurate Vacancy Superstructures in Natural Pyrrhotites
https://doi.org/10.2138/am-2020-7479CCBYJin et al. report that (1) Superstructures formed by Fe vacancy ordering in natural pyrrhotites are revealed using atomic-resolution scanning transmission electron microscopy; (2) Picometer-scale shifts of individual Fe atomic columns are consistent with a model for the structure of 4C pyrrhotite derived using X-ray diffraction; and (3), 5C-like unequally-sized nano-regions joining at anti-phase-like boundaries lead to the incommensurability observed in 4.91 ± 0.02 C pyrrhotite.
Three-dimensional and microstructural fingerprinting of gold nanoparticles at fluid-mineral interfaces
https://doi.org/10.2138/am-2021-7696Zhou et al. present the first 3D FIB/SEM tomography images and TEM microstructures of gold nanoparticles in an ore mineral, in this case from the world-class Beiya Au deposit, China. The results unravel how intrinsic and extrinsic factors drove the formation of Au nanoparticles at fluid-mineral interfaces. Non-lattice-bound gold in ore minerals is commonly interpreted to be remobilized from exsolution of earlier formed minerals. However, their work offers an alternative explanation that this gold can also be introduced by hydrothermal fluids during later mineralization stages. They also suggest that this gold in many ore minerals may reflect nano-scale permeability between nanopores that developed during dissolution-reprecipitation reactions involving earlier minerals.
Seaborgite, LiNa6K2(UO2)(SO4)5(SO3OH)(H2O), the first uranyl mineral containing lithium
https://doi.org/10.2138/am-2020-7540Kampf et al. report on the first uranyl mineral containing essential lithium, which plays an important role in this structure. The structure is unique among both natural and synthetic phases.
Reheating and magma mixing recorded by zircon and quartz from high-silica rhyolite in the Coqen region, southern Tibet
https://doi.org/10.2138/am-2020-7426Chen et al. report on: (1) High-silica rhyolites (HSRs) erupted at ~74 Ma from the Coqen region, southern Lhasa terrane; (2) Trace element characteristics of zircons from Nuocang HSRs indicate multi-stage magmatic processes involving magma mixing; (3) Ti geothermometers of zircon and quartz suggest reheating in the magma reservoir; and (4) Zircon and quartz with distinct internal textures provide critical insights into the formation of HSR.
Crystal-chemistry and thermal behavior of Fe-carpholite: a case of study from the Pollino Massif (southern Italy)
https://doi.org/10.2138/am-2020-7385Mesto et al. report on: (1) new insights into the crystal chemistry of Fe-Mg carpholites; (2) new data on the thermal evolution of the species, obtained by TG-DTA as well as in situ high-temperature powder diffraction analysis; (3) new Raman data; and (4) first study on the thermal behavior of the most common carpholite composition occurring in HP/LT metasediments in collisional contexts.
New insights into the control of visible gold fineness and deposition: A case study of the Sanshandao gold deposit (Jiaodong, China)
https://doi.org/10.2138/am-2020-7475Peng et al. report on: (1) Mineralogy and chemistry research on the gold-producing zone from –4000 m depth to surface; (2) Two generations of gold grains with different fineness show different gold-precipitating processes; (3) Three types of pyrite record the temporal and spatial evolution of ore-forming fluid; and (4) Integrated gold fineness, pyrite trace element, and sulfur isotope reveal a comprehensive genetic model.
A comment on "An evolutionary system of mineralogy: Proposal for a classification of planetary materials based on natural kind clustering"
https://doi.org/10.2138/am-2021-7590Reply to "A comment on 'An evolutionary system of mineralogy: Proposal for a classification of planetary materials based on natural kind clustering'"
https://doi.org/10.2138/am-2021-7773New Mineral Names
https://doi.org/10.2138/am-2021-NMN106131American Mineralogist Volume 105
December 2020
The Incompressibility of Atoms at High Pressures
https://doi.org/10.2138/am-2020-7123How compressible is an atom? Here, Gibbs et al. report the impact of pressure on the sizes of the bonded radii of a diverse range of atoms by calculating their bonded interactions and electron density distributions as a function of pressure. The results provide novel insights about the compressibility of atoms and changes in their chemical bonding at high pressure.
Phase transitions in ε-FeOOH at high pressure and ambient temperature
https://doi.org/10.2138/am-2020-7468Constraining the accommodation, distribution, and circulation of hydrogen in the Earth's interior is vital to our understanding of the deep Earth due to the significant influence of hydrogen on the material and rheological properties of minerals. Recently, a great deal of attention has been paid to the high-pressure polymorphs of FeOOH. These structures potentially form a hydrogen-bearing solid solution with AlOOH and phase H (MgSiO4H2) that may transport water (OH-) deep into the lower mantle. In this study, Thompson et al. re-evaluated the high-pressure evolution of FeOOH up to ~75 GPa using a combination of synchrotron X-ray diffraction, Fourier transform infrared spectroscopy, and optical absorption spectroscopy. Based on these measurements, the authors report on the phase transitions which occur in ε-FeOOH at ambient temperature and pressures up to 70 GPa.
Thermal state of the upper mantle and the origin of the Cambrian-Ordovician ophiolite pulse: Constraints from ultramafic dikes of the Hayachine-Miyamori Ophiolite
https://doi.org/10.2138/am-2020-7160Kimura et al. demonstrated that a global thermal state of the upper mantle in the Cambrian-Ordovician time, which corresponds to the period of high production of ophiolites, called a ophiolite pulse, was similar to the present one by quantitatively estimating the mantle potential temperature from an arc ophiolite. They also showed that a slab breakoff tectonics is responsible for the magma generation and further argued that such tectonics frequently took place during the Cambrian-Ordovician time resulting in the ophiolite pulse.
Quadrivalent praseodymium in planetary materials
https://doi.org/10.2138/am-2020-7325Praseodymium is a rare earth element (REE) that predominantly occurs in the trivalent oxidation state like most other REEs. However, in some cases it can occur as tetravalent Pr, leading to a subtle anomaly in REE patterns. It is stabilized by high oxygen fugacities and low temperatures. Anenburg et al. find Pr4+ in tetravalent Ce minerals (cerianite and stetindite), and they discuss the possibility of finding a pure tetravalent Pr mineral.
Quantitative microscale Fe redox imaging by multiple energy X-ray fluorescence mapping at the Fe K pre-edge peak
https://doi.org/10.2138/am-2020-7359Ellison et al. describe a synchrotron-based X-ray spectroscopic approach that allows microscale quantitative mapping of Fe valence state by extending the Fe XANES pre-edge technique. An area of interest is mapped at 10 excitation energies, allowing reconstruction, baseline subtraction, and integration of the pre-edge feature to determine Fe(III)/ΣFe at every pixel. By combining the Fe redox mapping approach with hyperspectral Raman mapping, the Fe oxidation state distributions of the major mineral phases can be revealed. They provide an example of application of this method to a partially serpentinized peridotite from the Samail Ophiolite (Oman) that displays a variety of secondary mineral phases with a range of Fe redox states. Quantification of Fe(II) and Fe(III) in these phases allows for determination of the sequence of reactions and Fe transformations that produced the observed alteration assemblage.
Quantification of excess 231Pa in late Quaternary igneous baddeleyite
https://doi.org/10.2138/am-2020-7449Initial excess protactinium (231Pa) is a frequently suspected source of discordance in baddeleyite geochronology, which limits accurate U/Pb dating, but such excesses have never been directly demonstrated. In this study, Sun et al. quantified Pa incorporation in late Holocene baddeleyite from Vesuvius (Campanian Volcanic Province, Central Italy) and Laacher See (East Eifel Volcanic Field, Western Germany) by U-Th-Pa measurements using a large-geometry ion microprobe. Measured 231Pa excesses indicate preferred Pa baddeleyite-melt partitioning relative to U equivalent to, or possibly even more severe, than for zircon. These excesses require significant corrections for 207Pb/206Pb and 207Pb/235U ages. Comparison with partitioning of other trace elements suggests that Pa is predominantly present as Pa4+, even in comparatively oxidized melts. The crystallization age of baddeleyite crystals obtained here for the first time for Somma-Vesuvius syenitic ejecta, which represent parts of the solidified magma chamber margin, is also crucial for constraining magma chamber evolution before major eruptions.
Magma oxygen fugacity of mafic-ultramafic intrusions in convergent margin settings: insights for the role of magma oxidation states on magmatic Ni-Cu sulfide mineralization
https://doi.org/10.2138/am-2020-7351Cao et al. investigated the magma fO2 of the mafic-ultramafic intrusions in typical convergent margin settings to examine the correlation of magma fO2 and Ni-Cu sulfide mineralization. They calculated the magma and mantle fO2 for a number of Ni-Cu-sulfide-bearing mafic-ultramafic intrusions in the central Asian orogenic belt (CAOB), based on the olivine-spinel oxygen barometer and the newly established method of modeling the partition coefficient of V between olivine and melt. The results show that the high magma fO2 for the intrusions in the CAOB is likely related to the fractionation of hydrous magmas derived from slightly oxidized metasomatized mantle. This may be a fundamental feature to distinguish the magmas produced in the subduction zones from those in the mid-ocean ridges. They also discussed the relationship between the redox conditions of basaltic magmas and formations of Ni-Cu sulfide deposits in various settings, and proposed that the high fO2 of mantle-derived magmas may be able to dissolve enough mantle-derived sulfur to form economic Ni-Cu sulfide deposits in convergent margin settings.
Investigation of the crystal structure of low water content hydrous olivine to 29.9 GPa: a high-pressure single-crystal X-ray diffraction study
https://doi.org/10.2138/am-2020-7444Xu et al. conducted high-pressure single-crystal synchrotron X-ray diffraction experiments on a synthetic low water content hydrous Mg-rich olivine (Fo95; 1538 ppm water) to determine the effects of minor hydration associated with the Si sites on the structural evolution under compression. Previous studies on hydrous olivine indicated that the incorporation of water affects the structure of olivine at high pressure; however, the water contents of the olivine are much higher than expected in natural mantle olivine. Field observation and petrological experiments suggest that the water storage capability of olivine in peridotite in the upper mantle is lower than 2000 ppm. In this study, the authors investigated the structural evolution of a hydrous olivine whose water content is more accessible in natural olivine. The results indicate that low water content (less than 2000 ppm) has negligible effects on the EoS of olivine, though the incorporation of water softens the Si-O1 and Si-O2 bonds.
Ferric-ferrous iron ratios of experimental majoritic garnet and clinopyroxene as a function of oxygen fugacity
https://doi.org/10.2138/am-2020-7265Rzehak et al. compared the Fe3+/ΣFe contents of experimental majorites and clinopyroxenes at 10 GPa with two analytical methods: EPMA flank and TEM-EELS analysis on the same samples. They used a FeO content of 8 wt% that is considered to be the natural bulk FeO content of the Earth's upper mantle. Previous studies have used much higher FeO contents (~25 wt%) to facilitate Fe3+/ΣFe measurements. Although results from the flank method are systematically lower than the EELS measurements, Fe3+/ΣFe obtained with both methods agree well within 2θ errors. The authors used their experimental results to compare four different geobarometers by Collerson et al. (2010), Wijbrans et al. (2016), Beyer and Frost (2017), and Tao et al. (2018) and found that all of them are recommendable for different settings.
The origin of Ti-oxide minerals below and within the eastern Athabasca Basin, Canada
https://doi.org/10.2138/am-2020-7235Titanium oxides along a major fault within and below the eastern Athabasca Basin record a protracted geological history of the area. Early rutile records regional metamorphism of the basement and reducing hydrothermal activity prior to the deposition of the Athabasca sandstones. Anatase in the sandstones and basement formed from oxidizing, acidic basinal fluids. Anatase in the basement records the incursion of basin fluids into the basement, which marks the onset of hydrothermal activity related to the formation of unconformity-type uranium deposits. This study by Adlakha et al. confirms that Ti-oxides are useful in unraveling the geological history of an area that underwent prolonged hydrothermal alteration.
Partition behavior of platinum-group elements during the segregation of arsenide melts from sulfide magma
https://doi.org/10.2138/am-2020-7477Pina et al. determined experimentally the partition behavior of platinum group elements (PGE) between arsenide and sulfide melt when an arsenide melt segregates by immiscibility from a sulfide melt after As oversaturation. The results show that PGE are strongly compatible into arsenide melt supporting empirical observations in several Ni-Cu-PGE sulfide deposits where PGE occur preferentially concentrated in the most As rich zones. The implications for exploration are considerable: the formation of arsenide melts in PGE-bearing natural sulfide systems can lead to the formation of As-PGE-rich orebodies associated with S-rich and As-poor mineralized zones.
Vapor-bubble growth in olivine-hosted melt inclusions https://doi.org/10.2138/am-2020-7377Olivine-hosted melt inclusions (MI) are the best tool for studying magmatic volatiles. Recent studies have shown that vapor bubbles, commonly found in MI, contain up to ~90% of entrapped CO2 contents. This revelation has put much of the last 30 years of MI work into question because many studies required knowledge of entrapped CO2 contents but did not account for bubble growth. CO2-reconstruction methods exist, but their accuracy has not been tested. Rasmussen et al. describe bubble growth, evaluate existing corrections, and develop improved experimental and computational (MIMiC program) methods.
November 2020
Parageneses of TiB2 in corundum xenoliths from Mt Carmel, Israel: Siderophile behavior of Boron under reducing conditions
https://doi.org/10.2138/am-2020-7375Natural titanium diboride (TiB2) occurs rarely on Earth, but is widespread in mantle-derived aggregates of corundum found in the volcanic ash of small Cretaceous volcanoes on Mt Carmel, Israel. Petrographic studies by Griffin et al. show that the highly reduced igneous melts trapped in these aggregates during their growth later separated into immiscible metallic (Fe-Ti-Si) and silicate (Ca-Mg-Al-Si-O) melts. TiB2 crystallized mainly from the metallic melts, and less commonly from the silicate melts. This indicates that boron, though usually a lithophile element, becomes a strongly siderophile element under reducing conditions, like those expected in the deep mantle. Metallic melts in the mantle may be the major reservoir for boron below the crust.
Crystal structure and Raman spectroscopic studies of OH stretching vibrations in Zn-rich fluor-elbaite
https://doi.org/10.2138/am-2020-7360Pieczka et al. present results of their study of OH stretching vibrations in the Raman spectrum of Zn-rich fluor-elbaite. Deconvolution of the bands provides new insights into crystal chemical and structural details of the studied crystals, even at the nano-scale, and direct evaluation of Li and OH (Li2O and H2O) concentrations, two components not measured in microprobe analysis and usually only calculated.
Crystal structure of Ag-exchanged levyne intergrown with erionite: single crystal X-ray diffraction and molecular dynamics simulations
https://doi.org/10.2138/am-2020-7500Cametti and Churakov report the crystal structure characterization of a Ag-exchanged levyne intergrown with erionite. Ag-exchanged zeolites are particularly interesting due to their improved catalytic, photocatalytic, adsorption, and luminescent properties. To resolve the extreme disorder of Ag ions within the zeolitc pores we combined experimental (X-ray diffraction) and theoretical approachs (Molecular dynamics).
Br diffusion in phonolitic melts: Comparison with fluorine and chlorine diffusion
https://doi.org/10.2138/am-2020-7372Balcone-Boissard et al. investigated the diffusion of Br in phonolitic melts with differing Na/K ratios, which had previously been used to study Cl and F diffusion, and compared their results to published studies of Br diffusion in magmatic melts. Br diffusion may be affected by the Na/K ratio of phonolitic melts, as previously seen for Cl, but not for F, diffusion. Similarly to noble gases, halogen diffusivity at a given temperature in the phonolitic melts appears related to the ionic porosity of the silicate structure, and Br diffusion appears to be at least partially decoupled from melt viscosity.
Crystal-chemistry and microfeatures in gadolinite imprinted by pegmatite formation and alteration evolution
https://doi.org/10.2138/am-2020-7355Alteration patterns observed by Tomašić et al. in gadolinite from pegmatites of southern Norway point to late magmatic and metasomatic changes of local mineral chemistry. The general alteration path suggests a shift to a more Ca-rich mineral chemistry within the gadolinite group. The changes induced by alteration mechanisms are related to the metamictization-inherited structural properties of gadolinite, thus promoting gadolinite as one indicator of late magmatic processes in these pegmatite systems.
A new occurrence of corundum in eucrite and its significance
https://doi.org/10.2138/am-2020-7361Corundum is an important indicator of Al-rich and Si-poor regions and/or lithologies. It is rarely observed in samples from extraterrestrial differentiated celestial bodies, although shock-induced corundum has recently reported by Li et al. In this study, Li et al. report the presence of non-shock-induced corundum in a eucrite. The petrographic texture of corundum and its associated pyroxenes and their compositions indicate that corundum has an indigenous origin and was captured during the ascent of a basaltic melt. Its presence suggests a hidden Al-rich and Si-poor region or lithology in the interior of Vesta and further implies that this asteroid’s internal evolution could be much more complicated than previously thought.
Zircon survival in shallow asthenosphere and deep lithosphere
https://doi.org/10.2138/am-2020-7402Borisova et al. experimentally investigated the behavior of zircon in mafic/ultramafic melts to predict zircon survival in the terrestrial lithosphere and shallow asthenosphere. They performed high-temperature experiments on zircon dissolution in natural mid-ocean ridge basaltic and synthetic haplobasaltic melts at temperatures of 1250 to 1300 degrees Celsius and pressures from 0.1 MPa to 0.7 GPa coupled with electron probe microanalyses of the experimental products. For the first time, the zirconium diffusion coefficient (2.87 x 10-12 m2/s) at 1300{degree sign}C and 0.5 GPa pressure in a natural tholeiitic basaltic melt has been estimated. The experimental data raise questions about the origin of zircons in mafic and ultramafic rocks, in particular, in shallow oceanic asthenosphere and deep lithosphere, as well as the meaning of the zircon-based ages estimated from these minerals. Large zircon megacrysts in kimberlites, peridotites, alkali basalts, and other magmas suggest the fast transport and short interaction durations between zircon and melt.
Reconsidering initial Pb in titanite in the context of in situ dating
https://doi.org/10.2138/am-2020-7274The mineral titanite has become a popular U-Pb geochronometer, but determination of dates requires a correction for non-radiogenic (initial) Pb. This contribution by Bonamici and Blum reviews different methods for handling initial Pb in titanite, specifically when U-Pb data are collected by in situ mass spectrometry techniques that sample small domains within grains. Their results show that initial Pb is heterogeneous both between and within the studied titanite grains. In addition, analysis of U-Pb data patterns suggests that some titanite grains have lost Pb by diffusion during a high-temperature metamorphic event. They demonstrate that even when U-Pb data are scattered and precise ages cannot be determined with in situ techniques, analysis of U-Pb data patterns can provide information about the processes that caused the scatter.
Solubility of Na2SO4 in silica-saturated solutions: Implications for REE mineralization
https://doi.org/10.2138/am-2020-7470The solubility of Na2SO4 is traditionally considered to display retrograde behavior in aqueous solutions. Chen et al. show experimentally that the solubility of Na2SO4 changes from retrograde to prograde in the presence of silica. The authors show that sulfate-rich fluids are capable of transporting large quantities of REE’s and that mass balance calculations demonstrate that hidden, deep-seated magma chambers are not necessary for the Maoniuping carbonatite-related rare earth element (REE) deposit (SW China).
Vanadium micro-XANES determination of oxygen fugacity in olivine-hosted glass inclusion and groundmass glasses of martian primitive shergottite Yamato 980459
https://doi.org/10.2138/am-2020-7321Nakada et al. focuses on the oxidation states of redox-sensitive elements in an olivine-hosted glass inclusion and groundmass glasses in a Martian meteorite; these glass phases reflect the earliest and latest stages of formation of basaltic rocks on Mars. The study shows that the oxygen fugacity (fO2) was almost equal to the Iron-Wüstite (IW) buffer (IW-0.07{plus minus}0.32) for the glass inclusion, whereas the groundmass glass is 0.9 log units more oxidized than the IW buffer (IW+0.93{plus minus}0.56). This fact suggests that the fO2 of the parent magma of Yamato 980459, which is believed to have evolved in a closed system, increased during magma ascent and emplacement. Our findings show that the µ-XANES technique is useful to investigate the redox conditions of volcanic rocks, when they do not possess mineral phases applicable to conventional oxybarometers, and surely contributes to better understanding of the redox evolution in the Martian interior.
Donwilhelmsite, [CaAl4Si2O11], a new lunar high-pressure Ca-Al-silicate with relevance for subducted terrestrial sediments
https://doi.org/10.2138/am-2020-7393Donwilhelmsite (IMA 2018-113) is a new lunar high-pressure mineral identical to the calcium aluminum silicate, CAS, phase first reported from static pressure experiments. This paper by Fritz et al. details the first extraterrestrial mineral with a crystal structure solved by three dimension electron diffraction (3D ED). The name honors the lunar geologist Don E. Wilhelms. In the feldspathic lunar meteorite Oued Awlitis 001, donwilhelmsite crystallized as micrometer-sized needles in shock melt pockets at high-pressure and temperature conditions mimicking the conditions in the Earth's mantle. On Earth, donwilhelmsite is an important mineral in continentally derived sediments subducted into the deep mantle (460 to 700 km) and likely associated with the enriched mantle components EM1 and EM2.
Magnetite texture and trace-element geochemistry fingerprint of pulsed mineralization in the Xinqiao Cu-Fe-Au deposit, Eastern China
https://doi.org/10.2138/am-2020-7414Zhang et al. use magnetite to investigate the Cu-Fe-Au mineralization of the Xinqiao ore deposit. They find 6 different generations of magnetite, including veins that formed later than pyrite and are genetically linked to elevated fO2 resulting from fracturing. Furthermore, the magnetite textures and chemistry imply that multiple pulses of fluids from a magmatic-hydrothermal system played an important role in the generation of the ore deposit.
Magmatic haggertyite in olivine lamproites of the West Kimberley region, Western Australia
https://doi.org/10.2138/am-2020-7456Jaques et al. report the first occurrence of the alkali titanate mineral haggertyite (BaTi5Fe6MgO19) as a magmatic phase in two individual olivine lamproite bodies from the classic West Kimberley lamproite province in Western Australia. This is only the second report of haggertyite. In the original description of haggertyite (American Mineralogist 1996) from the Prairie Creek lamproite, the haggertyite was inferred to be of metasomatic origin. The haggertyite in the West Kimberley lamproites varies in composition (Fe, Ti) and in the calculated Fe3+ and Fe2+ depending on the prevailing fO2 as a result of the coupled substitution between Ti + Fe2+ on one side of the reaction and and 2 Fe3+ On the other. They show that the haggertyite crystallized at relatively low temperatures (650 to 800 degrees Celsius) and that the haggertyite in one of the lamproite pipes crystallized under increasing fO2 conditions whereas in the other it formed under more reducing conditions and decreasing fO2. Jaques et al. present major and trace element analyses of the haggertyite and co-existing titanate and alkali titanate minerals. Their new data show that haggertyite is not an isolated occurrence, but one of an increasing number of new minerals in upper mantle rocks, and volcanics derived from the upper mantle, hosting large-ion-lithophile and high field strength cations. They suggest that haggertyite may be more widespread than thought, perhaps having been overlooked or misidentified.
Trace elements in sulfides from the Maozu Pb-Zn deposit, Yunnan Province, China: Implications for trace element incorporation mechanisms and ore genesis
https://doi.org/10.2138/am-2020-6950To understand the genesis of Pb-Zn deposits in the Sichuan-Yunnan-Guizhou Metallogenic Province (SYGMP), the trace elements in different sulfides (sphalerite and galena) from the Maozu Pb-Zn deposit, which is a representative Pb-Zn deposit in the SYGMP, were analyzed by Li et al. via LA-ICP-MS. Compared with trace elements in sulfides from the different genetic types of Pb-Zn deposits, the Maozu Pb-Zn deposit is characterized by enrichment of Ge, Fe, Mn, and Co in sphalerite and Ag, Sb, Cd, and Se in galena, which is similar to the composition of typical Mississippi Valley Type (MVT) deposits and different from those of SEDEX, VHMS, and skarn type Pb-Zn deposits, suggesting that the Maozu deposit is a MVT deposit.
New pressure-induced phase transition to Co2Si-type Fe2P
https://doi.org/10.2138/am-2020-7574The phase relations and elastic properties of Fe alloys with light elements under relevant high-pressure and high-temperature conditions are the keys to understanding the nature of planetary metallic cores. Phosphorous is one of the candidates for core light elements. Here, Nakajima et al. discovered a new phase transition in Fe2P from Co2P-type (C23) to Co2Si-type (C37) structure at 42 GPa based on in-situ high pressure-high temperature X-ray diffraction measurements. The results indicate that the new C37-type Fe2P phase can be stabilized as the most iron-rich phosphide phase under planetary core conditions where the pressure is higher than 42 GPa. Moreover, the present study reveals that (Fe,Ni)2(S,Si,P) could have wide solid solution and constitute planetary cores.
Effects of small crystallite size on the thermal infrared (vibrational) spectra of minerals
https://doi.org/10.2138/am-2020-7602The infrared spectra of minerals change when their crystallite sizes approach the wavelength of light. Hamilton et al. show that these changes can be used to recognize small crystallite sizes in samples lacking other indicators of this characteristic. Such small crystallite sizes are commonly associated with abbreviated crystal growth and reflect a potentially important aspect of the sample's geologic history.
October 2020
The effects of solid-solid phase equilibria on the oxygen fugacity of the upper mantle
https://doi.org/10.2138/am-2020-7162Terrestrial magmas and peridotites display large variations in oxygen fugacity, typically attributed to differences in the redox state of multivalent elements in the mantle or to processes acting on segregated melts. Using thermodynamic models, Stolper et al. show that the oxygen fugacity of subsolidus mantle peridotite of a fixed composition can vary by 1.5 log units in the upper ~100 km of the mantle due to the same reactions that produce transitions from plagioclase-spinel-garnet lherzolite and variations in Al in pyroxenes. These effects of phase equilibria on peridotite oxygen fugacity are likely to be superimposed on variations due to bulk composition and should be considered in efforts to understand variations in the oxygen fugacities of magmas and their mantle sources.
Structural and spectroscopic study of the kieserite-dwornikite solid solution series, at ambient and low temperatures, with cosmochemical implications for icy moons and Mars
https://doi.org/10.2138/am-2020-7287Talla et al. present new detailed data on the behavior of the kieserite-Ni-kieserite solid solution series. The study documents changes in the crystal structure along the binary join, as well as variations in the IR and Raman spectral band positions in relation to the changing chemical and structural properties. These sulfate phases are of cosmochemical importance, as they occur in considerable amounts on some planetary objects in our solar system.
Mineral compositions and thermobarometry of basalts and boninites recovered during IODP Expedition 352 to the Bonin forearc
https://doi.org/10.2138/am-2020-6640IODP Expedition 352 to the Bonin forearc recovered tholeiitic forearc basalt (FAB) and slightly younger basaltic, low-Si and high-Si boninite. Using mineral compositions and thermobarometry, Whattam et al. determined the conditions of crystal growth and differentiation for Expedition 352 lavas and compared these conditions with those recorded in lavas from mid-ocean ridges, forearcs, and ophiolites.
An evolutionary system of mineralogy, Part II: Interstellar and solar nebula primary condensation mineralogy
https://doi.org/10.2138/am-2020-7447The evolutionary system of mineralogy relies on varied physical and chemical attributes, including trace elements, isotopes, solid and fluid inclusions, and other information-rich characteristics, to understand processes of mineral formation and to place natural condensed phases in the deep-time context of planetary evolution. This paper by Hazen and Morrison considers the formation of primary crystalline and amorphous phases by condensation in interstellar molecular clouds and the earliest period of solar nebula evolution-environments that increased mineralogical diversity and distribution prior to the accretion of planetesimals greater than 4.5 billion years ago.
Swelling capacity of mixed talc-like/stevensite layers in white/green clay infillings ('deweylite'/'garnierite') found in serpentine veins of faulted peridotites (New Caledonia)
https://doi.org/10.2138/am-2020-6984Fonteneau et al. describe the occurrence of stevensite in vein-infillings of reactivated faults from peridotitic formations, New Caledonia. Five samples of white (deweylite) and bluish green (garnierite) clay infillings were selected from a large set of vein infillings to investigate their swelling ability and crystal chemistry. They belong to the ultimate phases of clay infillings, which are predominantly made of 2:1 layer silicates. Different treatments were used to link the gradual increase of the swelling capacity to charge deficiency and structural defects in mixed S and TL phases.
Experimental observations of TiO2 activity in rutile-undersaturated melts
https://doi.org/10.2138/am-2020-7391Over the past decade, trace-element thermobarometry (e.g., Ti-in-quartz, Ti-in-zircon) has emerged as a powerful tool to determine the thermal and barometric histories of igneous and metamorphic rocks. In many instances, the application of these tools requires an accurate estimate of Ti activity when rutile is not present. In this study, Ackerson and Mysen utilized two methods (a rutile-saturation model and a Ti-in-tridymite solubility calibration) to calculate Ti activity in rutile-undersaturated melts, and demonstrate that rutile-saturation model estimates consistently over-predict Ti activity. When applied to natural systems, this over-prediction will lead to an under-estimation of magmatic temperatures.
Direct evidence for the source of uranium in the Baiyanghe deposit from accessory mineral alteration in the Yangzhuang granite porphyry, Xinjiang Province, Northwest China
https://doi.org/10.2138/am-2020-7383Zhang et al. provide new insights into ore sources and enrichment processes of post-magmatic hydrothermal uranium deposits. This study also provides an example illustrating the application of primary versus altered U-bearing mineral-assemblages and their trace element abundances and textures to trace the source of uranium for uranium deposits. The results highlight the potential of element mapping in obtaining direct evidence for uranium leaching from source rocks and in tracing the source of uranium in ore deposits.
Extraction of high-silica granites from an upper crustal magma reservoir: insights from the Narusongduo magmatic system, Gangdese arc
https://doi.org/10.2138/am-2020-7369Although the magma dynamics for the genesis of crystal-poor high-SiO2 rhyolites has been a subject of many studies, the dynamics for the formation of high-silica granites are poorly understood. Yang et al. provide a scarce case that kilometer-scale high-silica granite bodies are the extracted products from a shallow magma reservoir. These high-silica granites can be regarded as failed eruptions of high-SiO2 rhyolites. The work addresses a broad range of issues concerning silicic magmatism, such as the behaviors of shallow magmatic systems, the volcanic-plutonic connections, and, particularly, the capability and efficiency of crystal-melt separation in upper crustal reservoirs.
Synthesis and crystal structure of Pb-dominant tourmaline
https://doi.org/10.2138/am-2020-7457Vereshchagin et al. synthesized a Pb-dominant tourmaline (up to 14.7 wt% PbO) at 700 °C and 200 MPa using a hydrothermal method. Structural analysis shows that tourmaline can incorporate significant amounts of Pb2+ (up to 0.71 apfu) in its X-site, which means that natural Pb-rich tourmalines belong to a Ca-dominant group. Similarities between (1) the paragenesis of Minh Tien tourmaline and (2) the observed, final experimental phase assemblage indicate comparable P-T conditions of formation.
Element loss to platinum capsules in high-temperature-pressure experiments
https://doi.org/10.2138/am-2020-7580Wang et al. investigate the loss behavior of 45 elements in high-temperature-pressure experiments and find (1)15 elements including V, Cr, Mn, Fe, Co, Ni, Cu, Zn, Ga, Ge, Cd, In, Sn, W, and Mo lose significantly from the sample by alloying with Pt at reducing condition; (2) Graphite- and Re-lined Pt capsules can prevent the loss of V, Cr, Mn, Fe, Zn, Ga, Ge, Cd, In, Sn, W, and Mo, but can barely reduce the loss of Ni and Cu; (3) Element loss can be reduced effectively at the oxidizing condition, and all the elements except Cu are retained at Ru-RuO2 buffered condition. The results provide several viable capsule assemblies that are capable of preventing or reducing element loss, which may prove useful in determining precise phase diagrams and accurate partition coefficients in HTP experiments.
Discussion
https://doi.org/10.2138/am-2020-7267Reply
https://doi.org/10.2138/am-2020-7616CCBYNew Mineral Names
https://doi.org/10.2138/am-2020-NMN1051022September 2020
How American Mineralogist and the Mineralogical Society of America influenced a career in mineralogy, petrology, and plate pushing, and thoughts on mineralogy’s future role
https://doi.org/10.2138/am-2020-7382This reflection by W.G. Ernst was a part of the recent yearlong centennial celebration of the MSA that took place at the 2019 GSA annual meeting in Phoenix. One day’s activities featured invited talks by extant MSA presidents. Most speakers described exciting new mineralogic studies in progress, whereas Dr. Ernst took a “walk down memory lane” -- a chronicle of 65 years of scientific studies and a few lessons learned from them, as well as concerns regarding the future habitability of the Earth. Dr. Ernst’s goals involved the integration of mineralogy, petrology, and geochemistry with regional geology and plate tectonics.
Petrographic and spectral study of hydrothermal mineralization in drill core from Hawaii
https://doi.org/10.2138/am-2020-7125This is the first analysis of alteration in basaltic materials obtained from the subsurface that can provide a unique analog for alteration environments on Mars. The paper by Calvin et al. describes the initial analysis of spectra taken of drill core in the field used to investigate a set of cut sections that represent the mineralogical diversity present. Petrography and infrared spectroscopy identify similar minerals and show that field spectra can be a useful reconnaissance tool for subsequent higher resolution and more time-consuming analysis in the lab.
Characterizing low-temperature aqueous alteration of Mars-analog basalts from Mauna Kea at multiple scales
https://doi.org/10.2138/am-2020-7131Rasmussen et al. characterized the low-temperature aqueous alteration of core samples documenting the shield-building phase of Mauna Kea’s formation. Trioctahedral Mg-Fe smectites and multiple zeolites make up the bulk of the alteration mineralogy, providing an opportunity to spectrally characterize these minerals in geologically realistic mixtures. Spectral characterization of these minerals and understanding their environment of formation are fundamental in the pinpointing of Martian alteration conditions.
Archean to Paleoproterozoic seawater halogen ratios recorded by fluid inclusions in chert and hydrothermal quartz
https://doi.org/10.2138/am-2020-7238Halogens are critical elements for sustaining life on Earth and control the salinity of the oceans. Changes in past ocean halogen compositions are largely unknown, especially for the least abundant halogen, iodine. Burgess et al. determine Cl, Br, and I abundances in chert from the 2.5 Ga Hamersley Banded Iron Formation and hydrothermal quartz from the 3.5 North Pole area, both in Australia. Comparable results from both imply that ancient oceans had Br/Cl and I/Cl about 30% and 30 times higher, respectively, relative to modern seawater. The higher I/Cl indicates a smaller organic reservoir in the ancient oceans.
Metasomatism-controlled hydrogen distribution in the Spitsbergen upper mantle
https://doi.org/10.2138/am-2020-7290This work by Tang et al. reports the hydrogen concentrations in olivine and pyroxene of Spitsbergen mantle xenoliths. The correlations between hydrogen and incompatible trace elements in xenoliths suggest that hydrogen distribution in the Spitsbergen upper mantle is controlled by metasomatism. The hydration of mantle was achieved by two different processes in this metasomatic event. Furthermore, the metasomatic melt could have derived from an OIB-type source, consistent with the Sr-Nd isotope compositions of the xenoliths.
Phase transformation of hydrous ringwoodite to the lower-mantle phases and the formation of dense hydrous silica
https://doi.org/10.2138/am-2020-7261Chen et al. performed new high-pressure experiments that showed that hydrous ringwoodite can break down to form bridgmanite, ferropericlase, and a dense hydrous silica phase. The new observation of hydrous silica formation may explain the coexistence of stishovite and ferropericlase inclusions in lower-mantle diamonds, which cannot be explained by the equilibrium phase equilibria of dry phases.
Density and sound velocity of liquid Fe-S alloys at Earth’s outer core conditions
https://doi.org/10.2138/am-2020-7349The Earth’s outer core is mostly composed of liquid iron (Fe) alloy with a small amount of light elements, such as S, O, C, Si, and H. Among these light elements, S stands as a key candidate element, but its concentration is very controversial. In order to constrain the S concentration, Fu et al. developed a thermal equation of state for liquid Fe-S alloys by first-principles molecular dynamics simulations. Comparing with Preliminary Reference Earth Model, they clarify that the S concentration range is from 10 ~ 14 wt%, assuming S is the only light element. Taking into account the geophysical and geochemical constraints, they propose the outer core contains no more than 3.5 wt% S, 2.5 wt% O, or 3.8 wt% Si.
Some geometrical properties of fission-track-surface intersections in apatite
https://doi.org/10.2138/am-2020-7271Apatite fission-track dating conventionally relies on counting the etched damage trails from uranium fission in apatite prism faces, which are considered to have high counting efficiencies. This presents a severe practical limitation. The work by Jonckheere et al. aims to lay the basis for extending fission-track counts to other apatite faces. It presents a model calculation of the orientations, lengths, and widths of the intersections of etched fission tracks with the prism and non-prism faces of apatite. The model predictions are consistent with extensive measurements. Their implications section discusses future practical uses of such measurements. Their contribution is a step toward understanding which tracks are counted and which are not in fission-track dating, and toward using other faces than prism faces for dating.
Thermal equation of state of post-aragonite CaCO3-Pmmn
https://doi.org/10.2138/am-2020-7279Lv et al. investigated the stability and physical properties of post-aragonite CaCO3-Pmmn, a high-pressure polymorph of CaCO3, at lower mantle pressure and temperature conditions. Using synchrotron X-ray diffraction measurements in a laser-heated diamond-anvil cell, they confirm the stability of CaCO3-Pmmn at pressures and temperatures corresponding to the middle of the lower mantle, and provide the first characterization of the thermal equation of state of this phase. The newly determined thermodynamic parameters of CaCO3-Pmmn are used to quantitatively model the density and seismic velocity of CaCO3 and carbonated eclogite. With the assumption that carbonates are homogeneously mixed into the slab, this work determines that the presence of carbonates in the subducted slab is unlikely to be detectable through seismic observations, and the buoyancy of carbonates will have a negligible effect on slab dynamics.
Structure of NaFeSiO4, NaFeSi2O6, and NaFeSi3O8 glasses and glass-ceramics
https://doi.org/10.2138/am-2020-7285This paper by Ahmadzadeh et al. studies the structure of melts of geologically relevant Na-Fe-silicates. Both as-quenched and heat-treated powders of three Na-Fe-silicates with compositions analogous to well-known aluminosilicate minerals were studied. The main focus of the paper is on the role of Fe and the effects of increasing Si/Fe ratio. It is found that most of the Fe occur as 4-coordinated Fe3+ in the glasses. The crystallization behavior upon both quenching and heat-treating varies depending on Si/Fe ratio. The correlation between Si/Fe ratio and Fe structural role, as well as its oxidation state in such glasses, is important for geosciences and nuclear waste management. The study of these simplified compositions can provide fundamental insight into the physical behavior of silicate melts as well as high-Fe nuclear waste glass crystallization, which is related to their aqueous alteration behavior.
Raman spectroscopic studies of OH stretching vibration in Mn-rich apatites: A structural approach
https://doi.org/10.2138/am-2020-7324In this manuscript, Pieczka et al. present results of their studies of the OH-stretching vibration band in Raman spectra of Mn-bearing apatites. Deconvolution of the complex band gives an opportunity to gain insight into the distribution of Mn among the M1 and M2 structural sites in the apatite structure. Thus, the data can, at least partly, verify (or complete) structural data obtained by single-crystal X-ray diffraction and structure refinement, and in the case of very small crystals or their zoned texture, the described method may be the only tool that is able to yield structural data.
Characterization of modified mineral waste material adsorbent as affected by thermal treatment for optimizing its adsorption performances
https://doi.org/10.2138/am-2020-7227In the paper by Su et al., physiochemical properties of mineral waste material (MMWM) can be modified by thermal treatment. Increase in the adsorption capacity of MMWM is attributed to the enlarged surface area and disappearance of -OH groups. Adsorption of Pb on MMWM could be assigned to monolayer coverage and chemisorption. Thermally treated MMWM is a good adsorbent for Pb in solution.
Morin-type transition in 5C pyrrhotite
https://doi.org/10.2138/am-2020-7266Haines et al. report the discovery of a low-temperature spin-flop transition in 5C pyrrhotite at ~155 K that is similar to those seen in hematite at 260 K and FeS (troilite) at 440 K. The 5C crystal was produced by annealing a 4C pyrrhotite crystal at 875 K to produce a change in the vacancy-ordering scheme that developed during cooling. The 5C structure is confirmed by single-crystal x-ray diffraction and the stoichiometry and homogeneity by electron microprobe and SEM BSE mapping. Resonant ultrasound spectroscopy, heat capacity, and magnetization measurements from room temperature down to 2 K are reported. The transition is marked by a steep change in elastic properties at the transition temperature, a peak in the heat capacity, and weak anomalies in measurements of magnetization. Magnetic hysteresis loops and comparison with the magnetic properties of 4C pyrrhotite suggest that the transition involves a change in orientation of moments between two different antiferromagnetic structures, perpendicular to the crystallographic c-axis at high temperatures and parallel to the crystallographic c-axis at low temperatures. The proposed structures are consistent with a group theoretical treatment that also predicts a first-order transition between the magnetic structures.
The formation of marine red beds and iron cycling on the Mesoproterozoic North China Platform
https://doi.org/10.2138/am-2020-7406Tang et al. makes three points in this paper. First, marine red beds (MRBs) have been identified in the 1.4 Ga Xiamaling Formation, North China. Second, continentally sourced iron reactivated by dissimilatory iron reduction and distal hydrothermal fluids have supplied Fe(II) for the MRBs Third, Xiamaling MRBs record a moderate oxygenation event and a long-lasting ferruginous deep water mass.
A multi-methodological study of kernite, a mineral commodity of boron
https://doi.org/10.2138/am-2020-7433Gatta et al. investigated the chemical composition and the crystal structure of kernite [Na2B4O6(OH)2·3H2O], one of the most important mineral commodities of B, by a series of analytical techniques, including single-crystal neutron diffraction. The general experimental formula of the kernite sample used in this study is Na1.99B3.99O6(OH)2·3.01H2O; the fraction of other elements (measured for more than 50 elements) is, overall, insignificant (<100 wt ppm); excluding B, kernite does not act as geochemical trap of other technologically-relevant elements (e.g., Li, Be, or REE). The H-bonding network in the structure of kernite is complex, pervasive, and plays a primary role on its structural stability. The potential utilizations of kernite, as a source of B (B2O3 ~ 50 wt%), are discussed on the basis of the experimental findings of this study.
Si-rich Mg-sursassite Mg4Al5Si7O23(OH)5 with octahedrally coordinated Si
https://doi.org/10.2138/am-2020-7533The crystal structure of a new high-pressure hydrous phase, Si-rich Mg-sursassite, of composition Mg4Al5Si7O23(OH)5 that was produced by sub-solidus reaction at 24 GPa and 1400 degrees in an experiment using a model sedimentary bulk composition has been determined by Bindi et al. using single-crystal X-ray diffraction. The reactions defining the stability of Si-rich Mg-sursassite are unknown, but are likely to be fundamentally different from those of Mg-sursassite, and involve other ultra-high-pressure dense structures such as Phase D, rather than Phase A.
Inherited Eocene magmatic tourmaline captured by the Miocene Himalayan leucogranites
https://doi.org/10.2138/am-2020-7608Tourmaline, which is very common in the Himalayan leucogranites and typically the dominant reservoir of B in the rocks, is stable in various P-T-X conditions and could record the physical and chemical conditions of its formation. Thus, tourmaline chemistry has been frequently used to investigate the petrogenesis of its host magmatic rocks. However, these studies relied on the assumption that the tourmalines formed cogenetically with their magmatic host rocks, as is widely interpreted in most of the global tourmaline occurrences. Han et al. applied the 40Ar/39Ar dating method to coarse-grained tourmalines from the Miocene Cuonadong leucogranite in the Tethyan Himalayan, Southern Tibet, which yielded Eocene ages. The results, together with textural observations and geochemical studies, clearly suggest that the coarse-grained tourmalines found in pegmatites were inherited. The identification of inherited tourmalines not only contributes new insights into the Himalayan collisional orogeny, but also provides constraints for the application of tourmaline chemistry to petrological studies. The capture of Eocene tourmaline by the Miocene leucogranites at Cuonadong suggests that the crust-derived Eocene magmatism may have occurred in the southern Tethyan Himalaya together. Identification of the inherited magmatic tourmaline, although not common, challenges the current application of tourmaline chemistry to the investigation of magmatic-hydrothermal systems.
Book Review: Geology of the Lassen Country: The Geologic Story of Lassen Volcanic National Park and Vicinity. (2019)
https://doi.org/10.2138/am-2020-B105921Memorial of Don Bloss
https://doi.org/10.2138/am-2020-m1002August 2020
Are quasicrystals really so rare in the Universe?
https://doi.org/10.2138/am-2020-7519Until 2009, the only known quasicrystals were synthetic, formed in the laboratory under highly controlled conditions. Conceivably, the only quasicrystals in the Milky Way, perhaps even in the Universe, were the ones fabricated by humans, or so it seemed. Then came the report that a quasicrystal with icosahedral symmetry had been discovered inside a rock recovered from a remote stream in far eastern Russia, and later that the rock proved to be an extraterrestrial, a piece of a rare CV3 carbonaceous chondrite meteorite (known as Khatyrka) that formed 4.5 billion years ago in the presolar nebula. At present, the only known examples of natural quasicrystals are from the Khatyrka meteorite. Does that mean that quasicrystals must be extremely rare in the Universe? In this speculative essay, Bindi et al. present a number of reasons why the answer might be no. In fact, quasicrystals may prove to be among the most common minerals found in the Universe.
Reaction between Cu-bearing minerals and hydrothermal fluids
https://doi.org/10.2138/am-2020-7114Qi et al. study the reaction between native Cu and NaCl solution that leads to the coexistence of fluid inclusions and Na-bearing silicate melt inclusions. Micrometer- to submicrometer-sized cuprite (Cu2O) crystals have been observed in both types of the inclusions, and they are formed most probably due to the dissociation of CuOH. When CuO reacts with HCl and CuCl solutions, or Cu+ reacts with NaCl solution, nantokite (CuCl) has been found in the fluid inclusions that were formed due to oversaturation. Cu solubility in pure water and in 1.5 m NaCl solutions are 0.004 ± 0.002 m and 0.16 ± 0.07 m, respectively. The main responsible Cu-bearing complexes are CuOH(H2O)x in water, NaCuCl2 in NaCl solutions, and HCuCl2 in alkali-free solutions.
Evaluation and application of the quartz-inclusions-in-epidote mineral barometer
https://doi.org/10.2138/am-2020-7379Epidote is one of the most common minerals found in igneous and metamorphic rocks. This study by Cisneros et al. introduces a new barometer that has the potential to be widely used among petrologists and other geoscientists. The barometer utilizes elastic modeling of quartz inclusions in epidote to constrain the pressure conditions of epidote growth. Modeling, pressure estimates, and applications of the technique are discussed.
Let there be water: how hydration/dehydration reactions control Earth and life key properties
https://doi.org/10.2138/am-2020-7380This paper by Brovarone et al. highlights some fundamental aspects of hydration and dehydration reactions in the solid Earth, in biology, and in modern society, as well as their connections to carbon cycling on our planet.Origin of corundum within anorthite megacrysts from anorthositic amphibolites, Granulite Terrane, Southern India
https://doi.org/10.2138/am-2020-7108Karmaker et al. describe the occurrence of and offer an explanation for the genesis of corundum in anorthositic amphibolites from ~2.5 Ga old basement of the Granulite Terrane of Southern India (GTSI). The studied amphibolites from the Manavadi (MvAm) and Ayyarmalai (AyAm) localities contain anorthite lenses (An90-99) with euhedral to elliptical outline set in a finer grained matrix of calcic plagioclase (An85-90) and aluminous amphibole (pargasite-magnesiohastingsite). The lenses, interpreted as primary magmatic phenocrysts, and the matrix are both recrystallized under static conditions presumably during the regional high pressure (HP) metamorphism (~800 °C, 8 to 11 kbar) at ~2.45 Ga. Combined petrological data and computed phase relations are consistent with metasomatic growth of corundum in an open system during infiltration-driven regional metamorphism. This study thus presents a new viable mechanism for the origin of corundum in anorthositic amphibolites, and basic-ultrabasic rocks in general, which should provide new insight into lower crustal processes.
Raman spectroscopy study of manganese oxides -- tunnel structures
https://doi.org/10.2138/am-2020-7390With the comprehensive Raman database of well-characterized Mn oxide standards, reported here (and as supplementary data) by Post et al., and use of appropriate data collection conditions, micro-Raman is a powerful tool for identification and characterization of biotic and abiotic Mn oxide phases from diverse natural settings (including on other planets) and thereby can provide new insights into the roles of these phases in our environment.
Experimental constraints on the partial melting of sediment-metasomatized lithospheric mantle in subduction zones
https://doi.org/10.2138/am-2020-7403Zhang et al. performed high-pressure experiments in sediment-harzburgite systems at 1.5-2.5 GPa and 800-1300 °C to investigate the partial melting behavior of mixed sediment-harzburgite. The results demonstrate that bulk sediment diapirs, in addition to sediment melt, may be another possible mechanism to transfer material from a subducting slab to an upper mantle wedge or lithospheric mantle. On the other hand, the breakdown of phlogopite may play an important role in the mantle source that produces potassium-rich arc lavas in subduction zones.
Interlayer energy of pyrophyllite: Implications for macroscopic friction
https://doi.org/10.2138/am-2020-7333The origin of macroscopic friction of clay minerals is one of the most crucial problems to be solved in the geoscience to understand the fault behavior in the crust. Low frictional strength of clay minerals seems to originate from the crystal structure; however, the underlying physics remains unclear. Here, Sakuma et al. succeed in bridging the gap by conducting the ab initio calculations and developing a simple model of randomly oriented pyrophyllite particles. This result would be a basis for future studies on the frictional properties of clay minerals in natural faults.
Thermodynamic and thermoelastic properties of wurtzite-ZnS by Density Functional Theory
https://doi.org/10.2138/am-2020-7330Since wurtzite is an important mineral in scientific and technological fields spanning from geology/mineralogy to materials science, this work by Ulian et al. aims at providing deep insights into its thermodynamic, thermomechanic, and electronic properties at pressures up to 20 GPa and temperatures between 0 to 2000 K for various applications. Several properties, such as phonon dispersion relations, elastic and piezoelectric constants, thermodynamic and thermoelastic behaviors were calculated and reported.
The nature of Zn-phyllosilicates in the nonsulfide mina grande and cristal zinc deposits
https://doi.org/10.2138/am-2020-7140Balassone et al. conducted a detailed TEM-HRTEM and AEM study of Zn-phyllosilicates in Mina Grande and Cristal Zn-sulfide and nonsulfide deposits of Bongara district, Amazonas region, Peru. They determined the amount/mode of metal incorporation in the lattices of Zn-phyllosilicates and the relationships of natural occurring clay-rich complex associations, which can act as models for possible synthetic counterparts.
Orthovanadate wakefieldite-(Ce) in symplectites replacing vanadium-bearing omphacite in the ultra-oxidized manganese deposit of Praborna (Aosta Valley, Western Italian Alps)
https://doi.org/10.2138/am-2020-7219This study by Tumiati et al. focuses on the occurrence of Ca-bearing REE orthovanadate wakefieldite in the Italian Western Alps. The authors identified wakefieldite and refined its structure following a cutting-edge approach, using a single ~10 micrometer-sized crystal and synchrotron X-ray microdiffraction on a fragment of thin section. In addition, they performed WDS-microprobe analyses, micro-Raman spectroscopy, and thermodynamic modeling.
A simple and effective capsule sealing technique for hydrothermal experiments
https://doi.org/10.2138/am-2020-7230Capsule sealing has always been a key procedure in hydrothermal experiments to explore the composition and properties of geo-fluids and their influence on various geological processes. Previously reported capsule sealing techniques have primarily focused on either weld-sealing or cold-sealing methods, which have some disadvantages and limitations. Here, Li et al. report on a newly developed, simple, and effective capsule sealing technique incorporating operations from the cold-sealing and weld-sealing techniques. The sealed capsules are suitable not only for piston cylinders but also for multi-anvil presses and other gas-media or hydrothermal-media apparatuses, such as autoclaves and pressure vessels.
Metamorphic amphiboles in the Ironwood Iron-Formation, Gogebic Iron Range, Wisconsin
https://doi.org/10.2138/am-2020-7211This paper by Green et al. characterizes the metamorphic amphiboles in the Ironwood Iron-Formation in northern Wisconsin. The Ironwood is one of the largest iron ore resources in the U.S., but the lack of definitive, unbiased information regarding the presence of amphiboles and their potential human-health impacts has been a source of great controversy. The authors present data that will allow further mineralogical, medical, and biological work to aid in understanding the potential impacts of developing iron resources affected by metamorphism.
The chlorine-isotopic composition of lunar KREEP from magnesian-suite troctolite 76535
https://doi.org/10.2138/am-2020-7467To characterize the isotopic composition of KREEP, a primordial geochemical reservoir enriched in incompatible lithophile elements, McCubbin and Barnes conducted in-situ Cl isotopic measurements of apatite within intercumulus regions and within a holocrystalline olivine-hosted melt inclusion in magnesian-suite troctolite 76535 from Apollo 17. The isotopic composition of Cl from this reservoir indicates volatile-loss from the Moon occurred prior to 4.31 Ga.
New Mineral Names
https://doi.org/10.2138/am-2020-NMN105819July 2020
Experimental determination of solubility constant of kurnakovite
https://doi.org/10.2138/am-2020-7212Xiong et al. first highlight that the solubility constant for Kurnakovite has been experimentally determined. Second, they note that a precise model describing the interactions between borate and sulfate is established.
Elastic properties of majoritic garnet inclusions in diamonds and the seismic signature of pyroxenites in the Earth’s upper mantle
https://doi.org/10.2138/am-2020-7136CCBYKoemets et al. report seismic velocities of natural majoritie garnets and use this information to evaluate the seismic signature of a possible pyroxenitic garnet layer in Earth’s mantle.
Establishing a protocol for the selection of zircon inclusions in garnet for Raman thermobarometry
https://doi.org/10.2138/am-2020-7246Campomenosi et al. present a systematic study of zircon inclusions coupling structural and chemical information by combining a wide range of analytical techniques (Raman spectroscopy, CC-imaging, and LA-ICP-MS) to define how the degree of metamictization, their inherent structural heterogeneity, chemical zonation, and metamorphic recrystallization processes can influence the Raman spectra of zircon. The experimental results allowed them to establish a protocol devoted to the selection of reliable buried zircon inclusions to use for elastic barometry applications relying only on Raman spectroscopic measurements.
Reversely zoned plagioclase in lower crustal meta-anorthosites: An indicator of multistage fracturing and metamorphism in the lower crust
https://doi.org/10.2138/am-2020-7284Soda et al. describe the formation mechanism of reversely zoned plagioclase, which has been observed frequently in lower crustal shear zones, by studying the microstructural and chemical characteristics of plagioclase from the Eidsfjord shear zone, northern Norway. The reversely zoned plagioclase is an indicator of multistage, brittle fracturing and subsequent hydration metamorphism during exhumation, providing information relevant to understanding the deep rupture process caused by repeated seismicity alternating with aseismic creep below the seismogenic zone.
High-pressure silica phase transitions: Implications for deep mantle dynamics and silica crystallization in the protocore
https://doi.org/10.2138/am-2020-7299Das et al. improved the constraints on the pressure-temperature locations and slopes by first-principles atomistic computations of two silica phase transitions occurring at the lowermost mantle and outer core conditions: (1) beta-stishovite (CaCl2-structure) to seifertite and (2) seifertite to pyrite-structured silica. Seifertite has a wide stability range, covering the lowermost 470 km of the mantle. The resulting incremental density increase for recycled oceanic crust might help to segregate and stabilize basaltic materials in the two thermochemical piles beneath Africa and the Pacific.
Cr-Zr-Ca armalcolite in lunar rocks is loveringite: Constraints from electron backscatter diffraction
https://doi.org/10.2138/am-2020-7260The compositional and EBSD study by Zhang et al. reveals that Cr-Zr-Ca armalcolite in lunar rocks should be loveringite, which could be an important mineral indicator and REE carrier of lunar Mg-suite rocks.
Effects of composition and pressure on electronic states of iron in bridgmanite
https://doi.org/10.2138/am-2020-7309The amount, valence, spin state, and site occupancy of iron in Earth’s most abundant mineral, bridgmanite, potentially controls dynamics and physical properties of heterogeneities in the lower mantle. Electronic states of iron in bridgmanite were probed by Dorfman et al. via Mossbauer spectroscopy for a wide range of compositions. The results provide constraints on the effects of pressure and composition on spin state, valence state, and charge transfer in bridgmanite in Earth’s mantle.
Ti Diffusion in feldspar
https://doi.org/10.2138/am-2020-7272These data by Cherniak and Watson indicate that Ti diffuses more slowly than most other impurities in feldspar, including Sr. For plagioclase, there is a dependence of Ti diffusion on feldspar An content, with more anorthitic feldspars having slower Ti diffusivities. Characterization of diffusivities for this trace impurity, which is relatively slow-diffusing and ubiquitous in feldspars, has the potential to expand the scope and applicability of modeling of time-temperature conditions for crustal rocks.
Radiation-induced defects in montebrasite: An electron paramagnetic resonance study of O- hole and Ti3+ electron centers
https://doi.org/10.2138/am-2020-7168Toledo et al. detail color improvements of the rare gemstone montebrasite and related minerals that contain hydroxyl ions in their crystalline structure. From detailed analysis of the EPR angular rotation patterns, microscopic models for the O− hole and Ti3+ electron centers are presented, as well as their role in the formation of color centers is discussed and compared to other minerals.
New IR spectroscopic data for determination of water abundances in hydrous pantelleritic glasses
https://doi.org/10.2138/am-2020-7363In view of the scarcity of data on peralkaline rhyolitic compositions compared with metaluminous rhyolites, new experimental data are provided by Stabile et al. that allow the use of IR spectroscopy to measure water abundance in melt inclusions in natural glasses and water content in hydrous experimental glasses of pantelleritic composition. Such compositions, once thought to be relatively H2O-poor, may have water contents as high as 5-6 wt% H2O, which needs to be considered to better gain insights into pantelleritic origins and eruption dynamics. This is of importance for such compositions because, despite the low viscosity, they can experience vastly different eruptive styles, including Plinian events involving large magma volumes; this study can also help in eruptive process modeling.
Experimental Investigation of the effect of nickel on the electrical resistivity of Fe-Ni and Fe-Ni-S alloys under pressure
https://doi.org/10.2138/am-2020-7301It is commonly assumed that nickel does not significantly affect the chemical and physical properties of core analogues. However, the effect of Ni on electrical and thermal resistivity is not well-constrained under temperature and pressure conditions, as the very few previous electrical studies on Fe-Ni alloys were conducted either at room temperature or at atmospheric pressure. Pommier shows experimentally that at defined temperature, Fe-Ni(-S) alloys are more resistive than Fe (by a factor of ~3) and Fe-Ni alloys containing 5 and 10 wt% Ni present comparable electrical resistivity values. A comparison is made with data on pure iron and Fe-5S samples. Pommier also provides estimates of the lower and upper bounds of thermal conductivity. These estimates suggest that the thermal conductivity values used as part of recent numerical modeling of the cores of Ganymede and the Moon are too high, and thus, this paper has the potential to better constrain future thermochemical models of planetary cores. Pommier shows that a similar amount of heat is conducted at any depth along the adiabat gradient of a Fe-Ni(-S) core, whereas less heat is conducted down this gradient at shallow depth in a Ni-free core. Because variation in heat conduction is critical for driving convection, this implies that it may be easier to drive convection in a Ni-free core than in a Ni-bearing core.
An experimental approach to examine fluid-melt interaction and mineralization in rare-metal pegmatites
https://doi.org/10.2138/am-2020-7216McNeil et al. establish an experimental technique to investigate solubilities of minerals in melts via fluid-melt interactions, through the interactions of a melt enriched in HFSEs with a hydrothermal fluid enriched in a fluid mobile element. HFSE minerals that crystallize from fluid-melt interactions texturally occur as euhedral crystals as phenocrysts in glass, i.e., are purely magmatic textures. Therefore, crystallization of HFSE minerals from fluid-melt interactions in rare metal granites and pegmatite deposits may be more widespread than previously recognized. This is significant because the formation of these deposits may require magmatic-hydrothermal interaction to explain the textures present in deposits worldwide, rather than always being the result of a single melt or fluid phase.
Crystal-chemistry of sulfates from the Apuan Alps (Tuscany, Italy). VI. Tl-bearing alum-(K) and voltaite from the Fornovolasco mining complex
https://doi.org/10.2138/am-2020-7320Sulfates play an important role in determining the dispersion in the environment of acids and potentially toxic metals related to the weathering of ore deposits, coals, and mine wastes. Alum-(K) and voltaite are two common phases in sulfate assemblages, and they may be able to host high contents of the toxic element Tl. Through a multi-technique study of these minerals, Biagioni et al. found that it was possible to highlight their role as scavengers of Tl in acid mine drainage systems.
First-Principles Modeling of X-ray absorption spectra enlightens the processes of scandium sequestration by iron oxides
https://doi.org/10.2138/am-2020-7308Chassé et al. investigate the mechanisms of scandium sequestration by iron oxides combining first-principles calculations with X-ray absorption near-edge structure spectroscopy. Their results show the specificities of scandium sorption processes, explaining its concentration in iron-oxide rich surficial environment, with implications for mineral processing. This work demonstrates the relevance of the approach to study the speciation of trace metals in the environment.
Effects of the dissolution of thermal barrier coating materials on the viscosity of remelted volcanic ash
https://doi.org/10.2138/am-2020-7334The chemical interaction between remelted volcanic ash and ceramic coatings of yttria-stabilized zirconia (YSZ) and/or gadolinium zirconate (GZO) is of special importance for the design of volcanic ash melt-resistant thermal barrier coatings (TBCs) for aviation turbine technologies. Müller et al.’s investigation of the high-temperature viscosity of five volcanic ash samples of basaltic, andesitic, rhyolitic, and phonolitic compositions, each doped with 6.5 wt% YSZ or GZO, revealed a reduction of viscosity for all samples compared to their natural counterparts. With respect to thermal barrier coatings, it can be concluded that once the dissolution of the YSZ or GZO coating material in contact with a silicate melt starts, the viscosity will decrease, enabling an enhanced spreading on the surface and/or infiltration in the coating. A simple parameterization of the effects of YSZ and GZO on the viscosity of melts of volcanic ash samples can be expressed as a linear relationship. This parameterization should be employed in any future modeling of the dynamics of CMAS or natural ash melt interaction with TBCs.
New Mineral Names
https://doi.org/10.2138/am-2020-NMN105714June 2020
Halogens in amphibole and mica from mantle xenoliths: Implications for the halogen distribution and halogen budget of the metasomatized continental lithosphere
https://doi.org/10.2138/am-2020-7174Hecker et al. conducted a study on the halogen contents (F and Cl) in amphibole and phlogopite in mantle xenoliths of variable modal compositions (dunites, harzburgites, lherzolites, wehrlites, olivine websterites, websterites, and clinopyroxenites) complemented by amphibole and phlogopite megacrysts from several localities and samples from the metasomatized Finero peridotite massif in northern Italy. For comparison, the authors also analyzed amphibole from several mafic magmatic cumulates (hornblendites). Totally, data for 12 localities in Europe and Africa are presented. The data provide insight into halogen storage and redistribution in the mantle and demonstrate the importance of amphibole and mica for the total halogen budget of the lithospheric mantle and their potential role as a halogen source for mantle-derived melts. The data also record large differences in Cl concentrations (which correlate negatively with F/Cl ratios) between samples of the same region, recording desiccation processes that may cause very large variations in mantle halogen contents on a regional scale.
New insights on Br speciation in volcanic glasses and structural controls on halogens degassing
https://doi.org/10.2138/am-2020-7273Despite their relatively low concentrations compared to H2O and CO2, halogens (F, Cl, Br, and I) are considered key actors in magmatic processes, because they may have a significant effect on melt properties. Their volcanic degassing may also lead to considerable perturbations of atmospheric chemistry, with the rapid transformation of HBr to reactive BrO during explosive eruptions notably known to trigger ozone depletion on various temporal and spatial scales. While recent analytical developments enable a better detection and characterization of their concentrations in magmas and volcanic gases, current understanding of incorporation mechanisms in silicate melts is scarce and potentially limits the interpretation of elemental ratios and modeling of degassing behavior. Here, Louvel et al. take advantage of the development of high-energy resolution fluorescence detection X-ray absorption spectroscopy (HERFD-XAS) to study Br speciation in natural volcanic glasses that contain 100-3000 ppm Br. The measurements reveal that Br speciation in basalt, andesite, and rhyodacite differs significantly from that previously recorded in haplogranite laboratory analogs, with Br being incorporated in at least three distinct sites, surrounded by Na, K, or Ca. The reported structural environment for natural glasses are similar to those found for Cl in silicate and borosilicate glasses, suggesting that melt composition may not play a significant control on Cl and Br incorporation in natural melts, and further supporting their coupled degassing in volcanic systems.
Decoupled water and iron enrichments in the cratonic mantle: A study on peridotite xenoliths from Tok, SE Siberian Craton
https://doi.org/10.2138/am-2020-7316Doucet et al. provide new data of water contents of olivine and pyroxenes from peridotite xenoliths that represent the cratonic lithospheric mantle at the edge of the Siberian Craton. The peridotite xenoliths display significant iron enrichment due to complex metasomatic history that might have led to the destruction/delamination of the cratonic root beneath the Anabar shield in the southeastern part of the Siberian Craton. This study addresses the fundamental question of the role of water and iron on the stability and longevity of the cratonic lithosphere and describes, in particular, the role of “wet” and “dry” metasomatism on the physical properties of the lithospheric mantle.
Deconvolution of the composition of fine-grained pyrite in sedimentary matrix by regression of time-resolved LA-ICP-MS data
https://doi.org/10.2138/am-2020-7202Sedimentary pyrite is an important host of many trace elements. The LA-ICP-MS analysis of sedimentary pyrite by Stepanov et al. results in variable, mixed data. Pyrite composition is calculated from the mixed data using an algorithm based on linear regression. The algorithm involves segmentation of the time-resolved signal, normalization to total, calculation of the regression equations, and estimation of explanatory variable normalizing to total. Regression analysis of time-resolved mixed LA-ICP-MS data is a powerful technique for the analysis of complex substances.
A multi-method characterization of natural terrestrial birnessites
https://doi.org/10.2138/am-2020-7303Ling et al. investigate a variety of natural birnessites collected from freshwater environments. Birnessite exists in two varieties, triclinic and hexagonal birnessite, which can determine the metal cycling and redox reactions in which they are involved. Laboratory experiments suggest that biotically produced birnessite is hexagonal, although little is known about the varieties of birnessite that exist in freshwater environments. From the analyses of 11 non-marine birnessite samples, they found that the birnessite samples span the entire spectrum of triclinic to hexagonal birnessite, including intermediate structures.
REE redistributions during granite weathering: Implications for Ce anomaly as a proxy for paleoredox states
https://doi.org/10.2138/am-2020-7148Different response of Ce to redox state from those of the other light rare earth elements (LREEs) can be used to understand paleo redox states. In order to establish the possibility of using Ce anomaly as a proxy for paleo-environments, Ichimura et al. examined the mineralogical and chemical characteristics of bulk samples and REE-bearing minerals of a modern weathering profile developed on granite using a number of experimental techniques. They have classified LREE redistributions in both secondary minerals and bulk weathered samples during oxic weathering and suggested that Ce anomaly can provide useful information on paleoredox states if Ce anomalies of both bulk samples and secondary REE-bearing minerals are determined.
Formation of destinezite in relationship with the acid-sulfate alteration associated to the magnetite deposit of El Laco, Chile
https://doi.org/10.2138/am-2020-7122The giant El Laco magnetite-(apatite) deposit, Chile, has been interpreted as a complex polyphase magmatic-hydrothermal mineralization in which most of the existing magnetite and apatite formed as lava flows or (crypto)-domes. However, there are opinions that support a genesis exclusively related to an intense hydrothermal replacement of preexisting andesitic rocks. Among the geological evidences that support the first explanation has been argued the presence of unconsolidated Fe-P-oxide materials ejected as tephra (volcanic bombs) by the El Laco volcano, currently replaced by destinezite forming enigmatic lumps. However, these rare (almost) monomineralic rocks, affected by hydrothermal alteration and weathering, have received little attention. In this study, Velasco et al. use conventional geological information and mineralogical identification of the lumps, besides thermal decomposition of pure destinezite to derive its thermodynamic properties. These new data have been used to discuss the extension of fields of stability of this mineral and predict the favorable conditions for destinezite formation. Finally, they propose that quoted destinezite formed at low-temperature hydrothermal conditions, an origin very different from the traditionally proposed one for this mineral (i.e., supergene).
Implications for anisotropy at the slab–mantle interface due to Si-metasomatism
https://doi.org/10.2138/am-2020-7006Nagaya et al. carried out EBSD measurements of talc grains and obtained the crystallographic preferred orientation (CPO) of talc from talc schists formed due to Si-metasomatism by subduction zone fluids into ultramafic rocks. Talc CPO shows a strong concentration of the pole to the (001) plane, which is normal to the foliation. The strongest concentration of the [100] direction is parallel to the lineation. The combination of TEM and EBSD observations of talc grains implies that the use of EBSD orientation data of talc grains with MAD values in the range of less than 1.3° to less than 0.7° enables determination of a relatively accurate talc CPO when using the thin-section parallel to the foliation of talc schist. As with talc, CPO measurements of most clay minerals have not been reported. The methods used here may be applicable to other mechanically-weak minerals. Their calculations of seismic anisotropies based on the talc CPO obtained demonstrate that Vp and AVp of talc schist can show slower and stronger values, respectively, as compared with antigorite schist at deeper domains. Therefore, if S-waves with different ray paths through the same anisotropic domain show a larger variation in Vp values than that predicted in antigorite schist, this anisotropic domain may be composed of talc schist. The strong talc CPO and the significantly weak layer developed in the slab-mantle interface in the subduction zone can promote spatial expansion of the slip area during an earthquake in the wedge.
The occurrence, origin, and fate of water in chromitites in ophiolites
https://doi.org/10.2138/am-2020-7270Su et al. presented direct petrographic, mineralogical, and geochemical evidence confirming the occurrence, origin, and fate of water in podiform chromitites. They provided the petrological evidence in clinopyroxene-bearing chromitites for the presence of water, self-alteration features in podiform chromitites, chemistry of clinopyroxene associated with chromite, and water contents and Li isotopic ratios of olivine and clinopyroxene. Additionally, the fate of surface fluids on chromite grains and the importance of water in the formation and evolution of chromite deposits, as inferred by earlier experimental studies, were described.
Thermoelasticity of tremolite amphibole: Geophysical implications
https://doi.org/10.2138/am-2020-7189Peng and Mookherjee evaluate whether anomalously low seismic velocities observed in the mid lithospheric discontinuities (MLD) could be caused by mantle metasomatism i.e., hydrated layer consisting of amphibole and phlogopite. In order to do so, they determine thermoelastic parameters of tremolite using density functional theory calculations. Then they calculate the velocity depth profile for hydrated lithologies with varying amphibole and phlogopite contents. They compare the velocity of hydrated lithology with that of the dry lithology and deduce that while the presence of amphibole and phlogopite in metasomatized mantle might reduce velocity, it may not be the sole mechanism to explain MLD.
Stability of fcc phase FeH to 137 GPa
https://doi.org/10.2138/am-2020-7153Kato et al. propose a revised phase diagram of iron hydride, FeH, which is a candidate component of the Earth’s core. Contrary to previous studies, the experimental results suggest that face-centered cubic (fcc) FeH has a wide stability field at high pressure and temperature instead of a double-hexagonal closed packed (dhcp) FeH. They also examined the compression behavior of fcc FeH and observed a change in compressibility at about 60 GPa, which could be due to a magnetic transition, as suggested by ab initio computation. Fcc FeH could be an endmember in the Fe-FeH system at the Earth’s core conditions.
Partition coefficients of trace elements between carbonates and melt and supra-solidus phase relation of Ca-Mg-Carbonates at 6 GPa
https://doi.org/10.2138/am-2020-7098The presence of Ca-Mg-carbonates affects the melting and phase relations of peridotites and eclogites in the mantle and (partial) melting of carbonates liberates carbon from the mantle to shallower depths. The onset and composition of incipient melting of carbonated peridotites and carbonated eclogites are influenced by the pure CaCO3-MgCO3-system, making understanding of the phase relations of Ca-Mg-carbonates fundamental in assessing carbon fluxes in the mantle. By performing high pressure and temperature experiments, Sieber et al. clarify the supra-solidus phase relations of the nominally anhydrous CaCO3-MgCO3-system at 6 GPa (~200 km deep) showing that Ca-Mg-carbonates will (partially) melt at temperatures above ~1300 °C. A comparison with data from thermodynamic modelling confirms the experimental results. Furthermore, partition coefficients for Li, Na, K, Sr, Ba, Nb, Y and rare earth elements between calcite and dolomitic melt, Ca magnesite and dolomitic melt and magnesite and dolomitic melt are established.
Systematics of H2 and H2O evolved from chlorites during oxidative dehydrogenation
https://doi.org/10.2138/am-2020-7326Lempart et al. performed thermogravimetric analysis combined with quadrupole mass spectrometry to give a complete picture of the thermal decomposition for Fe(II)-containing phyllosilicates. The evolved gas analysis of chamosties, Fe-clinochlores, and biotite revealed that H2 was generated along with H2O under inert atmosphere conditions in the temperature range 500-1000 °C. The higher the Fe(II) content in the chlorites, the more intense the H2 evolution, which results in the increased oxidation of structural Fe(II). H2O and H2 gas are, respectively, indicators of dehydroxylation and oxidative dehydrogenation. These molecules evolve simultaneously, but independently, as they are governed by different mechanisms and kinetics. Despite ongoing dehydrogenation, under oxidizing gas conditions, no H2 was detected, as it immediately combines with an external oxygen to form H2O in the carrier gas. Potential dehydrogenation of Fe(II)-bearing phyllosilicates affects the pathways of fluids released during the deep burial of sedimentary rocks and during metamorphism and melting. This changes the budget of both water and H2 gas in metamorphic and magmatic processes, not only on Earth but also on Mars. The thermal heating experiments were carried out under both an inert and oxidizing atmospheres, which closely reflect anaerobic and aerobic geological environments. The occurrence of oxidative dehydrogenation complicates the interpretations based on the valence of Fe (or Mn), because the Fe(III)/Fetot ratio does not directly reflect oxygen fugacity nor temperature of formation.
Texture and geochemistry of multi-stage hydrothermal scheelite in the Tongshankou porphyry-skarn Cu-Mo (-W) deposit, eastern China: Implications for ore-forming process and fluid metasomatism
https://doi.org/10.2138/am-2020-7194SEM and CL imaging of scheelit can reveal textures that are not seen under transmitted light imaging and shed light on the dissolution-reprecipitation process in scheelites. During this process, the REE patterns and Sr isotopes of the scheelites were modified without significantly modifying their morphology and appearance. Therefore, it is crucial to evaluate such a process according to the textures and geochemical characteristics before using scheelite as an indicator mineral for hydrothermal ore deposits. This study by Han et al. provides a good example of the use of scheelite textures and geochemistry to reveal the nature and source of ore-forming fluids. The scheelite-precipitating fluids were not depleted in Mo based on the coexistence of molybdenite and chalcopyrite and the coexistence of scheelite and chalcopyrite. The Mo contents in the scheelites reflect the fluid oxygen fugacity and, together with the observed Eu anomalies, suggest that the scheelite-fluid partition coefficient of Eu2+ may be greater than that of Eu3+.
EXCALIBR to EXCELIBR and the optical orientation of minerals: Correcting the optical orientation of clinoamphiboles
https://doi.org/10.2138/am-2020-7336CCBYNCNDSteven and Gunter revise the crystallographic setting depicted in crystal form drawings of clinoamphiboles. The crystal form drawings were originally made for a body-centered setting for clinoamphiboles in contrast with the standard C-centered lattice. They also present a methodology for relating the optical geometry of a crystal with its crystallographic setting for orientation-dependent analytical methods, and for future characterization of minerals.
A refined zirconium-in-rutile thermometer
https://doi.org/10.2138/am-2020-7091The zirconium-in-rutile thermometer is widely applied to natural metamorphic rocks, but its accuracy has not been evaluated. Here, Kohn refined the thermometer using a combination of experimental data and data from natural rocks whose pressures and temperatures of formation are well known. The new calibration reduces thermometer uncertainty by a factor of 2-3, and improves retrieval of thermodynamic properties, but also points to as-yet poorly understood causes of compositional variability in natural rocks.
New Mineral Names
https://doi.org/10.2138/am-2020-NMN105612Book Review
https://doi.org/10.2138/am-2020-B105615High Temperature Gas-Solid Reactions in Earth and Planetary Processes
RiMG volume 84, editors: P.L. King, B. Fegley, Jr., and T. Seward
May 2020
Texture constraints on crystal size distribution methodology: An application to the Laki fissure eruption
https://doi.org/10.2138/am-2020-7007Crystal size distributions (CSDs) in igneous rocks are used to tease out evolving environments of crystallization. Kinetic information, such as residence times and average crystal sizes in a sample, is extracted from CSDs. However CSDs are produced from assessments of crystal length measurements, which Cone et al. find vary based on the method and type of image used. They use a combination of techniques ranging from traditional manual approaches and polarized optical microscopy to high-resolution automated mineralogy (automated scanning electron microscopy that relies on elemental information in a mineral) to demonstrate that the resulting CSDs do indeed differ. Even localized fabric can affect the interpretation of crystal lengths. A protocol should be developed that focuses on the textural effects on crystal length determinations, as each image type has the potential to skew measurements.
Hydrogenation reactions of carbon on Earth: Linking methane, and margarine, and life
https://doi.org/10.2138/am-2020-6928CCBYNCNDHydrogenation reactions involve the coupled movement of protons and electrons. These reactions are responsible for numerous molecular transformations deep in the Earth, in biological systems, and also in the atmosphere. Here McGlynn et al., in this open-access paper, survey hydrogenation reactions with a focus on those involving carbon.
Abiotic and biotic processes that drive carboxylation and decarboxylation reactions
https://doi.org/10.2138/am-2020-7166CCBYNCNDSheik et al., in their open-access paper, explain that carboxylation and decarboxylation are an important class of chemical reactions that have helped shape life on the surface of Earth for billions of years. These reactions can be driven through biotic and abiotic processes at different locations in and on Earth’s crust. It is likely that prebiotic synthesis of carboxylic acids was critical for the origin and development of life, which has been using this class of reactions for energy generation and reproduction for billions of years. It is apparent that the location of where these processes are occurring in/on the crust (i.e., subduction zone vs. coal deposit vs. aerobic water column vs. tropical forest) will greatly dictate the rates of reaction and the diversity of carboxylic acids that may be produced. Whether produced biotically or abiotically, these individually mundane reactions are at the heart of complex and even elegant pathways that are extremely important on Earth and potentially on extraterrestrial planets.
In-situ measurements of magmatic volatile elements, F, S, and Cl, by electron microprobe, secondary ion mass spectrometry, and heavy ion elastic recoil detection analysis
https://doi.org/10.2138/am-2020-7221F and Cl measurements by EPMA and SIMS have a generally good agreement on standard glasses available to the scientific community (MPI-DING, Jochum et al. 2006). These are the two most common instruments for in situ halogen measurements and their performance has now been analyzed and compared by Rose-Koga et al. Elastic Recoil Detection Analysis (ERDA) of F and Cl on three standards independently anchors the EPMA-SIMS comparison curves and gives absolute F and Cl concentrations for these standards. The ERDA results also highlight the fact that there is a matrix effect on SIMS measurements of F and Cl in the high-SiO2 standard. Rose-Koga et al. propose a new equation to correct this matrix effect in the SIMS F and Cl data of high-SiO2 samples. We can now use, and rely upon, EPMA or SIMS for F and Cl measurements of glasses on a large SiO2 range covering most of the geological samples.
An evolutionary system of mineralogy. Part I: Stellar mineralogy
https://doi.org/10.2138/am-2020-7173Hazen and Morrison introduce an “evolutionary system of mineralogy” — an approach to classification that links mineral species to their sources and modes of origin. Part I of this system examines stardust, including 41 kinds of minerals that originate in three very different types of stars: classic novae, Type II supernovae, and asymptotic giant branch stars, each of which forms stellar mineral grains under different physical conditions and contributes distinctive isotopic signatures to those grains. They construct a “bipartite network graph” that displays 41 links between these three types of stars (the three larger star-shaped symbols) and 27 different kinds of condensed phases (the smaller diamond-shaped symbols).
A structural study of size-dependent lattice variation: In situ X-ray diffraction of the growth of goethite nanoparticles from 2-line ferrihydrite
https://doi.org/10.2138/am-2020-7217Heaney et al. explore an important but poorly understood nanoparticle behavior — that lattice parameters change systematically for a given metal oxide when crystal diameters fall below 100 nm. This paper documents nanoscale-induced lattice expansion in goethite (FeOOH) through synchrotron X-ray diffraction of ferrihydrite gels that transform to nanogoethite. They demonstrate that the crystallographic changes that accompany expansion due to nanodimensionality in goethite are identical to those operative during heating of goethite.
Cassiterite crystallization experiments in alkali carbonate aqueous solutions using a hydrothermal diamond-anvil cell
https://doi.org/10.2138/am-2020-7118This paper by Liu et al. demonstrates that alkali carbonate can enhance the solubility of SnO2 in aqueous solutions and investigates cassiterite crystallization conditions and the tin-transport species in alkali carbonate aqueous solutions. They find that alkali carbonate-rich aqueous solution or hydrous melt can be a favorable transport medium for tin, and that carbonate or CO2 could be fluxes that promote the formation of rare metal pegmatites.
New insights into the nature of glauconite
https://doi.org/10.2138/am-2020-7341The work by Lopez-Quiros et al. shows that “glauconite” must be assessed as a mica-rich, mica-smectite (R3 ordered), interstratified mineral. Furthermore, they argue that the 100%-mica extreme term (i.e., the glauconitic mica) should not have been described in nature (at least in the usual glauconitic genetic contexts) and that its K+ content would be ~0.8 a.p.f.u. In addition, their new findings give insights into the glauconitization process and, at the same time, investigate the K-deficient character of the dioctahedral mica glauconite.
Kaolinization of 2:1 type clay minerals with different swelling properties
https://doi.org/10.2138/am-2020-7339Li et al. demonstrate that montmorillonite, illite, and rectorite in acidic Al3+-containing solutions can be transformed into kaolinite. Interstratified kaolinite-smectite (K-S), kaolinite-illite (K-I), and kaolinite-rectorite (K-R) form as the intermediate phases during the transformations. The kaolinization of 2:1 type swelling and non-swelling clay minerals is most likely via a local dissolution-crystallization mechanism, mainly from the layer edges rather than from the swelling interlayers. Sizes and stacking order of the newly formed kaolinite strongly depend on those of the precursor minerals. The findings in the present study by Li et al. provide new insights for understanding mineral-water interactions that are central to all geochemical processes.
The quintet completed: The partitioning of sulfur between nominally volatile-free minerals and silicate melts
https://doi.org/10.2138/am-2020-7188Sulfur partitioning between clinopyroxene crystals and melts of compositions from basaltic to dacitic was measured by Callegaro et al. using synchrotron X-ray fluorescence. Crystal chemistry, oxygen fugacity, and meltwater concentration influence the partitioning. These measurements can be applied to natural crystals to determine sulfur concentrations in coexisting melts and estimate sulfur concentrations in magmatic systems in the absence of melt inclusions.
222Rn and 220Rn emanations from powdered samples of samarskite as a function of annealing temperature
https://doi.org/10.2138/am-2020-6988Malczewski and Dziurowicz measured variations in the 222Rn and 220Rn emanation coefficients with a temperature of fully metamict samarskite-(Y) ground to a five-micrometer grain size fraction and annealed from 473 to 1373 K, which cross the structural conversion from the low- to high-temperature samarskite phase. The same mineral species showed noticeably different emanation coefficients of radon and thoron depending on the crystallographic system induced by annealing in an argon atmosphere. The 222Rn emanation coefficients obtained both for 1 h and 24 h annealing were significantly lower than the values reported in the literature for comparable metamict minerals. The results indicate that samarskite-(Y) behaves as a closed system for radon retention across a very broad temperature range, from an untreated sample to a sample annealed at 1373 K, despite high concentrations of uranium and unusual structural complexity.
Polymerization during melting of ortho- and meta-silicates: Effects on Q species stability, heats of fusion, and redox state of MORBs
https://doi.org/10.2138/am-2020-6841The fusion of crystalline orthosilicates and metasilicates produces melts more polymerized than their precursor crystals, as discussed by Nesbitt et al. in this paper. Polymerization during melting can be rationalized from an energetics perspective. Si-NBO-M moieties are more subject to librational, rotational, and vibrational modes than Si-O-Si moieties. Conversion of the former moieties to the latter results in stabilization of melts subject to strong thermal agitation.
Formation of native arsenic in hydrothermal base metal deposits and related supergene U6+-enrichment: The Michael vein near Lahr, SW Germany
https://doi.org/10.2138/am-2020-7062Arsenic is a common constituent of upper crustal fluids, but native arsenic is a rare commodity in natural systems. Understanding the thermodynamically constrained formation conditions of native arsenic for base metal hydrothermal mineralization, as an example, also sheds light on the formation of other As-minerals in other types of hydrothermal deposits. Furthermore, as shown by Scharrer et al., the presence of native arsenic in combination with base metal sulfides produces a unique uranium trap under oxidized weathering conditions.
Lingbaoite, a new silver telluride from the Xiaoqinling gold district, central China
https://doi.org/10.2138/am-2020-7167Lingbaoite (AgTe3) is a new silver telluride discovered in Xiaoqinling gold district of central China. Full details of this new telluride by Jian et al. are in the paper. Lingbaoite probably formed through the cooling of polymetallic melts. Lingbaoite and associated minerals reveal a previously unrecognized magmatic-hydrothermal process, suggesting gold mineralization in the Xiaoqinling gold district involves multiple superimposed processes of gold enrichment.
Oxygen isotope fractionation between gypsum and its formation waters: Implications for past chemistry of the Kawah Ijen volcanic lake, Indonesia
https://doi.org/10.2138/am-2020-7298Utami et al. determined the fractionation of oxygen isotopes between gypsum and its formation water for both water and sulfate, and applied these to gypsum from the Kawah Ijen volcanic lake in Indonesia to reconstruct the oxygen isotopic composition of the lake water during passive degassing and before the last eruption in 1817. Their study shows that gypsum can provide a historical isotopic record of water chemistry for volcanic lakes and other environments where gypsum forms.
2019 MSA Awards
https://doi.org/10.2138/am-2020-AP10552
https://doi.org/10.2138/am-2020-AP10553
https://doi.org/10.2138/am-2020-AP10554
https://doi.org/10.2138/am-2020-AP10555
https://doi.org/10.2138/am-2020-AP10559
https://doi.org/10.2138/am-2020-AP10558
https://doi.org/10.2138/am-2020-AP10556
https://doi.org/10.2138/am-2020-AP10557
https://doi.org/10.2138/am-2020-AP105510
https://doi.org/10.2138/am-2020-AP105511
April 2020
Buoyant rise of anorthosite from a layered basic complex triggered by Rayleigh-Taylor instability: Insights from a numerical modelling study
https://doi.org/10.2138/am-2020-6985Massif type anorthosites constitute a major puzzle of Proterozoic geology. Formation of these rocks from basaltic magma by floatation of plagioclase crystals at the top of a magma chamber is not supported by observations of the major layered basic intrusions of the world. This numerical modelling study by Mukherjee et al. proposes a new genetic process for the origin of massif type anorthosites. It shows that layered basic intrusions may generate Rayleigh-Taylor instability between the anorthosite layers and the layers of denser rocks, when all these rocks behave like Newtonian or non-Newtonian power law fluids. This instability would trigger a vertical movement of the unstable anorthosite as anorthosite diapirs across the lower and middle crust. The result provides for the first time a dynamic basis and a testable hypothesis for the rise of anorthosite from the Earth's mantle into crust.
Chemically oscillating reactions in the formation of botryoidal malachite
https://doi.org/10.2138/am-2020-7029The Belousov-Zhabotinsky reaction displays the same kind of patterns as botryoidal malachite. Arborescent organic structures commonly occur in the geometric center of malachite botryoids. Papineau interprets this in the context of chemical oscillations during the diagenetic decarboxylation of biological organic matter.
Micro- and nano-size hydrogarnet clusters in calcium silicate garnet: Part II. Mineralogical, petrological and geochemical aspects
https://doi.org/10.2138/am-2020-7257Geiger and Rossman investigate mineralogical, petrological, and geochemical characteristics of hydrogarnet clusters in nominally anhydrous garnet. New scientific understanding is obtained because the cluster model not only explains measured infrared spectra, but also permits an atomistic interpretation of many varied experimental results obtained on various calcium silicate garnets over the years.
Petrogenetic insights from chromite in ultramafic cumulates of the Xiarihamu intrusion, northern Tibet Plateau, China
https://doi.org/10.2138/am-2020-7222Song et al. study the geochemistry of chromite, one of the first crystallized phases in mafic melts. Although it can be altered by reaction with hosting mineral and trapped liquid, important clues of parental magma and magma evolution can be preserved. In the Xiarihamu ultramafic intrusion, northern Tibet Plateau, China, the most primitive chromites hosted in olivine with Fo > 87 have similarities with MORB chromite in TiO2 and Al2O3, depleted in Sc and enriched in Ga relative to MORB chromite. This indicates a partial melting of the metasomatized asthenospheric mantle at garnet stable pressures.
Enigmatic diamonds from the Tolbachik volcano, Kamchatka
https://doi.org/10.2138/am-2020-7119Diamonds, found in products of the recent Tolbachik volcano eruption in Kamchatka, Russia, were studied comprehensively by Galimov et al. Their mineralogical and geochemical features demonstrate that the diamonds were formed from volcanic gases under low pressure conditions. This implies that diamond is a polygenetic mineral, which may be formed at both high- and low-pressure conditions.
Volcanic SiO2-Cristobalite: A natural product of chemical vapor deposition
https://doi.org/10.2138/am-2020-7236Schipper et al. study the formation of vapor-phase SiO2-cristobalite in volcanic rocks, which is a natural proxy for Chemical Vapor Deposition. Although metastable, cristobalite may be the most widely occurring SiO2 polymorph in extrusive igneous rocks.
Alteration of magmatic monazite in granitoids from the Ryoke belt (SW Japan): Processes and consequences
https://doi.org/10.2138/am-2020-7025Skrzypek et al. describe two different processes responsible for the alteration of magmatic monazite in granitic rocks from SW Japan: monazite replacement by allanite, apatite, and Th-U phases at low temperature and monazite recrystallization with the formation of U-Y-enriched domains at high temperature. The versatility of the accessory mineral monazite gives a unique chance to constrain the nature and timing of metasomatic events. The different alteration processes can be ascribed to hydrous fluid or granitic melt infiltration. In situ age dating of monazite alteration textures gives information on: the age of magmatic crystallization (primary monazite domains), the age of high-temperature recrystallization (secondary monazite domains), and the timing of low-temperature replacement (Th-U-rich phases).
Smamite, a new mineral and a possible sink for Sb during weathering of fahlore
https://doi.org/10.2138/am-2020-7133Plášil et al. discovered smamite, a new supergene arsenate-hydrate phase that contains both calcium and antimony. Since this new mineral is rather inconspicuous, it is highly possible that it has remained overlooked by mineralogists for decades. As one of the few Sb(V) supergene minerals, it can serve as a sink for Sb during supergene weathering.
The new K, Pb-bearing uranyl-oxide mineral kroupaite: Crystal-chemical implications for the structures of uranyl-oxide hydroxy-hydrates
https://doi.org/10.2138/am-2020-7311Plášil et al. report on the new mineral, kroupaite, which is an additional member of the schoepite-family of minerals. This family is important due to environmental issues associated with the storage of irradiated or spent nuclear fuel in geological repositories.
Mg diffusion in forsterite from 1250-1600 °C
https://doi.org/10.2138/am-2020-7286New Mg isotopic diffusion experiments in forsterite show a clear dependence of diffusivity on silica activity, with higher Mg diffusivities at protoenstatite-buffered conditions than in experiments buffered by periclase. The new data by Jollands et al. reconciles some previous discrepant datasets.
Changes of antigorite cell parameters close to the antigorite dehydration reaction at subduction zone conditions
https://doi.org/10.2138/am-2020-7159Shen et al. report TEM studies of natural antigorite after being equilibrated in a piston cylinder in the P-T range of 600-650 °C and 25-45 kbar. The length of the a-axis of antigorite samples (can be expressed as m value) was determined and a P-T-m diagram has been constructed based on the new experimental and previous data. The m-value of antigorite changes systematically with pressure and temperature (due to loss of water), which could be used to better constrain the formation conditions of subducted serpentinites.
The issue ends with a memorial of Edward J. Olsen (1927-2020) by Ian Steele and Nancy Hutcheon
https://doi.org/10.2138/am-2020-m697March 2020
Heavy halogen geochemistry of martian shergottite meteorites and implications for the halogen composition of the depleted shergottite mantle source
https://doi.org/10.2138/am-2020-7237Volatile elements (e.g., H, C, N) influence the physical and chemical properties of planets. The halogen group elements, Cl, Br, and I can provide insight into volatile distribution and transport processes due to their ability to track with water. By measuring halogens in shergottites, martian meteorites that are derived from volcanic processes, Clay et al. show that the halogen geochemistry of different shergottites indicates distinct mantle sources. They calculate that the halogen composition of bulk silicate Mars is very similar to that of bulk silicate Earth, suggesting common volatile source(s) or delivery mechanism(s) to the terrestrial planets.
The distribution and abundance of halogens in eclogites: An in situ SIMS perspective of the Raspas Complex (Ecuador)
https://doi.org/10.2138/am-2020-6994Subduction of oceanic crust cycles F and Cl from surface reservoirs to the mantle, however the minerals responsible for hosting these halogens, and the bulk halogen content of deeply subducted oceanic crust, remains poorly understood. Urann et al. analyze halogen abundances of individual minerals and bulk rocks to better quantify the halogen content of high pressure metamorphic altered oceanic crust during subduction.
Pressure dependence of Si diffusion
https://doi.org/10.2138/am-2020-7197Tsujino et al. investigate the pressure dependence of Si diffusion in gamma-Fe at pressures of 5-15 GPa and temperatures of 1473-1673 K using a Kawai-type multi-anvil apparatus. Even after 1 billion years, maximum of Si diffusion length at planetary and satellites' core conditions is less than ~1.2 km. On the other hand, the estimated strain of plastic deformation by Harper-Dorn creep based on Si diffusivity in gamma-Fe reaches more than 103 at a stress level of 103-104 Pa.
Seismic detectability of carbonates in the deep Earth: A nuclear inelastic scattering study
https://doi.org/10.2138/am-2020-6901Elastic property measurements of two synthetic iron-containing carbonates reveal the extent to which carbonates are seismically detectable using geophysical methods. Chariton et al. confirm that nuclear inelastic scattering is a reliable method for the in situ study of the elastic properties of Fe-bearing systems at high pressures and temperatures, and reveals that seismic detectability of carbonates requires a high concentration of carbon in subducted slabs. Such contents may be reached in several present-day subduction trenches, which are potential targets for geophysical studies investigating carbonates in the deep Earth.
Equations of state, phase relations, and oxygen fugacity of the Ru-RuO2 buffer at high pressures and temperatures
https://doi.org/10.2138/am-2020-7060Oxygen fugacity (fO2) is a critical variable in magmatic systems, affecting phase relations and element partitioning. At present, large uncertainties in fO2 exist in high-pressure experimental work. Armstrong et al. report in situ X-ray diffraction measurements of Ru and RuO2, from which they have calculate the absolute oxygen fugacity of the reaction at P/T conditions up to the top of the lower mantle. This allows utilization of this buffer to impose a known fO2 on experiments, reducing uncertainty in the exploration of mantle processes.
Experimental study of the Pt and Pd antimonides and bismuthinides in Fe-Ni-Cu sulfide systems between 1100 and 700 degrees C and applications to nature
https://doi.org/10.2138/am-2020-7154Helmy and Botcharnikov present phase relations in the Pt-Sb and Pt-Bi sulfide systems in the temperature range 1100 to 750 °C and in the Pd-Sb and Pd-Bi sulfide systems in the temperature range 1100 to 700 °C. Partitioning behavior of Pt, Pd, Ni, and Cu between monosulfide solid solution and Sb-bearing and Bi-bearing sulfide melts is discussed, as well as Sb and Bi behavior in Pt and Pd-bearing sulfide systems. These results are used to investigate the behavior of Sb and Bi during mantle melting processes.
Layers stacking disorder in Mg-Fe chlorites based on powder X-ray diffraction data
https://doi.org/10.2138/am-2020-6982Luberda-Durnas et al. investigate the effects of semi-random stacking in chlorites on powder X-ray diffraction patterns of bulk powder samples. A complete set of information about the stacking sequences in chlorite structures was determined based on XRD pattern simulation and study of a suite of natural samples. A detailed study of semi-random stacking sequences shows that simple consideration of the proportion of IIb-2 and IIb-4/6 polytypes, assuming equal content of IIb-4 and Iib-6, is not sufficient to fully model the stacking structure in chlorites. Several, more general, possible models were therefore considered. In the first approach, a parameter describing a shift into one of the ±1/3b directions (thus, the proportion of IIb-4 and IIb-6 polytypes) was refined. In the second approach, for samples with slightly distinguishable hkl reflections with k≠3n, some kind of segregation of individual polytypes (IIb-2/4/6) was considered. In the third approach, a model with rotations of 2:1 layers about 0°, 120°, 240° were shown to have the lowest number of parameters to be optimized and therefore, giving the most reliable fits.
Elasticity of single-crystal Fe-enriched diopside at high-pressure conditions: Implications for the cause of upper mantle low-velocity zones
https://doi.org/10.2138/am-2020-7075Fan et al. report the acoustic wave velocities and density of a single-crystal Fe-enriched diopside by Brillouin light scattering combined with in situ synchrotron X-ray diffraction in a diamond anvil cell up to ~18.5 GPa at room temperature. Based on a comparison of the obtained elastic moduli of Fe-enriched diopside with those of Fe-free diopside (CaAlSi2O6) and hedenbergite (CaFeSi2O6) reported in the literature, the authors evaluate the Fe composition effect on the elasticity and seismic parameters of diopside at the upper mantle P-T conditions. The comparison shows systematic linear correlations between the Fe composition and single-crystal elastic moduli of diopside, while the modeling indicates that substitution of 20 mol% Fe in diopside can reduce VP and VS by ~1.8% and ~3.5%, respectively, along both the expected normal mantle geotherm and a representative cold subducted slab geotherm. Furthermore, modeling also shows that the VP and VS profiles of Fe-enriched pyroxenite along the cold subducted slab geotherm are ~3.2% and ~2.5% lower than AK135 model at 400 km depth, respectively. These results lead to the conclusion that the presence of Fe-enriched pyroxenite (including Fe-enriched clinopyroxene, Fe-enriched orthopyroxene, and Fe-enriched olivine) can be an effective mechanism for causing low-velocity anomalies in the upper mantle regions atop the 410 km discontinuity at cold subducted slab conditions.
XANES spectroscopy of sulfides stable under reducing conditions
https://doi.org/10.2138/am-2020-7146Anzures et al. detail new S K-edge XANES spectra for sulfides stable under reducing conditions for comparison to potential extraterrestrial analogs. This information can be used to determine the coordination chemistry and oxidation state of S species in geologic materials. These new sulfide standards should improve studies of sulfur speciation in reduced silicate glasses and minerals with applications for the early Earth, Moon, Mercury, and enstatite chondrites.
Zircon and Apatite geochemical constraints on the formation of the Huojihe Porphyry Mo Deposit in the Lesser Xing'an Range, NE China
https://doi.org/10.2138/am-2020-7226Northeastern China is an important Mo resource region, with more than 80 Mo deposits and occurrences. However, the major factors controlling large-scale porphyry Mo mineralization in this region are still unclear, and whether there is any inherent Mo enrichment of the source region and/or any pre-degassing magmatic processes leading to high-Mo melts remains enigmatic. Xing et al. use a combination of geochemistry and isotopic analyses of zircon and apatite from the Huojihe porphyry Mo deposit in NE China to provide insights into the characteristics of the parental magma and ore-forming mechanisms responsible for the porphyry Mo mineralization. The results suggest that pre-degassing enrichment of Mo and S in the original magma is not necessarily important in the formation of the Huojihe Mo deposit and that factors other than melt composition, including (but not limited to) a large magma chamber, may be more critical in forming a porphyry Mo deposit. The conclusions from this study might also apply to other porphyry Mo deposits worldwide.
Textural and compositional evolution of iron oxides at Mina Justa (Peru): implications for mushketovite and formation of IOCG deposits
https://doi.org/10.2138/am-2020-7024Hu et al. provide mineralogical evidence to support that the platy magnetite in IOCG systems is mushketovite. The textural and compositional data on magnetite from the Mina Justa deposit provide new insights into evolutionary mechanisms of iron oxides in IOCG deposits, which are dominated by oxygen fugacity and temperature changes and lead to the formation of multiple generations of magnetite at Mina Justa. The primary hematite transformed into TM1-1 magnetite due to a decline in fO2 and then was replaced by TM1-2 magnetite with increased temperature. Meanwhile, granular TM2-1 magnetite directly precipitated from hydrothermal fluids. With the decrease of temperature, TM1-2 and TM2-1 magnetite were replaced by TM1-3 and TM2-2 magnetite, respectively.
Siwaqaite, a new mineral of the ettringite group from the pyrometamorphic Daba-Siwaqa complex, Jordan
https://doi.org/10.2138/am-2020-7208Juroszek et al. focus on a new mineral, siwaqaite, from the pyrometamorphic Daba-Siwaqa complex in Jordan, which is a Cr6+-analog of ettringite. They provide a detailed description of physical properties, structural parameters and spectroscopic results for siwaqaite, as well as comparisons with other members of the ettringite group. Siwaqaite is an interesting phase because it contains Cr, a redox-sensitive transition element. Wide usage of Cr in the industry, such as the production of stainless steel, metal finishing, wood preservatives, and refractory products, causes release of high concentrations of chromates in surface water. Due to the high solubility and oxidizing potential, hexavalent Cr is a toxic, teratogenic, carcinogenic, and mutagenic element in biological systems, which can damage kidney and other tissue structures. Juroszek et al. suggest that siwaqaite, in analogy to ettringite, can be used for immobilization of toxic cations and oxyanions because the structure of ettringite has a wide ability to ions exchange. Moreover, slight changes of the chemical composition do not modify the structure. Therefore, minerals form ettringite group could be used for selected contaminant removal by incorporating toxic ions into their structures.
Negevite, the pyrite-type NiP2, a new terrestrial phosphide
https://doi.org/10.2138/am-2020-7192Britvin et al. report the discovery of negevite, natural pyrite-type NiP2, that might open new insights into the role of phosphides as a source of phosphorus in prebiotic phosphorylation processes. They discuss the aquatic oxidation of meteoritic phosphides leading to formation of diverse phosphorus compounds, which could serve as prebiotic phosphorylation agents at the early stages of Earth's evolution. In that respect, the unique catalytic and electrochemical properties of NiP2 could provide new routes for further exploration of phosphorylation pathways.
Transjordanite, a new terrestrial and meteoritic phosphide, and natural solid solutions barringerite-transjordanite
https://doi.org/10.2138/am-2020-7275Britvin et al. report the presence of natural hexagonal solid solutions along the join Fe2P-Ni2P. These Fe2P-Ni2P solid solutions are related to the Fe-Ni-P system that, along with the Fe-Ni-S and Fe-Ni-C ternaries, are the most significant reduced cosmochemical systems. They discuss how knowledge of phase relationships and compositional variations of Fe-Ni phosphide minerals has a broad range of implications, including the origin of the solar system, composition of deep planetary interiors, meteoritics, terrestrial processes in super-reduced environments, and processes of prebiotic phosphorylation on early Earth.
February 2020
A new emerald occurrence from Kruta Balka, Western Peri-Azovian region, Ukraine
https://doi.org/10.2138/am-2020-7010Franz et al. investigated emerald, the bright-green gem variety of beryl, from a new locality at Kruta Balka, Ukraine, and compare its chemical characteristics with those of emeralds from selected occurrences worldwide to clarify the types and amounts of substitutions as well as the factors controlling such substitutions. They applied secondary ion mass spectrometry and showed that—although beryl in general has quite variable Be-contents—emeralds are surprisingly simple. However, Li-contents, even small, are important; the new occurrence is unusual, because Li (together with Na) is substituting for Al, and not, as at most other emerald localities, for Be, and small amounts of Li can sit in a 'hollow' channel position. The green color of emerald is produced by the incorporation of small amounts of Cr and V.
Dissolution of poorly soluble uranyl phosphate Phases in the Metaautunite Supergroup
https://doi.org/10.2138/am-2020-7106Uranyl phosphate minerals of the meta-autunite group are sparingly soluble in water under most conditions and have been targeted in remediation strategies for contaminated sites. Here Lobeck et al. demonstrated that under some aqueous peroxide-bearing conditions these minerals dissolve readily to form complex cage clusters built of uranyl hexagonal bipyramids that persist in solution as complex macro-anions.
Quartz crystals in Toba rhyolites show textures symptomatic of rapid crystallization
https://doi.org/10.2138/am-2020-6947Barbee et al. studied quartz crystals in rhyolites erupted from Toba caldera (Indonesia) that show abundant textural signs of rapid crystallization. This study is the first to document diverse quartz textures as consequences of rapid, disequilibrium crystal growth in a caldera-forming rhyolite. Studies that seek to extract time information from the crystal record have found that trace element diffusion in quartz indicates crystallization, or residence, timescales that are much shorter than previously thought for large silicic magma systems. This work provides at least some physical, textural evidence that dynamic processes must operate on short geologic timescales in these systems. The documentation of skeletal to dendritic quartz textures is expected to provide vital context to interpreting the chemical stratigraphy of quartz and may impact the application of quartz as a petrologic tool.
Constraints on non-isothermal diffusion modeling
https://doi.org/10.2138/am-2020-7193This study by Rout et al., through a series of diffusion experiments, evaluates the non-isothermal diffusion model, which gives more realistic estimates of diffusion time in natural samples, assesses the errors, and provides a simpler and more accurate approach to better implement the model.
Machiite, a new oxide mineral from the Murchison carbonaceous chondrite: A new ultrarefractory phase from the solar nebula
https://doi.org/10.2138/am-2020-7185Krot et al. report on a discovery of a new ultra-refractory mineral, machiite, found in the CM carbonaceous chondrite Murchison. Machiite is intergrown with corundum and most likely formed by gas-solid condensation from a gas of solar composition at the very beginning of the protoplanetary disk evolution. Machiite is in O-isotope disequilibrium with corundum. They suggest that both minerals condensed from an 16O-rich solar nebula gas. Subsequently, machiite experienced O-isotope exchange with an aqueous fluid on the CM chondrite parent body.
Partitioning of V and 19 other trace elements between rutile and silicate melt as a function of oxygen fugacity and melt composition: Implications for subduction zones
https://doi.org/10.2138/am-2020-7013Holycross and Cottrell detail the results of 16 experiments to measure the partitioning of 20 trace elements between the mineral rutile (TiO2) and silicate melt as a function of redox state and melt composition. Special attention is given to the behavior of the multivalent trace element vanadium (V), which may speciate as V2+, V3+, V4+, or V5+ in the silicate Earth. The distribution of V between rutile and melt is a direct function of both oxidation state and melt composition. Vanadium is strongly compatible in rutile when V4+ is the dominant species, and when melt compositions are highly polymerized and contain low concentrations of TiO2. Their new data indicate that rutile may be a significant sink for V in the solid Earth. The flux of V will be coupled to Ti during mass transfer through the subduction system when rutile is present and will change as a function of the oxidation state of the subducting slab.
Cl-bearing Fluorcalciobritholite in high-Ti basalts from Apollo 11 and 17: Implications for volatile histories of late-stage lunar magmas
https://doi.org/10.2138/am-2020-7180Greenwood et al. report the second fluorine-bearing mineral found from the Moon, a Cl-bearing fluorcalciobritholite. This mineral has only been found in mesostasis of high-Ti magmas with fluorapatite. This F- and Cl-bearing mineral crystallizes after fluorapatite, implying that high-Ti lunar magmas were not volatile-poor.
Amphibole-rich cumulate xenoliths in the Zhazhalong intrusive suite, Gangdese arc: Implications for the role of amphibole fractionation during magma evolution
https://doi.org/10.2138/am-2020-7199Although the importance of precipitating amphibole has been inferred through distinctive geochemical fingerprints of derivative products, this study by Zhou et al. provides a direct snapshot of this differentiation process. With progressive accumulation, the amphibole-rich sponge will form in the arc crust, which is fundamental to interpreting many issues in petrology. Particularly, the amphibole-rich xenoliths have petrological similarities to appinites, and their results suggest that some appinites likely represent amphibole-rich cumulates.
Extraterrestrial, shock-formed, cage-like nanostructured carbonaceous materials
https://doi.org/10.2138/am-2020-7305Németh and Garvie report the first natural carbon nano-onions resembling large multilayered fullerenes and bucky-diamonds from the impact-shocked Gujba (CBa) meteorite. They propose that the carbon nano-onions formed from the primitive carbonaceous materials, whereas bucky-diamonds could have formed through the high-pressure transformation of nano-onions. Both nano-onions and bucky-diamonds are fullerene-type structures, and as such could be a component of the astronomical 217.5 nm absorption feature.
Minerals Matter: Pyrite: fool's gold records starvation of bacteria
https://doi.org/10.2138/am-2020-7234This paper by Gregory and Kohn gives an introduction to a high-school level audience on pyrite, and the sulfur isotopes contained within it, and how that can be used to understand past ocean conditions.
The issue ends with a list of 2019 reviewers for American Mineralogist (https://doi.org/10.2138/am-2020-696) and a book review by Tony Barber on Geological Belts, Plate Boundaries, and Mineral Deposits in Myanmar (2017) by Andrew Mitchell, Elsevier, eBook ISBN: 9780128033838; Paperback ISBN: 9780128033821, 524 p. (https://doi.org/10.2138/am-2020-B10521)
January 2020
Highlights and Breakthroughs
https://doi.org/10.2138/am-2020-7312Elliott’s Highlights and Breakthroughs paper details how Li and Zhou describe the role of halloysite in sorbing rare-earth elements (REE) in a regolith setting in SE China. This mineral-based model explains the occurrences of the ion-sorbed REE in this regolith setting, which is a leading source of the heavy, and technologically important, rare-earth elements (Gd-Lu, Y).
Review Paper
https://doi.org/10.2138/am-2020-7205The founding of the MSA in 1919 followed the discovery of X-ray diffraction so closely that one might hypothesize a causal link between the events. This review by Peter Heaney explores the efforts of the MSA founders to communicate the importance of the crystallography revolution to their contemporaries, and it argues that the then-new conceptions of atomicity differentiated mineralogy from other styles of geological investigation, thereby warranting the establishment of a specialized professional society.
Deep Earth Carbon Reactions through Time and Space
https://doi.org/10.2138/am-2020-6888CCBYCarbon reactions take place in the deep Earth and have played a critical role in our planet's development. McCammon et al. describe these reactions, including carbon capture during Earth's formation, freezing of liquid iron-carbon to form Earth's inner core, and oxidation-reduction reactions in the modern-day mantle. Although these reactions are "invisible" because they are not observed on Earth's surface, our planet would be vastly different if none of these reactions had taken place during its history.
Magmatic Carbon Outgassing and Uptake of CO2 by Alkaline Waters
https://doi.org/10.2138/am-2020-6986CCBYThe Earth in Five Reactions project was part of the Deep Carbon Observatory program, a decade-long effort to understand the quantities, movements, forms, and origin of carbon in Earth. In this paper, Edmonds et al. review the reactions involved when carbon exsolves from silicate melts in magmatic intrusions and volcanic eruptions and the reactions that occur when carbon is dissolved into aqueous solutions, with a particular reference to alkaline lakes. They discuss the implications of our understanding of these natural reactions for forecasting the longevity and effects of anthropogenic carbon release.
New Insights into the Evolution of MVT Hydrothermal System
https://doi.org/10.2138/am-2020-7021The Upper Yangtze Pb-Zn metallogenic province (South China) covers an area of 170,000 km2 and contains approximately 400 carbonate-hosted epigenetic Pb-Zn deposits totaling ~26 Mt of metal reserves. Despite the Wusihe deposit resembling Mississippi Valley Type, MVT, deposits in North America, it shows many distinctive geological and geochemical features warranting further investigation. Luo et al. take the Wusihe Pb-Zn deposit as a case study, employing LA-ICPMS in situ quartz trace element analysis, bulk and NanoSIMS in situ S isotopes, and femtosecond LA-MC-ICPMS in situ Pb isotopes to reveal the evolution of this MVT hydrothermal system. The trend of trace element compositions in quartz indicates the role of an acid-producing process resulting from sulfide precipitation and acid consumption by carbonate buffering. New bulk δ34S values of sulfides imply that in addition to thermochemical sulfate reduction (TSR), bacterial sulfate reduction (BSR) may also play an important role in the formation of S2-. In situ δ34S values suggests a mixing of two isotopically distinct sources of S2- produced by TSR and BSR. New galena Pb isotopic ratios suggest that the metal sources in the Wusihe deposit are mainly Proterozoic basement rocks. Hence, a multi-process model (i.e., basin-mountain coupling, fluid mixing, local sulfate reduction, in situ acid-production, and involvement of black shales and carbonate sequences) was responsible for the formation of the Wusihe deposit, whilst S2- was produced by both TSR and BSR, providing new insights into the evolution of MVT hydrothermal systems.
Celestine Discovered in Hawaiian basalts
https://doi.org/10.2138/am-2020-6995Celestine was discovered in basalt from a Hawaii. This represents the first report of celestine in an oceanic basalt. Garcia and Hellebrand document the mode of occurrence of celestine in Ka'ula Island basalts using backscattered electron imagery, present high precision electron microprobe analyses of celestine, examine the effects of alteration on the geochemistry of Ka'ula basalts, and discuss possible origins for the formation of celestine in these rocks. Future studies of hydrothermally altered basalts from active volcanoes on oceanic islands, especially for basalts with elevated Sr contents (e.g., 1000 ppm), should be aware of the possible presence of celestine in moderately altered lavas.
Microstructural Controls on the Chemical Heterogeneity of Cassiterite Revealed by Cathodoluminescence and Elemental X-ray Mapping
https://doi.org/10.2138/am-2020-6964Cassiterite (SnO2) has gained attention recently as a geochronometer for Sn-bearing mineralized systems, but there is no clear framework for the interpretation of cathodoluminescence imaging frequently employed prior to analysis. In this paper, Bennett et al. use a combination of hyperspectral cathodoluminescence and quantitative X-ray element mapping to constrain the growth history of cassiterite crystals.
Hornblende as a Tool for Assessing Mineral-Melt Equilibrium and Recognition of Crystal Accumulation
https://doi.org/10.2138/am-2020-6972Recent studies have proposed that hornblende major element compositions can be used to calculate equilibrium anhydrous melt compositions. In this study, Fe/Mg partitioning relationships and hornblende chemometry are used to evaluate the extent to which hornblende from several plutonic and volcanic rocks are in equilibrium with their associated bulk-rock and/or glass compositions. Werts et al. found that hornblendes from many plutonic and volcanic rocks are not in equilibrium with their bulk-rock compositions; instead they are in equilibrium with melts that are more silicic than the bulk-rock sample, indicating that some degree of crystal accumulation and/or melt loss has occurred and requiring removal and/or redistribution of silicic melts. The application of hornblende partitioning relationships and chemometry to bulk-rock and glass samples has wide-ranging implications on discerning volcano-plutonic connections, the ways in which magmatic processes are determined and quantified, and on the distribution of melt compositions in the crust.
The Role of Clay Minerals in Forming the Regolith-hosted Heavy Rare Earth Element Deposits
https://doi.org/10.2138/am-2020-7061Heavy rare earth elements (HREEs) have become significantly important in our modern society. The dominant source of HREE is from regolith-hosted deposits, presumably occurring as adsorbed on the clay minerals in weathering crusts. However, the actual relationship between the HREEs and clay minerals has not been comprehensively examined before. In this study, for the first time, Li and Zhou link the properties of clay minerals to HREE accumulation through comprehensive mineralogical and geochemical studies exemplifying the world's largest regolith-hosted HREE deposit, Zudong, South China. Abundant poorly crystallized halloysite and kaolinite of high specific surface area, pore volumes, and low crystallinity in the lower part of soil profiles transform to well-crystallized vermicular kaolinite of low specific surface area, pore volumes, and high crystallinity in the upper part of the soil profile during progressive weathering, which is correlated with a significant decrease in the clay-adsorbed REE concentrations. Variation in physicochemical properties of the clay minerals during progressive weathering is one of the key factors for REE accumulation and enrichment in weathering crusts to form the regolith-hosted deposits.
The Tetrahedrite Group: Nomenclature and classification
https://doi.org/10.2138/am-2020-7128Biagioni et al. show how the new IMA-CNMNC approved classification of the tetrahedrite group allows the full description of the chemical variability of these widespread sulfosalts and is able to convey important information not only to mineralogists but also to ore geologists and industry professionals.
Caseyite, a New Mineral Containing a Variant of the flat-Al13 Polyoxometalate Cation
https://doi.org/10.2138/am-2020-7181The new mineral caseyite records the presence of technologically important polyoxometalate ions in geochemical fluids, both as cations and as anions. This report by Kampf et al. is the first of a naturally occurring nanocluster resembling the “flat-Al13” polyoxocation that is used to make thin films for electronics, optics, and optoelectronics applications, and it lends greater credence to research suggesting that the flat-Al13 cluster is an important aqueous ionic species in natural systems. The discovery of a new flat-Al13 variant containing a V5+ substituent suggests new avenues for developing technologically useful modifications of the flat-Al13 structure.
Incorporation of Mg in Phase Egg
https://doi.org/10.2138/am-2020-7204Bindi et al. demonstrate that Phase Egg, considered one of the main players in the water cycle of the mantle, can incorporate large amounts of Mg in its structure and that there exists a solid solution with a new hypothetical MgSiH2O4 end-member. The new hypothetical MgSiH2O4 end-member would be a polymorph of Phase H, a leading candidate for delivering significant water to the deepest part of the lower mantle.
Imaging Trace Element Zoning in Pyroxenes Using Synchrotron XRF Mapping with the Maia Detector Array: Benefit of Low Incident Energy
https://doi.org/10.2138/am-2020-7228Microbeam XRF mapping with synchrotron radiation lets Barnes et al. reveal subtle trace element zoning in igneous phases. Scanning with incident energy below the Fe K edge improves sensitivity and precision. An entire thin section can be imaged in a few hours. This is a powerful technique for imaging and understanding crystal cargoes in magmatic systems.
Memorial of Enver Murad 1941-2019
https://doi.org/10.2138/am-2020-m695American Mineralogist Volume 104
December 2019
This issue of American Mineralogist starts with a Centennial Review on high-pressure minerals by Tschauner (page 1701). The review is dedicated to the occurrence, relevance, and structure of minerals whose formation involves high pressure. Over the past ten years more high-pressure minerals have been discovered than during the previous fifty years. The previously unexpected richness in distinct high-pressure mineral species allows for assessing differentiation processes in the deep Earth.
Talla and Wildner (page 1732) performed detailed crystal-chemical and spectroscopic studies on a continuous solid solution series between kieserite and szomolnokite, (Mg,Fe)SO4·H2O, under ambient and Martian temperature conditions. Their verification of the existence of this sold solution series suggests that respective intermediate compositions are expected to occur in Fe-rich environments, such as the surface of Mars. The presented data can generally assist in the characterization of kieserite, szomolnokite and their solid solutions based on FTIR-, Raman-spectroscopic and structural data.
Brenna et al. (page 1750) investigate the role of crystal mushes on intra-eruptive trachyte-phonolite transition from natural evidences and experimental constraints. The transition of magma composition from trachyte to phonolite within a single eruption is an uncommon occurrence and cannot be derived from closed-system fractionation processes. The authors performed experiments to test the conditions that generated such a transition. Both experiments and natural samples suggest the formation of phonolite from trachyte as a result of open-system interaction between trachyte magma and extensively crystallized framework of feldspar with interstitial phonolitic glass.
Yakymchuk and Acosta-Vigi (page 1765) compare the measured concentrations of P2O5 in nanogranites and melt inclusions with existing solubility expressions for apatite in anatectic systems. They first identify discrepancies between the measured values and the predicted values and then use this information to discuss the implications for the behavior for apatite in anatectic systems. They provide a detailed analysis of the factors that contribute to apatite behavior in high-temperature metamorphic rocks and finally highlight some broader implications for the Nd isotope values of granites and the mechanisms of anatexis in migmatites and granulites.
Breton et al. (page 1781) conducted static compression experiments of γ’-Fe4N up to 77 GPa to examine its stability and elastic properties. From the obtained data together with existing data, the authors established a pressure-temperature phase diagram for the system Fe-N. The diagram shows the high-pressure stability of γ’-Fe7N3 to 56 GPa at 300 K beyond which β-Fe7N3 phase is stable. If Earth's core contains nitrogen, β-Fe7N3 is the most likely stable iron nitride phase in the solid inner core.
Almeida and Jenkins (page 1788) investigate the stability field (P-T-X) of an intermediate composition scapolite and compare the stability field of this Cl-rich scapolite with that of the end-member marialite. The results demonstrate that a small change in the scapolite composition from marialite to intermediate scapolite (Ma85Me15) causes a shift in its stability, relative to the plagioclase plus salt assemblage, by 260 °C from 990 to 730 °C at 2.0 GPa. The combined effects of a broader thermal stability field and tolerance to lower salinities mean that intermediate scapolites will be more commonly encountered in shallow- to mid-crustal levels at temperatures of ~500 °C or higher.
Amulele et al. (page 1800) conducted high-pressure electrical conductivity measurements, via impedance spectroscopy, on the silica polymorphs quartz and coesite. The authors compare their data to previously published values for quartz and stishovite and apply these data to a magnetotelluric profile to provide geophysical context. This study holds significance as quartz and the associated polymorphs of coesite and stishovite are prevalent in the Earth's crust and upper mantle. The authors propose that these data and the interpretations therein will serve as a useful tool to the geophysical and mineral physics communities.
Owen et al. (page 1806) investigated REE-, Sr-Ca-aluminum-phosphate-sulfate (APS) minerals of the alunite supergroup and their role as hosts for radionuclides. Woodhouseite-svanbergite-dominant APS assemblages are interpreted as paragenetically late with respect to the Cu-(Fe)-sulfides and associated gangue which they consistently replace (they also replace earlier REE-bearing phosphates). This interpretation adds to the growing body of evidence for a low-temperature hydrothermal overprint at Olympic Dam and represents a valuable contribution to a holistic mineralogical-geochemical-geometallurgical model of REE behavior in the Olympic Dam IOCG-U-Ag deposit. The observed sorption properties of the Ca-Sr-dominant APS minerals and their ability to scavenge radionuclides from the 238U decay chain from 226Ra onwards highlight both the need to suppress natural APS phases in copper concentrates and prevent crystallization of new, anthropogenic APS phases during leaching or elsewhere during the processing-refining cycle. Alternatively, the capacity of APS minerals to scavenge radionuclides could be used in the removal of the products of U decay from processing streams, provided that these phases can be re-dissolved or otherwise removed from the final product.
Vigliaturo et al. (page 1820) determined the Fe-valence state on amosite-asbestos boundaries using acSTEM dual-electron energy-loss spectroscopy (EELS). The authors determined the Fe-valence state from the core-loss region of the electron energy-loss spectra and compared the results with those obtained independently from X-ray photoelectron spectroscopy and a simple geometric model of the fiber boundaries. Dual-EELS allows collection of simultaneously low-loss and high-loss spectra, making it possible in the near future to apply this approach to the "soft" organic materials, which can be intimately associated with asbestos fiber surfaces after the interaction with human cells. This study will help in promoting the use of acSTEM dual-EELS in both medical mineralogy and general mineralogy.
Oberti et al. (page 1829) investigated the thermoelastic behavior, cation exchange and deprotonation processes of a Fe-rich holmquistite using high-temperature single-crystal X-ray diffraction up to 1023 K. The results contribute to paving the way towards a general crystal-chemical model that can explain how orthorhombic amphiboles responds to changes in physiochemical conditions, especially high temperature, and allows the release of water in diverse geological settings.
Lorenz et al. (page 1840) conducted a detailed textural and geochemical study on a relatively unknown but important rare-earth element bearing mineral group called britholite. The studied britholite occurrence at the Rodeo de los Molles deposit, central Argentina, is exceptional in its high concentration of this unusual mineral within vein mineralizations. The authors discovered intergrowth textures of apatite and britholite, which makes it very likely that a miscibility gap between these two phases exists. The paper also discusses the strong alteration of the mineral, which could be of particular interest for the field of nuclear waste containment, as phases with the britholite structure have been used to store liquid nuclear waste.
Kampf et al. (page 1851) describe a new mineral, bicapite, KNa2Mg2(H2PV5+14O42)·25H2O, from the Pickett Corral mine, Montrose County, Colorado. The structure of this mineral contains the [H2PV5+12O40(V5+O)2]7- protonated bicapped α-isomer of the Keggin heteropolyanion. The mines of the Colorado Plateau are a rich source of minerals with complex heteropolyions, which have formed in low-temperature, post-mining mineral assemblages. The discovery of these minerals advances our knowledge and understanding of mineral complexity on Earth, provides new insights into complex ions that can exist in near-surface environments, and informs researchers in other fields of new avenues to explore in developing phases with potential technological uses.
Sokol et al. (page 1857) investigated carbon solubility in a Fe melt and the iron-loving behavior of C and N in a carbon-saturated Fe-C-N-S system at 5.5 and 7.8 GPa and 1450-1800 °C. Carbon becomes twice less soluble as nitrogen increases in a graphite-saturated melt but the solubilities of C and N are commensurate in the presence of 1-1.7 wt.% S. Nitrogen partitioning between diamond and an N- and S-bearing Fe melt, DNDm/Met, is in the range 0.012-0.025. This study is another step forward to the understanding of nitrogen "deficit" and carbon "excess" in the mantle.
Belakovskiy et al. (page 1866) introduce 11 new minerals, including cesiodymite, cryptochalcite, feodosiyite, fluoro-tremolite, itelmenite, ozerovaite, ramazzoite, redcanyonite, selivanovite, vanderheydenite, and wrightite.
This issue ends with an Editors’ Note (page 1872) on mineral localities in Kaminsky et al. (2019, American Mineralogist, 104, 140-149).
November 2019
Fu et al. (p. 1533). Granite is one of the most abundant rock types of the continental crust. Heat transfer and temperature distribution in the crust could be strongly influenced by the thermal properties of granite. In this study, the thermal diffusivity and thermal conductivity of four natural granitoids are simultaneously measured under high pressure and high temperature. The calculated geotherms suggest that the presence of partial melting induced by muscovite or biotite dehydration likely occurs in the upper-middle crust of southern Tibet. This study provides new insights into the origin of low-velocity and high-conductivity anomaly zones revealed by geophysical observations in this region.
Trudu et al. (p. 1546). A long-standing issue is the presence of linear Si-O-Si bridges in silicates. We investigate the structure of the all silica zeolite ferrierite and explain the origin of the instability of its linear linkages. The relationship between linear Si-O-Si angles and metastability of a zeolite framework is a key to better understanding of the possible phase-transition mechanisms of open-framework (alumino-) silicate minerals at elevated pressure and temperature conditions of geological relevance.
Díaz et al. (p. 1556). Composite materials based on natural stilbite zeolites from Faroe Islands have been prepared where nanoparticles of hydroxyapatite are grown in a particular orientation on the zeolite external surface. Such nanoparticles are very efficient for the removal of fluoride from water through anion-exchange with hydroxides, allowing reducing the fluoride concentration below the admissible level.
Flahaut et al. (p. 1578). The characteristics and mineralogic diversity of the Italian Solfatara fumarolic field were surveyed as an analog for some of Mars' hydrothermal environments. Several techniques of mineral identification (VNIR spectroscopy, Raman spectroscopy, XRD) were used and reveal the presence of varying alteration patterns as a distance from the crater center and vents. The authors discuss the possibility of identifying such environments -which represent prime astrobiological targets- on Mars.
Xie et al. (p. 1565). The results of our research have two major implications. Firstly, it can be used for continuous monitoring of burial sites and other geothermal engineering projects. The acoustic emission (AE) energy distribution exponent can easily be measured using highly sensitive thermostability AE sensors attached to the host rock or normal sensors located around the burial site. The data analysis method used in this study also can be easily implemented through online evaluation of the AE signals. In addition, the relationship between mineral structure and avalanche dynamics is the focus of many studies of crackling noise. Here we show that avalanches are heavily influenced by temperature and that local 'crumbling' is a key mechanism for softening of geomaterials (rather than elastic softening). This result will now attract more theoretical and simulation studies in this field.
Williams et al. (p. 1585). The Adirondack Highlands have been held as an example of the middle crust during continental collision. Gneissic rocks interpreted to have undergone partial melting are common, but the timing and setting of melting are uncertain. This contribution uses in-situ monazite petrochronology to suggest that melting occurred at two main times, ca. 1.15 Ga and 1.05 Ga. Further, the amount of melting in the second event depends on the degree of melt segregation in the first event. High-resolution microprobe mapping and analysis provide a powerful tool for linking geochronology to deformation fabrics and metamorphic reactions.
Oka et al. (p. 1603). We performed melting experiments on Fe-O alloys to 204 GPa and determined the change in liquidus phase diagram in the Fe-FeO system, in particular the eutectic liquid composition, with increasing pressure on the basis of ex-situ textural and chemical characterizations of recovered samples. Our main conclusions are the following: (1) The oxygen content in the Fe-FeO eutectic liquid increases remarkably upon reduction in the immiscible two-liquids region to ~40 GPa. (2) Such observation is different from that in the recent study by Morard et al. (2017) that was mainly based on in-situ XRD measurements. The difference is likely attributed to the contamination by carbon that occurred in their experiments and some of ours. (3) Our data to 204 GPa allows extrapolation to 330 GPa, indicating the eutectic liquid includes ~15 wt% O at the ICB. We estimated the range of possible liquid core compositions in Fe-O-Si-S-C, which crystallize solid Fe that forms a dense inner core and account for the density and sound velocity observed in the outer core.
Cuadros et al. (p. 1608). The controls on Fe(III) distribution between tetrahedral and octahedral sites in phyllosilicates have been a matter of great interest in order to understand the interplay between formation environment and crystal-chemical factors during crystallization of Fe-phyllosilicates. Here, for the first time, we present a model describing the controls of Fe(III) distribution between octahedral and tetrahedral sites in all 2:1 phyllosilicates. The results are from a survey of 70 analyses covering a wide range of dioctahedral and trioctahedral phyllosilicates, including endmember and interstratified minerals. Dioctahedral phyllosilicates have a steric control whereby tetrahedral Fe(III) is only allowed if at least five out of six octahedral atoms are larger than Al (typically Fe[III], Fe[II], Mg). In this expanded structure tetrahedral sites can accommodate Fe(III). After this threshold, ~73 % of further Fe(III) atoms occupy tetrahedral sites. In trioctahedral 2:1 phyllosilicates there is no steric hindrance; on average, Fe(III) enters tetrahedral and octahedral sites in similar proportion, and the only control on Fe(III) abundance is Fe(III) availability during crystallization.
Zhang et al. (p. 1620). In this study, Fe-bearing phase E (one of the dense hydrous magnesium silicates) coexisting with ringwoodite and wadsleyite has been synthesized at 18 and 19 GPa, and 1400 °C. The long heating duration time (27 hours) of syntheses implies that phase E may be a stable component of the mantle transition zone at near-geotherm temperatures, if transition-zone regions adjacent to subducting slabs are hydrated by fluids generated at the top of the lower mantle.
Maner IV et al. (p. 1625). The enrichment of Mn to the point of spessartine saturation in granitic liquids is explained using experimentally derived mineral-melt partition coefficients for garnet, tourmaline, and cordierite. The partition coefficients are used in a Rayleigh fractional crystallization model to show the amount of crystallization required to concentrate enough Mn to saturate melt with respect to spessartine. The model indicates that approximately 95% of an S-type granitic melt must crystallize to achieve spessartine saturation.
Cherniak and Watson (p. 1538). This paper reports on experimental measurements of Al and Si diffusion in rutile, investigating effects of oxygen fugacity and orientation on diffusion. Little diffusional anisotropy is observed, with only minor effects of oxgyen fugacity over the range of conditions studied. Si and Al are among the slowest-diffusing species in rutile. These slow diffusivities indicate that the recently developed Al-in-rutile crystallization geothermobarometer (Hoff and Watson, 2018) will be more resistant to diffusional alteration than the Zr-in-rutile crystallization thermometer.
Wei et al. (p. 1650). We have determined the compressional-wave velocity (VP) of Ne at simultaneously high P-T condition up to 53 GPa and 1100 K. The obtained results provide crucial constraints on the combined effect of pressure and temperature on VP. In particular, VP of super-critical Ne fluid exhibit a weak dependence on pressure above 800 K and 20 GPa. The velocity contrast in VP between the super-critical Ne fluid and the solid phase increases with increasing temperature.
O'Bannon, III and Williams (p. 1656). Topaz is an important subduction zone mineral that transports water and fluorine into the deep Earth. Previous studies have characterized the bulk crystallographic response of topaz to high-pressure. Here we used Cr3+ luminescence which is a sensitive site-specific probe of the bonding environment of the Al-sites in topaz. We have characterized for the first time the high-pressure behavior up to 60 GPa of all three Al environments simultaneously. Our results reveal that topaz is one of the most metastable tetrahedrally coordinated silicates that is known.
Pittarello et al. (p. 1663). Mesosiderite meteorites consist of a mixture of crustal basaltic or gabbroic material and core metal, and have experienced an extremely slow cooling rate from ca. 550 °C, as recorded in the metal (0.25-0.5 °C/Ma). Here we present a detailed investigation of two generations of exsolution features in a pyroxene from the Antarctic mesosiderite Asuka 09545, in an attempt to constrain the evolution of pyroxene from 1150 to 570 °C.
Epp et al. (p. 1688). Pyromorphite-group minerals are the most common Pb-secondary minerals worldwide and their halogen incorporation is not only governed by crystallographic effects, but more predominantly by the fluid-composition. This enables further fluid monitoring applications such as deciphering single fluid flow episodes and the determination of surface processes. Furthermore, we developed a methodology to simultaneously determine Cl, F, Br and I. Since halogen data, especially concerning Br and I, is generally scarce, this is of interest to the broad geoscience audience.
Feisel et al. (p. 1689). We applied the diffusion couple technique on a homogeneous rhyodacitic melt over a temperature range relevant for natural systems. We analyzed the samples for diffusion induced concentration profiles using an electron microprobe. Our analytical conditions allow for very low F and Cl detection limits, which in turn results in very clear and smooth F- and Cl-concentration gradients across the experimental couples. Our results show that both F and Cl exhibit the same temperature dependence on diffusivity, but the diffusion of F is significantly faster than that of Cl. This means that the potential for diffusive fractionation exists and may occur especially under conditions of slow magma ascent and bubble growth. Compared to other melt compositions for which F and Cl diffusion data is available, our diffusivities are generally slower and correlate well with the atomic radius of the diffusing ion. With this investigation we have conducted the first study that quantifies and compares the diffusion rates of F and Cl elements in natural high-silica melts.
October 2019
Kellogg et al. (page 1365) discuss two alternative hypotheses on the origin of carbon in Earth's continental crust. In the first hypothesis, the atmosphere is extracted directly from the early atmosphere through the Urey reaction. In the second hypothesis, the atmosphere is extracted from the mantle by volcanism. The authors argue that the current equilibrium content of carbon in the atmosphere is a balance between injection by volcanism and loss by the Urey reaction.
Stewart et al. (page 1369) discuss carbonation and decarbonation reactions, which are the primary mechanism for transferring carbon between the solid Earth and the ocean-atmosphere system, and their implications for Earth's climate and planetary habitability. These processes can be broadly represented by the reaction: CaSiO3 (wollastonite) + CO2 (gas) = CaCO3 (calcite) + SiO2 (quartz). The authors summarize their significance as part of the Deep Carbon Observatory’s “Earth in Five Reactions” project.
Sugiura et al. (page 1381) investigated the effects of PO4 on CaCO3 formation from calcium sulfate anhydrate (CaSO4) at high-pH conditions, under which calcite is likely to form. PO4 regulates calcite formation and growth by adsorbing onto the surface of calcite. Therefore, under high PO4 concentrations, calcite crystals become small and porous. In addition, vaterite is likely to form to counteract the thermal instability of the solution. The results also indicate that PO4, which is an essential component of carbonate apatite, can control the physico-chemical properties of CaCO3, which is a precursor of carbonate apatite, a bone-replacement material.
Vennari and Williams (page 1389) conducted high-pressure Raman and Nd3+ luminescence spectroscopy of bastnäsite, a rare earth element (REE)-bearing carbonate that has geologic and economic importance due to its high REE content. The high-P measurements on bastnäsite allowed investigation of bonding changes in carbonate ions juxtaposed with halogens and REE within a carbonate matrix. This study provides evidence for how REE ions are coordinated with carbonate ions at depth with implications for both REE and carbon retention at depth.
Hill et al. (page 1402) observed nanocrystals of -cristobalite that are epitaxially exsolved in cores of ultrahigh-pressure (UHP) clinopyroxene from the Bohemian Massif, Czech Republic, using transmission electron microscopy. This study revealed vacancies and OH- in high-pressure pyroxene that formed during subduction. When the host rock is exhumed, the incorporated OH- migrates out from the UHP pyroxene structure together with vacancies and excess silica in multiple forms of glass and cristobalite/amphibole precipitates. If the pyroxene-bearing slab subducts further into the mantle, the pyroxene may release the incorporated OH- into mantle during its phase transformation to garnet and/or perovskite.
Xu et al. (page 1416) investigated phase relations in the AlOOH–FeOOH system at 15–25 GPa and 700–1200 °C. δ-AlOOH and ɛ-FeOOH, which have a CaCl2-type structure, were found to form partial solid solutions over wide ranges of pressures and temperatures. This result implies that CaCl2-type hydroxides may carry a certain amount of hydrogen into deeper regions of Earth interior and even Fe-rich Martian core in cold subduction regions.
Tamer et al. (page 1421) investigated analyst and etching protocol effects on the reproducibility of apatite confined fission-track length measurement, and ambient-temperature annealing at decadal timescales. The authors show that fission-track etching is a continuous process, and they illustrate possible differences between two major apatite fission-track etching procedures, analyst biases on track recognition and measurements, and long-term ambient temperature fission-track annealing.
Wang et al. (page 1436) developed a new approach combining multiple scanning/transmission electron microscopic techniques (HRTEM, STEM with EDS) to identify mixed layers in a chlorite sample. Since interstratified mineral phases appear only as a few layers, they are difficult to identify by conventional methods. Via lattice spacings, HRTEM and HAADF images, and EDS mapping, the authors distinguished the parallel-growing monolayer mica and pyrophyllite from chlorite domains. This method may be extended to the studies of other minerals, especially those e-beam sensitive phases.
González-García et al. (page 1444) measured the diffusive exchange of six major elements in diffusion couple experiments, using natural shoshonitic and rhyolitic melts as endmembers. Experiments were run at atmospheric pressure and temperatures between 1230 and 1413 °C, and the concentrations were measured by electron microprobe. Arrhenius relationships were obtained. The results suggest that diffusive coupling is prevalent, i.e., all elements show similar diffusivities. These also expand a previous database with variably hydrous melts, allowing establishment of a linear relationship between diffusivity and the square root of water concentration. The results are of particular interest in the study of mass transfer phenomena in alkaline volcanic systems.
Fei et al. (page 1455) investigated the factors that control the crystal morphology and chemistry of garnets in the Cuihongshan polymetallic skarn deposit, China. Major findings include: 1) Strain from lattice mismatch at substitution and twin probably causes the birefringence in garnets; 2) REE concentrations are probably influenced by the relative proportion and temperature of the system; 3) LREE-HREE fractionation of garnets can be attributed to relative compositions of grossular-andradite system; and 4) W and Sn concentrations in garnets can be used as indicators for the exploration of W-Sn skarn deposits.
Vereshchagin et al. (page 1469) report a new mineral, gasparite-(La), La(AsO4), from Mn ores of the Ushkatyn-III deposit, Central Kazakhstan and from alpine fissure in metamorphic rocks of the Wanni glacier, Binn Valley, Switzerland. Gasparite-(La) has a monoclinic structure with the space group P21/n. The samples from the Ushkatyn-III deposit and Wanni glacier have different origins. La/Ce and As/P/V ratios in gasparite-(La) may serve as an indicator of formation conditions. Raman spectra of gasparite-(La) can be used to identify its origin.
Stachowicz et al. (page 1481) investigated the cation ordering, valence states, and symmetry breaking in a Nb-rich chevkinite-(Ce) from the Biraya rare-metal deposit, Russia, using electron probe microanalysis, X-ray diffraction, and photoelectron spectroscopy. Annealing the sample at 750 °C resulted in the structural transformation C2/m→P21/α, which defines chevkinite stability relations. This transformation seems to be a rapid version of a naturally occurring process that possibly involves twinning of the crystals. Nb-rich chevkinite-(Ce) occurs naturally as two polymorphs, C2/m and P21/α, the latter of which is the stable form under ambient conditions. There are some distinct differences between the two structures, such as their mean M-O distances and site scattering values of particular sites, which can be associated with the redistribution of lighter cations, mainly Mg2+, within the structures.
Qiu et al. (page 1487) present geochronological data showing that Zr and REE mineralization events of the Baerzhe rare-metal deposit in Northeastern China spanned roughly two million years. The Zr and REE mineralization events are temporally distinct but both are genetically related to the same mineral system. Three distinct types of magmatic through deuteric zircon along with hydrothermal monazite are considered the most significant ore minerals.
Marger et al. (page 1503) present one of the first in-situ oxygen isotope analyses from zoned tourmaline crystals using secondary ion mass spectrometry. The authors document that tourmaline is refractory for oxygen isotope compositions, and can be used to track fluid-rock interactions. They developed a set of tourmaline reference materials spanning the composition range of the most abundant metamorphic and igneous tourmalines. By combining a detailed petrologic study with the oxygen isotope compositions of quartz and tourmaline, this study demonstrates that oxygen isotopes measured in-situ can be used to resolve the debated question about whiteschist formation in the western Alps. It also shows that the protolith of the whiteschists were Hercynian granites, and the metasomatism predated Alpine metamorphism.
Ma et al. (page 1521) report a new mineral, chenmingite, FeCr2O4, with a Pnma CaFe2O4-type structure. This is the third high-pressure mineral the authors have discovered (in addition to ahrensite and tissintite) during their nanomineralogy investigation of the Tissint shergottite. Further, chenmingite is one of thirteen newly-approved high-pressure minerals discovered in shocked meteorites since 2013. Their meteoritic occurrences can also inform the study of deep earth systems at high pressures and temperatures.
Satta et al. (page 1526) determined the elastic properties of phase E single crystals using Brillouin spectroscopy. The results show that phase E has the lowest isotropic compressional and shear velocities among the Dense Hydrous Magnesium Silicate (DHMS) phases and other relevant upper mantle and transition zone minerals. This study suggests that even small amounts of phase E can significantly reduce acoustic wave velocities in isotropic aggregates, opening the possibility of linking the presence of water-rich phase E assemblages with velocity anomalies detected by seismology in subduction scenarios.
Chen (page 1530) reviews a book entitled “Understanding Minerals and Crystals” by Terence McCarthy and Bruce Cairncross (2015), Struik Nature, 312 pp. The focus of this book is mineral resources education, and it distinguishes itself from other mineralogy books by containing rich conceptual and visual content as an introductory guide for mineralogy.
Johnson (page 1531) reviews a book entitled “A Practical Guide to Rock Microstructure (2nd edition)” by Ron H. Vernon (2019), Cambridge University Press, 440 pp. This book contains detailed descriptions of multiple hypotheses on the generation of each texture and provides extensive references for exploring the origin of textures in more depth. It can serve as a reference for graduate students, professionals, or undergraduate research projects.
September 2019
Steele-MacInnis this issue (page 1217) highlights the recent publications by Gavrilenko et al. (July issue, p. 936; https://doi.org/10.2138/am-2019-6735) that investigated the contradiction between experimental studies that predict that primitive arc melts may contain up to and greater than 15 wt% H2O, but that, curiously, the breadth of analytical data on melt inclusions consistently show to have lower values, mostly less than 6 wt%. Gavrilenko et al. showed experimentally that this apparent contradiction is likely rooted in a “quench control,” whereby wetter melts are incapable of being quenched to glass. This study neatly reconciles experimental and observational data and provides a key insight into how best to analyze and interpret the H2O contents of melt inclusions from subduction settings. https://doi.org/10.2138/am-2019-7124
Takahashi (page 1219) discusses the breakthrough by Sato and Ozawa’s study of the lithosphere under the Japanese subduction zone (page 1285; https://doi.org/10.2138/am-2019-6858). The LABZ (lithosphere-asthenosphere boundary Zone) under oceanic plates, young continents and subduction zones has remained largely in question, due to the lack of proper geobarometry for spinel lherzolite facies. Y. Sato and K. Ozawa, for the first time, describe the petrologic features of the LABZ beneath a subduction zone. A reconstructed LABZ beneath Ichinomegata is similar to those reported from the bottom of the subcratonic lithospheric mantle in various aspects, but the boundary layer beneath Ichinomegata is much shallower (40-60 km) and colder (~1050 °C). https://doi.org/10.2138/am-2019-7113
Hikosaka et al. (page 1356) present the results of their study of the stability of Fe5O6 and Fe4O5 at high pressures. While several new iron oxides were discovered above 10 GPa in the last decade, their stabilities at high pressure and temperature (P-T ) are not understood yet. Here we examined phase relations in both Fe5O6 and Fe4O5 and found that both decompose into the FeO + Fe3O4 assemblage above ~40 GPa. It indicates that the intermediate compounds between FeO and Fe3O4 (i.e., Fe4O5, Fe5O6, Fe7O9, and Fe9O11) are formed only in the deep upper mantle to the shallow lower mantle. https://doi.org/10.2138/am-2019-7097
Boujibar et al. (page 1221) investigate the origin of the elevated concentration of K relative to U and Th on the surface of Mercury found by the MESSENGER mission. These measurements suggested an enrichment of volatile elements (such as K) relative to refractory elements (such as U and Th), in comparison to Earth and Venus. They conducted experiments in an O-depleted environment typical of Mercury's formation, to assess whether K, U, and Th were distributed differently between the different layers inside Mercury (mantle, core, and possible sulfide layer) and found that if an iron sulfide layer exists between the core and mantle, more U and Th then K would be sequestered in that layer, leading to higher K/U and K/Th ratios in the crust and mantle, measurable on the surface by MESSENGER spacecraft. By taking into account this possible sulfide layer, Mercury appears to be as volatile depleted as the Earth and Venus and more depleted than Mars. These results confirm the depletion of volatile elements in the inner part of the Solar System, where Mercury, Venus, and Earth formed. https://doi.org/10.2138/am-2019-7000
Lee et al. (page 1238) study a new magnetic mineral (valleyite) that was discovered in a basaltic scoria. Valleyite has sodalite-type structure with low density. The magnetization hysteresis loop indicates the magnetic exchange coupling between valleyite (soft magnet) and luogufengite (hard magnet), which will help us better understand magnetic properties and paleo-magnetism of basaltic rocks. The new mineral with the sodalite-type cage structure, and the magnetic property is potentially a functional material. https://doi.org/10.2138/am-2019-6856
Geiger et al. (page 1246) analyzed the magnetic and and Néel temperature, TN, properties of four silicate solid solutions. The four systems are: fayalite-forsterite olivine, Fe2+2SiO4-Mg2SiO4, and the garnet series, grossular-andradite, Ca3(Alx,Fe3+1-x)2Si3O12, grossular-spessartine, (Cax,Mn2+1-x)3Al2Si3O12, and almandine-spessartine, (Fe2+x,Mn2+1-x)3Al2Si3O12. https://doi.org/10.2138/am-2019-6839CCBYNCND
Mukherjee et al. (page 1256) demonstrate that the trace-element concentrations (and their ratios) of sedimentary pyrite in three black shale formations of the McArthur Basin provide evidence for a gradual increase in atmospheric oxygenation from 1730 to 1360 Ma. They observe a marked change in pyrite sulfur isotopic compositions in the three black shale formations, i.e., a marked increase in mean δ34Spyrite values from the Wollogorang Formation to the Velkerri Formation. This change is possibly indicative of the expansion of oxygenated waters and decreasing areal extent of anoxia. Results from both techniques have major implications on the atmospheric redox evolution in “Boring Billion”, a period known to witness non-fluctuating redox conditions. https://doi.org/10.2138/am-2019-6873
Ohira et al. (page 1273) investigate the δ-(Al,Fe3+)OOH solid solution, one of the hydrous phases stable under lower mantle conditions. This study shows that the δ-(Al,Fe)OOH could cause an anomalously high ρ/νΦ ratio at depths corresponding to the spin crossover region (~900 to ~1000 km depth), whereas outside the spin crossover region a low ρ/νΦ anomaly would be expected. The results suggest that the presence of δ-(Al,Fe)OOH could be detectable and provide new insight for understanding the heterogeneity in the lower mantle. https://doi.org/10.2138/am-2019-6913
Sato and Ozawa (page 1285) develop a method for accurate estimation of derivation depths of shallow upper mantle materials (xenoliths) occurring as fragments in volcanic ejecta. Its application to xenoliths from the Ichinomegata maar in the back-arc side of Northeast Japan Arc was successful in revealing, for the first time, the structure of lithosphere-asthenosphere boundary zone in arc settings, which was found to consist of water-saturated lithospheric mantle and underlying partially molten asthenosphere. https://doi.org/10.2138/am-2019-6858
Li et al. (page 1307) investigate ammonia-water mixtures that are an important component of the giant ice planets. Using Raman spectroscopy, they investigate the phase stability of ammonia hydrate (AHH) in different ratios at high pressures. The experimental results show that all the ammonia hydrate will dehydrate into ammonia hemihydrate and ice-VII. More importantly, ice-VII will gradually separate out from ammonia hydrate and could grow to be a single crystal. They also measure the sound velocity and elasticity of ammonia hydrate and single-crystal ice-VII using Brillion scattering up to 53 GPa. Measured elasticity of ice-VII shows anomalous variations at 10-20 GPa and 42-53 GPa, respectively, which is associated with the structural change of single-crystal ice-VII. Modeling the velocity of AHH and ice-VII mixture indicates that the mantle of ice giants may have strong anisotropies in velocity. https://doi.org/10.2138/am-2019-7057
Diego Gatta et al. (page 1315) re-investigate the crystal structure and crystal chemistry of kurnakovite by a multi-methodological approach (i.e., single-crystal neutron diffraction at room and low temperature, titrimetric analysis for the determination of B and Mg content, inductively coupled plasma atomic emission spectroscopy for REE and other minor elements, ion selective electrode for F, high-T mass loss for H2O content). Kurnakovite does not act as a geochemical trap of industrially relevant elements (e.g., Li, Be, or REE). It is a potentially B-rich addition to concretes used for the production of radiation-shielding materials due to the elevated ability of 10B to absorb thermal neutrons, because it can mitigate the risk of releasing undesirable elements, for example sodium, which could promote deleterious reactions affecting the durability of cements. https://doi.org/10.2138/am-2019-7072
Yuan et al. (page 1323) treated diatomaceous opals using Focused Ion Beam (FIB) to characterize separately the composition of the internal siliceous parts and the surface/near-surface locations by means of EDS on a TEM. The results demonstrate that minor elements, such as Al, Fe, Ca, and Mg, conclusively exist within the siliceous framework. In addition, foreign minerals (mostly aluminosilicates) largely contribute to the bulk and surface properties of diatomaceous opal. These findings renew the knowledge that diatomaceous opal is “pure” silica mineral or materials. https://doi.org/10.2138/am-2019-6917
Anzolini et al. (page 1336) report the discovery and full description of a new mineral, nixonite, ideally Na2Ti6O13 (IMA 2018-133). Nixonite was found as a part of a complex reaction rim around a rutile grain within a heavily-metasomatized pyroxenite xenolith from the Darby kimberlite field, beneath the west-central Rae Craton, Canada. Nixonite is the first occurrence in nature of Na2Ti6O13, which was previously known only as a synthetic material used in batteries, and is the Na-rich analogue of jeppeite, K2Ti6O13, which is commonly observed as a groundmass mineral in lamproites. This finding represents not only the first natural occurrence of Na2Ti6O13, but also the unique coexisting assemblage of the minerals rutile, priderite, perovskite, freudenbergite, ilmenite, and nixonite. We suggest that this complex Na-K-Ti rich metasomatic mineral assemblage may have been produced by an unusual metasomatic melt that percolated through the lithospheric mantle beneath the Darby field. https://doi.org/10.2138/am-2019-7023
Meyer et al. (page 1345) report the discovery of the new mineral Goldschmidtite. New minerals are not common after years of mineral research, and new perovskite-structured minerals are even more rare. This new mineral, coming from a diamond, highlights a case of extreme metasomatic conditions in the lithosphere. Remarkably, this mineral is a well-known synthetic in ceramic science. It is also, justifiably, named after one of, if not the, founders of geochemistry, whose scientific impact is well known and far reaching. https://doi.org/10.2138/am-2019-6937
Chi Ma and Alan Rubin (page 1351) report the discovery of the new mineral edscottite that occurs with low-Ni iron (kamacite), taenite, nickelphosphide (Ni-dominant schreibersite), and minor cohenite in the Wedder-burn iron meteorite, a Ni-rich member of the group IAB complex. The end-member formula is Fe5C2. The new mineral is named in honor of Edward (Ed) R.D. Scott, a pioneering cosmochemist at the University of Hawai’i at Manoa, for his seminal contributions to research on meteorites. Since the Carbon Mineral Challenge was launched in 2015, edscottite is the first carbide mineral approved by the IMA-CNMNC. https://doi.org/10.2138/am-2019-7102
Belakovskiy et al. report on the new mineral names (page 1360). The paper contains entries for nine new minerals, including argentotetrahedrite-(Fe), bytízite, calamaite, fluorlamprophyllite, honzaite, katerinopoulosite, meitnerite, melcherite, and rozhdestvenskayaite-(Zn). http://dx.doi.org/10.2138/am-2019-NMN104914
August 2019
This issue of American Mineralogist starts with the MSA Presidential Address given by Brown (Past-President) and Johnson (page 1065): Metamorphism and the evolution of subduction on Earth. Whether Earth always had plate tectonics or, if not, when and how a globally-linked network of narrow plate boundaries emerged are matters of debate. Here the authors use a dataset of the pressure (P), temperature (T), and age of metamorphic rocks to evaluate secular change associated with subduction and collisional orogenesis at convergent plate boundaries. The widespread appearance of two types of metamorphism with different thermobaric ratios (T/P) at the beginning of the Neoarchean is interpreted to be evidence of the stabilization of subduction during the emergence of plate tectonics.
Cerantola et al. (page 1083) studied the stability of siderite (FeCO3) in the Earth's lower mantle. The experimental results using Fe K-edge X-ray absorption near-edge structure (XANES) spectroscopy are supported by first-principles calculations and match well with recently reported observations on FeCO3 at extreme conditions. At conditions of the mid-lower mantle, ~50 GPa and ~2200 K, FeCO3 melts and partially decomposes to high-pressure Fe3O4. Diamond and oxygen are also inferred products of the reaction. Moreover, the incongruent melting of FeCO3 could be a key mechanism that partially preserves FeCO3 from decomposition, potentially supporting carbon influx into the deep Earth via carbonate subduction.
Pan et al. (page 1092) conducted a comparative study of fluid inclusions in coexisting wolframite and quartz crystals from a giant vein-type tungsten deposit, South China. A combined in situ analytical procedure, including cathodoluminescence (CL) imaging, infrared microthermometry, Raman microspectroscopy, and fluid inclusion LA-ICP-MS analysis, was used to reconstruct the detailed fluid evolution history. Based on elaborate petrography on the crystal scale, fluid inclusions in wolframite and coexisting quartz are found to exhibit distinct chemical signatures, despite sharing similar overall ranges of salinity and homogenization temperature. The revealed fluid evolution history provides insight into the fluid source and the wolframite deposition mechanism in vein-type tungsten deposits.
Rusiecka and Baker (page 1117) obtained new data on monazite and xenotime solubility in boron-bearing rhyolitic melts at 1000-1400 °C and 800 MPa in a piston cylinder apparatus, as well as the diffusivity of the components of these two minerals (LREE, P, and Y). This study provides first-ever data on the solubility of xenotime-(Y) in rhyolitic melt, as well as new data on monazite solubility in the boron-bearing rhyolitic melt. The authors also discussed implications of the results on the understanding of natural, silicic (granitic/rhyolitic), and magmatic systems.
Moy et al. (page 1131) report a new low-accelerating voltage electron probe microanalysis (EPMA) method to quantify Fe using the combined Fe L X-ray lines. This method takes advantage of the reduction of the electron beam offered by Schottky field emission source in conjunction with the reduction of the electron interaction volume permitted by decreasing the accelerating voltage from traditional 15-20 kV to 7 kV, to accurately quantify submicrometer-sized features. The method has been successfully applied to olivines as a test bench. Compared with large errors using the traditional EPMA quantification method using the Fe L X-ray line, the new method gives much more accurate results, with average relative deviations of 3.6% from actual compositions.
Locmelis et al. (page 1143) present the results of a comprehensive study on the concentrations of first-row transition elements, Ga and Ge in olivine from komatiites measured via laser ablation ICP-MS. The data show that (1) elevated Ga/Sc ratios in olivine reflect garnet retention in the komatiite source, (2) high Ge contents in olivine may be indicative of melting under hydrous conditions, (3) V/Sc and Mn/Fe ratios in olivine can potentially be used to constrain local oxygen fugacity in the komatiite magma, and (4) Cu-abundances reflect the sulfide saturation state of a komatiite magma during olivine crystallization.
Tao et al. (page 1156) report a mechanism of hydrotalcite (Ht) conversion into saponite after formed by the hydrothermal alteration of metal oxides. The conversion was through a hydration-dissolution-precipitation pathway. It was favored under the conditions of relatively low Mg/Al ratios with high Al and Si contents, and greatly accelerated by the Al3+—Si4+ substitution in silicate oligomers to generate the extra negative charge in tetrahedral sheets. During the process, CO2 was initially incorporated into the interlayer of Ht as CO32-, but was expelled by the formation of saponite, due to the layer charge reversal from positive in Ht to negative in saponite. These findings provide an explanation for the rare occurrence of hydrotalcite deposits on the Earth's surface.
Křížová et al. (page 1165) report the occurrence of shenzhuangite close to its ideal end-member composition (NiFeS2), Ni1.007Fe0.998Cu0.016Co0.058S1.922, in Australasian Muong Nong-type tektites from Laos. This was the first discovery of shenzhuangite in terrestrial materials; originally it was found in meteorite Suizhou. This shenzhuangite was identified by electron probe microanalysis and electron back scatter diffraction. The authors also presented a Raman spectrum with a tentative assignment of spectral bands based on the analogy with synthetic chalcopyrite-structured phases.
Wang et al. (page 1173) conducted acoustic velocity measurements of a natural orthopyroxene, (Mg1.77Fe0.22Ca0.01)Si2O6, up to 13.5 GPa and 873 K. It is known that the end-member orthoenstatite, Mg2Si2O6, undergoes velocity softening at high pressure and room temperature, due to its transition to the metastable, high-pressure clinoenstatite. This study reports a similar, high-pressure velocity softening in Fe-bearing orthopyroxene at temperatures up to at least 673K, providing an upper bound on the P-T conditions where such softening may occur in subduction zones. The direct measurement of velocity jump across the orthopyroxene to high-pressure clinopyroxene transition further proves that it could be a possible contributor to the seismic X-discontinuity.
Yin et al. (page 1180) present a potential mechanism for the uptake of trace element at a fluid-mineral interface in hydrothermal systems. More specifically, they determine the role of mineral nanoparticles at a fluid-magnetite interface using high-resolution transmission electron microscopy. The results show that the Al concentration in magnetite measured on a micron-scale is caused by three different effects: Al solid solution, Al-rich nanometer-sized lamellae and zinc spinel nanoparticles in the host magnetite, and the authors propose a genetic relationship among the three phases. The fluid-mineral interface in this mechanism has been repeatedly utilized during crystal growth, providing an efficient way for the uptake of trace element from a related undersaturated bulk fluid.
Deng and Lee (page 1189) investigated the effects of the electronic spin transition of iron on the melting temperature of Earth's two most abundant minerals, ferropericlase, and bridgmanite, using both Lindemann's Law and thermodynamic analyses. Lindemann's Law predicts a negligible melting temperature depression for bridgmanite but a substantial depression for ferropericlase across the spin transition of iron, consistent with extant experimental results. Thermodynamically, the melting depression likely derives from a more negative Margules parameter for a liquid mixture of high- and low-spin end-members as compared to that of a solid mixture. This melting depression across the spin transition of iron may be the process responsible for the formation of a deep molten layer during the crystallization of a magma ocean in the past, and a reduced viscosity layer at present.
Chen et al. (page 1197) investigated epidote spherulites and radial euhedral epidote aggregates in a greenschist facies metavolcanic breccia hosting an ultrahigh-pressure eclogite in Dabieshan, China, and discussed the implications for dynamic metamorphism. Because these non-equilibrium textures would recrystallize into equilibrium ones if the P-T -H2O conditions were maintained sufficiently long, they likely formed in response to P-T and fluid pulses, possibly related to seismicity.
Liu et al. (page 1213) synthesized a new (Mg0.5Fe3+0.5)(Si0.5Al3+0.5)O3 LiNbO3-type phase (space group R3c) at lower-mantle conditions. Fe3+ and Al3+ cations substitute into A (Mg2+) and B (Si4+) sites, respectively, through a charge-coupled substitution mechanism. This phase is probably recovered from bridgmanite at lower-mantle conditions by a diffusionless transition because of the displacement of A cations and distortion of BO6 octahedra on releasing pressure. Bridgmanite can thus contain the FeAlO3 component (50 mol%) beyond the previously reported solubility limit (37 mol%). The present study shows that the Earth's most abundant elements form a new FeAlO3-dominated LiNbO3-type compound from bridgmanite at lower mantle conditions. It provides new insight into the complicated crystal chemistry of LiNbO3-type phase/bridgmanite and constrains the P-T conditions for shocked meteorites.
July 2019
Swanner et al. (page 917) in their article “Fate of cobalt and nickel in mackinawite during diagenetic pyrite formation” report their investigations of Ni and Co incorporation into freshly-precipitated mackinawite, and after experimental diagenesis to pyrite at 65 °C. Fe and S K-edge micro-X-ray absorption near edge spectroscopy identified the oxidation state and mineralogy within experimentally synthesized and diagenetically transformed minerals. Results indicate that Co and Ni may inhibit the transformation of mackinawite to pyrite or slow it down. Cobalt concentrations in the solid diminished by 30% during pyrite transformation, indicating that pyrite Co may be a conservative tracer of seawater or porewater Co concentrations. Ni concentrations increased several-fold after pyrite formation, suggesting that pyrite may have scavenged Ni from the dissolution of primary mackinawite grains. Nickel in pyrites thus may not be a reliable proxy for seawater or pore water metal concentrations.
Liu et al.’s research on page 929 in the “Influence of aluminum on the elasticity of majorite-pyrope garnets” used an ultrahigh pressure convection technique to fabricate a series of gem-quality majorite-pyrope garnets and measured the velocity of these garnets by means of ultrasonic interferometry measurements. They found that both velocity and elastic moduli increase linearly with increasing Al along the majorite-pyrope system. The Al component plays a dominant role for the variation of elasticity (velocity and modulus) for majorite-pyrope garnets, while the phase transition due to cation ordering or disordering cannot significant affect these elastic properties. Therefore, seismic velocity modeling of a garnet-bearing mantle transition zone is more associated with garnet’s composition rather than the phase transitions due to cation order or disorder in garnet.
Gavrilenko et al.’s article on page 936, “The quench control of water estimates in convergent margin magmas,” explores the limits of melt inclusions (MIs) as hydrous magma recorders in an experimental study, showing that there is a limit of dissolved H2O that mafic glassy MIs can retain. These results show there is potential bias in the glassy MI data set; they can only faithfully record pre-eruptive H2O contents in the upper-most part of the Earth’s crust where H2O-solubility is low. The current MI database cannot be used to robustly estimate the full range of arc magmas and therefore assess volatile budgets in primitive or evolved compositions. Such magmas may contain much larger amounts of H2O than currently recognized, and the diversity of magma evolutionary pathways in subduction zones is likely being significantly underappreciated.
Yin et al. (page 949) studied the “Textural and chemical variations of micas as indicators for tungsten mineralization: Evidence from highly evolved granites in the Dahutang tungsten deposit, South China.” They found that micas are effective indicators not only for the magmatic-hydrothermal evolution of granite but also for the tungsten mineralization. The texture of zoned micas and geochemical variations of micas are important for reconstructing tungsten ore-forming processes, including the enrichment and transportation of tungsten during the magmatic-hydrothermal evolution. Tungsten is unlikely to be deposited directly in the granite, and reducing fluids and fluid-rock interaction play an import role in forming large ore deposits.
Hirth et al. (page 966) in “A topological model for defects and interfaces in complex crystal structures” introduce a tractable method for applying the topological model to characterize defects in these complex materials. They illustrate how structural groups, each with a motif containing multiple atoms, provide lattices and structures that are useful in describing dislocations and disconnections in interfaces. They illustrate the model for twinning in albite owing to its potential application for constraining the rheological properties of the crust at conditions near the brittle-plastic transition, where plagioclase is a major constituent of common rock types. The concept of structural groups makes an analysis of the twinning process easier in complex minerals and explicitly predicts the interface structure of the deformation twins.
Kampf et al. (page 973) describe the new mineral “Phoxite, (NH4)2Mg2(C2O4)(PO3OH)2(H2O)4, the first phosphate-oxalate mineral.” Phoxite is a new mineral species found in an unusual bat-guano-related, post-mining assemblage of phases in the Rowley mine, Maricopa County, Arizona, U.S.A. It is the first mineral known to contain both phosphate and oxalate groups and it possesses a novel layer structure that can be considered a “soft framework” due to strong hydrogen bonding between layers. The phase may have potential uses in agricultural applications for soil conditioning, fertilizing, and as a natural pesticide.
Keller and Ague (page 980) investigated “Corundum, and apatite lamellae from garnet.” Multiple lines of evidence support the precipitation of rutile, ilmenite, apatite, and corundum lamellae from garnet. Crystallographic orientation relationships (COR) between inclusion and host are consistent from independent occurrences worldwide, and minerals of the same crystal system share preferred relationships. Rutile forms a COR angularly equivalent to the Widmanstätten pattern in meteorites. These COR are valuable for comparing precipitation behavior across materials and suggest lamellae are precipitates indicative of precursor garnet chemistry.
Brugman and Till’s (page 996) “A low-aluminum clinopyroxene-liquid geothermometer for high-silica magmatic systems” presents a new clinopyroxene-liquid geothermometer calibrated for use with high-Fe, low-Al clinopyroxene from high-silica systems. It lowers calculated temperatures by 85 °C on average relative to a popular geothermometer (Putirka 2008, Eq. 33) and reduces the uncertainty by a factor of two (standard error of estimate ± 20 °C). When applied to natural systems, this clinopyroxene-liquid geothermometer reconciles many inconsistencies between experimental phase equilibria and preexisting geothermometry results for silicic volcanism, including those from the Bishop Tuff and Yellowstone caldera-forming and post-caldera rhyolites. Clinopyroxene is found not restricted to near-liquidus temperatures in rhyolitic systems and can be stable over a broad temperature range, often down to the solidus.
Berryman et al. (page 1005) in “Compressibility of synthetic Mg-Al tourmalines to 60 GPa” established the pressure-volume equation of state of tourmaline and its room-temperature metastability to 60 GPa in a series of high-pressure single-crystal X-ray diffraction experiments. The use of synthetic tourmalines representing five distinct end-member species revealed the primary role of the octahedral sites, particularly the Z site, on tourmaline’s compressibility and its remarkable metastability. This study highlights the utility of synthetic crystals in exploring compositional and structural controls on mineral properties at high pressure.
Hao et al. (page 1016) measured “The single-crystal elastic properties of the jadeite-diopside solid solution and their implications for the composition-dependent seismic properties of eclogite.” The 13 single-crystal adiabatic elastic moduli (Cij) of a C2/c jadeite sample close to the ideal composition (NaAlSi2O6) and a natural P2/n diopside-rich omphacite sample were measured at ambient conditions by Brillouin spectroscopy. Voigt-Reuss-Hill averaging of the Cij values yields an aggregate bulk modulus, KS, = 138(3) GPa and shear modulus, G, = 84(2) GPa for jadeite. The vpvs of omphacite decrease with diopside content, though the velocity changes are small as diopside component exceeds 70%. They also found that both the isotropic vpvs, as well as the seismic anisotropy of eclogite, changed strongly with the bulk-chemical composition. The relationship between the anisotropic velocities of eclogite and the chemical composition can be a useful tool to trace the origin of the eclogitic materials in the Earth’s mantle.
Fan et al.’s (page 1022) “Elasticity of single-crystal low water content hydrous pyrope at high-pressure and high-temperature conditions” reports measurements of the acoustic wave velocities and density of a single-crystal, hydrous pyrope with ~900 ppmw H2O by Brillouin light scattering combined with in situ synchrotron X-ray diffraction in the diamond anvil cell up to 18.6 GPa at room temperature and up to 700 K at ambient pressure. The modeling results indicate that hydrous pyrope remains almost elastically isotropic at relevant high P-T conditions and may have no significant contribution to seismic anisotropy in the upper mantle. Furthermore, hydrogen has no significant effect on the seismic velocities and the Vp/Vs ratio of pyrope at the upper mantle P-T conditions, especially for the limited hydration level (<100 ppmw H2O) of mantle-derived garnets.
Le Losq et al.’s study (page 1032) “Determination of the oxidation state of iron in Mid-Ocean Ridge basalt glasses by Raman spectroscopy” used conventional, as well as machine learning, data reduction methods to measure the iron oxidation state of Mid-Ocean Ridge Basalt (MORB) glasses, a key parameter for understanding upper mantle conditions and oceanic seafloor production, from their Raman spectra. The approaches allow evaluation of the average iron oxidation state in MORB glasses as 0.09 and to predict MORB glass chemical composition. Because Raman spectroscopy is fast, non-destructive, has microscale resolution and has the potential to be portable (e.g., the SHERLOC system that equips the Mars 2020 rover), its combination with machine learning approaches have a strong potential for analysis of materials in environments inaccessible by other conventional techniques, like Mid-Ocean ridges.
Pieczka et al.’s paper (page 1043) “Lepageite, Mn2+3(Fe3+7Fe2+4)O3[Sb3+5As3+8O34]], a new arsenite-antimonite mineral from the Szklary pegmatite, Lower Silesia, Poland” presents data on a new mineral, lepageite, that is a representative of a rare mineral group of arsenite-antimonites, discovered in the Szklary LCT pegmatite in Poland. We describe its chemical composition and crystal structure and finally explain by genetic implications why arsenite-antimonite minerals are rare species in a pegmatitic environment.
Igami et al. (page 1051) investigated the “High-temperature structural change and microtexture formation of sillimanite and its phase relation with mullite.” Synchrotron X-ray diffraction experiments and transmission electron microscopy of heated sillimanite at various pressures were conducted to clarify the detailed phase relations between sillimanite and mullite. As a result, they propose a new P-T diagram for the Al2SiO5 system with the mullitization boundary and the Al/Si order parameter of sillimanite. Investigations of sillimanite/mullite based on the present results can yield new information about thermal histories in high-temperature regions that is easy to be lost in general.
Hawthorne et al. (page 1062) in their “Memorial of Paul Brian Moore 1940–2019” remember one of the most prolific mineralogists of the 20th century and highlight his contributions to our science.
June 2019
This issue of American Mineralogist starts with an interesting and thoughtful Editorial by the former Editor Keith Putirka (page 785): Why scientists should study chess.
In a “highlights and breakthroughs” article, Ferrand (page 788) comments on the paper “Kinetics of antigorite dehydration: Rapid dehydration as a trigger for lower-plane seismicity in subduction zones” by Liu et al. (2019, Vol. 104, no. 2, pages 282-290). The study by Liu et al. confirms that antigorite dehydration is fast enough to trigger brittle failure under subduction conditions. The dehydration was found to involve two dehydroxylation mechanisms, allowing better understanding of the two-step antigorite destabilization observed in high-pressure experiments.
Dunn et al. (page 791) performed calcite-graphite carbon isotope thermometry on 150 marble samples from the western Central Metasedimentary Belt (CMB) of the Ontario segment of the Grenville Province, which represents the deeply eroded and exposed core of a major mountain chain that existed one billion years ago. The obtained data show a gradual increase in the peak metamorphic temperature from <500 °C in the Tudor Township area in the east to >700 °C along the western margin of the CMB. These results refine our understanding of orogenic buildup and collapse in the Grenville, including the styles of deformation of crystalline rocks in continental crust. The authors also found unusually high carbon isotope ratios (13C/12C) in the sample which provides a useful chemical marker to correlate rocks that formed in similar marine settings at around the same time roughly 1.3 billion years ago, well before the large mountain-building event.
In his Roebling Medal Paper, Hazen (page 810) describes a classification of planetary materials based on natural kind clustering. Minerals reveal the nature of the co-evolving geosphere and biosphere through billions of years of Earth history. Mineral classification systems have the potential to elucidate this rich evolutionary story; however, the present mineral taxonomy, based as it is on idealized major element chemistry and crystal structure, lacks a temporal aspect and thus cannot reflect planetary evolution. A complementary evolutionary system of mineralogy based on the quantitative recognition of "natural kind clustering" for a wide range of condensed planetary materials with different paragenetic origins (best revealed through the data-driven methods of cluster analysis) has the potential to amplify, though not supersede, the present classification system.
Putirka and Rarick (page 817) survey the compositions and mineralogy of >4000 nearby stars from the Hypatia Catalog (the most compositionally broad of such collections) to determine whether such exoplanets might be geologically similar to Earth. They find that most exoplanets will have rocky compositions that are similar to Earth and so most exoplanets might exhibit plate tectonics, and so might also be Earth-like in other respects, for example, by harboring life.
Stangarone et al. (page 830) used density functional theory (DFT) simulations of the structures of zircon and reidite (polymorphs of ZrSiO4) to show that above 20 GPa zircon undergoes a displacive phase transition to a new polymorph (space group I-42d) which may trigger the reconstructive transformation to reidite. Thus, this study provides new insights into the zircon-reidite transition, which may be induced by shock in meteorite impacts. The results clarify the discrepancies between previous observations on natural and experimental samples.
Papike et al. (page 838) investigated the effects of contrasting Ti and Al activities on Mn/Fe systematics in pyroxene from lunar mare basalts. In terms of using Mn/Fe ratios for determining planetary parentage and the reasons for dispersion in this trend for each body, variation in oxygen fugacity appears to be the most important factor for Martian basalts. For lunar mare basalts, however, high Ti activity in the melting zone and the melts, and crystallization sequence differences among high-Ti, low-Ti, and very low-Ti basalts account for almost all of the observed dispersion in the Mn/Fe ratios. This study gives important insight into the challenges of establishing planetary parentage by use of Mn/Fe ratios in lunar pyroxene, and explores the effects of crystallization sequence (order of appearance on liquidus) on this parameter.
Wood et al. (page 844) recalculated the temperatures at which the so-called "moderately volatile elements" such as Zn, In, Tl, Ga, Ag, Sb, Pb, and Cl, would condense from a gas of solar composition at 10-4 bar during formation of the solar system. The calculations highlighted three areas where currently available estimates of condensation temperature could be improved. The newly calculated 50% condensation temperatures are generally similar to or, because of the improvements, lower than those of Lodders (2003). Thus, this work provides a more accurate measure of the relative volatilities of the elements at the earliest stages of planetary formation.
Heller et al. (page 857) explore the possibilities of estimating radiation damage (-dose) in titanites using Raman spectroscopy. Raman spectra of randomly oriented titanite fragments with known thermal history were related to their -dose, calculated from the concentration of -emitting elements. The intensity-weighted mean width of all Raman bands of a spectrum is independent from orientation and proves to be the most robust measure of -dose. This approach provides a pre-selection method to optimize the range of -doses of titanite crystals to be dated by (U-Th)/He thermochronology.
Yoder et al. (page 869) have explored the IR spectra of carbonated calcium and strontium apatites that contain carbonate in the channel (A-type substitution) as well as in both the channel and in place of phosphate (B-type substitution). The results show that a correlation of the band position of the high frequency A-type carbonate band with weight percent carbonate exists for the calcium apatites, whereas a correlation of the band positions of both the low and high frequency B-type carbonate bands with carbonate weight percent occurs for the strontium apatites. On the other hand, correlations of band frequencies with sodium content are weaker than those for carbonate, even though carbonate and sodium are correlated with each other in the calcium apatites. This study also confirms previous conclusions about the distribution of A and B-type carbonate for most synthetic calcium apatites formed under a wide range of temperature and pressure conditions.
Yang et al. (page 878) carried out in situ high-temperature and high-pressure IR spectroscopic investigations on hydrogen storage sites in the natural olivine and synthetic Fe-free forsterite. The results show that hydrogen does not transfer between storage sites with increasing temperature, but displays disordering at temperatures over 600 °C. In contrast, pressure can induce re-configuration of hydrogen storage sites corresponding to the 3610 and 3579 cm–1 bands. Hydrogen storage sites also exhibit disordering at high pressure. In addition, the dehydrogenation experiments of the natural olivine indicate interactions of hydrogen storage sites. Protons released from titanium-clinohumite defects move to pure Si vacancies, and also to Mg vacancies coupling with trivalent cations. This study is of importance to understand water distribution and its impact on the upper mantle.
Glazner (page 890) conducted a thermodynamic analysis to reevaluate the ascent of water-rich magma and decompression heating. It has long been assumed that water-saturated magmas move into the subsolidus field and freeze upon ascent. However, this assumption ignores the considerable thermal energy released by crystallization. The new analysis shows that if magma ascent is treated as an adiabatic, reversible (isentropic) process then water-saturated magma can ascend without freezing, following the solidus to shallow depth and higher temperature as it undergoes modest crystallization and vapor exsolution. Decompression heating is an alternative to magma recharge for explaining pre-eruptive reheating seen in many volcanic systems and accounts for paradoxical growth of quartz during heating events. The viscosity increase that accompanies vapor exsolution as magma rises to shallow depth explains why silicic magmas tend to stop in the upper crust rather than erupting, producing the observed compositional dichotomy between plutonic and volcanic rocks.
Lazarz et al. (page 897) conducted in situ single-crystal X-ray diffraction experiments to investigate high-pressure phase transitions of clinoenstatite (Mg2Si2O6): 1) from a low-pressure form (LPCEN, space group P21/c) to a high-pressure form (HPCEN, space group C2/c) at ~6 GPa; and 2) from HPCEN to a P21/c-structured polymorph (HPCEN2) at ~45 GPa (discovered in this work). High-pressure structure refinements of HPCEN were carried out to determine its P-V equation of state and structural evolution over an expanded pressure range relevant to pyroxene metatstability. The newly discovered HPCEN2 phase is related to the P21/c structure previously observed in diopside at 50 GPa and in clinoferrosilite at ~30-36 GPa.
Matrosova et al. (page 905) performed in situ high-pressure single-crystal X-ray diffraction experiments to determine compressibility of two synthetic Na-rich clinopyroxenes, a Na-Ti-pyroxene with formula (Na0.86Mg0.14)(Mg0.57Ti0.43)Si2O6 and a Na-pyroxene with composition (Na0.886Mg0.085Fe0.029)(Si0.442Mg0.390Fe0.168)Si2O6, up to 40 GPa. These phases were found to be monoclinic with the space group C2/c and exhibit KTo of 106.8(2) and 121.8(4) GPa, respectively. Na-Ti-pyroxene is more compressible than Fe-bearing Na-Mg-Si-pyroxene, likely because the FeO6 octahedron is significantly more rigid than MgO6 at high pressure. The formation of Na-rich pyroxenes in the deep mantle is related to crystallization of low-degree alkaline carbonate-silicate melts formed when the crust and mantle interact during the slab descent and its stagnation in the transition zone.
This issue ends with a review by Hummer (page 914) on a textbook entitled “An Introduction to X-ray Physics, Optics, and Applications” by Carolyn MacDonald (2017), Cambridge University Press, 368 pp. The book presents a comprehensive, rigorous, but understandable explanation of X-ray physics and the many contexts in which this physics is useful in modern technologies. Hummer highly recommends this book to any student or researcher with an adequate background in physics who is seeking advanced knowledge of any system that utilizes X-rays.
May 2019
Hudson-Edwards (p. 633) discusses research on the importance of alunite, jarosite, and beudantite group minerals as sinks for arsenic and antimony. These minerals can immobilize these elements and restrict their bioavailability in acidic, oxidizing environments. This ability to store arsenic and antimony can protect humans and other biota from their toxic effects. Aerobic and abiotic As release from alunite and natroalunite is limited, especially between pH 5 and 8. Release of As is also very limited in As-bearing jarosite, natrojarosite, and ammoniumjarosite at pH 8 due to the formation of secondary maghemite, goethite, hematite, and Fe arsenates that resorb the liberated As. Abiotic reductive dissolution of As-bearing jarosite at pH 4, 5.5, and 7 is likewise restricted by the formation of secondary green rust sulfate, goethite, and lepidocrocite that take up the As. Similar processes have been observed for the aerobic dissolution of Pb-As-jarosite (beudantite analog), with secondary Fe oxyhydroxides resorbing the released As at pH 8. Higher amounts of As are released, however, during microbial-driven jarosite dissolution. Natural jarosite has been found to contain up to 5.9 wt% Sb5+ substituting for Fe3+ in the B-site of the mineral structure. Sb(V) is not released from jarosite at pH 4 during abiotic reductive dissolution, but at pH 5.5 and 7, up to 75% of the mobilized Sb can be structurally incorporated into secondary green rust sulfate, lepidocrocite, or goethite. Further research is needed on the co-incorporation of As, Sb, and other ions in, and the uptake and release of Sb from, alunite, jarosite, and beudantite group minerals, the influence of microbes on these processes, and the long-term (>1 yr) stability of these minerals.
Barton (p. 641) examines the social, cultural, scientific, and technological factors that affected the rate and types of the 4,046 mineral discovery reports (roughly 3/4 of all known minerals) from 1917 to the present. The number of new minerals discovered per year was steady over time from 1917 to the early 1950s, when it began a rapid increase punctuated by spikes in 1962-1969, 1978-1982, and 2008-2016, the last of which is probably still ongoing. A detailed breakdown of the technological, geographic, institutional, and other characteristics of mineral discovery demonstrates that the availability of instrumentation for a particular analytical technique has a far larger impact on the rate of its uptake in mineral discovery than the technique's invention or computer-automation. Around 2/3 of all new mineral discoveries are found in samples associated with resource exploration and exploitation, with peralkaline intrusions and volcanic fumaroles as the next most productive source of new mineral discoveries. New mineral discovery is highly concentrated in specific laboratories or work groups Interestingly, although the number of analytical techniques continues to grow, the average number of methods used to characterize new minerals has not changed significantly since 1960, and about half of new mineral descriptions are made using roughly the minimum of analyses required for a new mineral to be recognized. Although some minerals have been discredited or redefined, most of those were due to changes in nomenclature and classification, and only five cases of fraudulent mineral discovery are known. This article presents the data underlying these analyses and discusses some possible reasons for the observed trends in the rate of new mineral discovery, as well as the implications for the history (and future) of mineralogy.
Gu et al. (p. 652) investigated enigmatic milky diamonds and revealed that dislocations, nano-inclusions, and polycrystalline textures are the possible origins of their appearance. Through application of cathodoluminescence (CL), electron backscatter diffraction (EBSD), and transmission electron microscopy (TEM), Gu et al. suggest dislocation loops, nano-sized inclusions (negative crystals) and/or characteristic grain boundaries of the radiating fibrous crystals are the origins for the milky appearance of the type IaB diamonds studied.
Spürgin et al. (p. 659) present an unconventional zeolite deposit type and discuss the geological framework as an important factor for the generation of zeolite deposits hosted in subvolcanic alkaline rocks. This work is focused on the Miocene Kaiserstuhl Volcanic Complex, Germany, which shows that economic grades of zeolitization occur in phonolites intruded into water-rich, shallow marine sediments; whereas low degrees of zeolitization are found in phonolite intruded in dry, subaerial pyroclastic strata of the volcanic edifice. Pseudomorphic replacement textures indicate that zeolites formed from magmatic feldspathoid minerals. The common sequence from Ca-Na-rich zeolites (thomsonite, mesolite, gonnardite) towards pure Na-zeolites (natrolite, analcime) is the result of closed-system alteration. The late formation of Ca-K-rich chabazite in the latter phonolite is interpreted as open-system behavior with an influx of water in equilibrium with the leucite-bearing country rock, after the main alteration phase. This work concludes that zeolite deposits may not only be found in sheet-like, often very young tuffs but also in older rocks in a different geological context.
Dong et al. (p. 671) measured the melting point of barium carbonate (BaCO3) at pressures up to 11 GPa using the ionic conductivity and platinum (Pt) sphere methods in a multi-anvil press. The melting point decreases with pressure from 2149 ± 50 K at 3 GPa to a minimum of 1849 K at 5.5 GPa, and then it increases with pressure to 2453 ± 50 K at 11 GPa. The negative slope of the BaCO3 melting curve between 3 and 5.5 GPa indicates that the liquid is denser than the solid within this pressure range. Synchrotron X-ray diffraction measurements in a laser-heated diamond-anvil cell showed that crystalline BaCO3 transformed from the aragonite structure (Pmcn) to the post-aragonite structure (Pmmn) at 6.3 GPa and 1026 K as well as 8 GPa and 1100 K, and the post-aragonite structure remained metastable upon quenching and only reverted back to the witherite structure upon pressure release. The local minimum near 5 GPa is attributed to the triple point where the melting curve of BaCO3 meets a phase transition to the denser post-aragonite structure (Pmmn). Local minima in the melting curves of alkaline earth carbonates would lead to incipient melting of carbonated rocks in Earth's mantle.
Williams et al. (p. 679) demonstrate that packing changes in glasses have little influence on how glasses compact under pressure through their study of the elasticity of a sequence of SiO2-TiO2 glasses at high pressures and temperatures. The effect of changing Ti content on the bulk moduli of these glasses is monotonic, and no systematic effect of possible coordination changes is observed. In contrast, there is an apparent decrease in the pressure derivative of the bulk modulus above ~3 wt% TiO2. This change occurs at a similar composition to that at which a transition from predominantly 5-fold to 4-fold of Ti has been proposed to occur in these glasses. This shift in the pressure derivative of the bulk modulus is attributed to a stiffening of the equation of state for these glasses generated by the substitution of 5-fold Ti species relative to TiO4 units. These results provide rationales for the onset of coordination changes producing a minimal change in the equation of state of silicate melts/glasses, and for bulk moduli determined at ambient pressure producing relatively accurate silicate melt volumes even within liquids that have begun to undergo coordination changes. Thus, this study supports the general validity of the single equation of state formulations that describe the densities of silicate melts through the transition zone and shallow upper mantle.
Wu et al. (p. 686) explored the thermodynamics of interaction between nano oxides and small gas molecules by gas adsorption calorimetry to investigate the energetics of ethanol and carbon dioxide adsorbed on surfaces of nanoscale anatase, rutile, and γ-alumina particles. The measured zero-coverage adsorption enthalpies per mole of gas adsorbed are -97.7, -107.3, and -84.4 kJ/mol for C2H5OH on anatase, rutile, and γ-Al2O3, respectively. The corresponding values for CO2 adsorption are -61.6, -47.4 and -47.1 kJ/mol. The results indicate the ethanol adsorption is generally more exothermic than carbon dioxide and water adsorption. The isotherm and differential enthalpies show type II isotherms and step-wise patterns for ethanol adsorption in all three oxides. However, CO2 adsorption shows simple continuous isotherms and energetics that suggest dominantly physical adsorption occurred. The repeated adsorption cycle shows that ethanol adsorption on these nanoparticles is partially reversible at room temperature. This thermodynamic evidence indicates that ethanol and similar organics may protect mineral oxide surfaces from reaction with aqueous solutions, which may affect crystal growth, dissolution, and biomineralization.
Knafeic et al. (p. 694) investigated the effect of olivine oxidation on its magnetic properties using a time series of 1 bar oxidation experiments at 600 °C and 900 °C. They found rapid olivine oxidation and alteration at both 600 and 900 °C, forming magnetite and hematite associated with a change from paramagnetic to ferromagnetic behavior after oxidation. Magnetite and hematite nucleated along dislocations and impurities in the crystal structure, along with surface coatings and within cracks in the crystals. Fresh, unaltered mantle xenoliths containing magnetite have been interpreted as having formed in cold tectonic regimes in the mantle, rather than through oxidation during or after the ascent. Mantle xenoliths rapidly ascend through the mantle with estimates of ascent of up to 90 km/hour (3 GPa/hour) based on the diffusion profile of water in mantle olivine. The rates correspond to xenoliths ascending through the mantle over hours and not days or weeks. Our results show that olivine oxidation and alteration can occur in days to weeks at 600°C and within minutes at 900 °C. Therefore, if the xenolithic material is transported to the surface in a cold magma (at temperatures less than or equal to 600 °C), then the time scale of ascent is likely not long enough for oxidation to cause magnetite formation or a ferromagnetic signature to occur. However, if the material is transported in a hot oxidized basaltic magma (with temperatures greater than or equal to 900 °C), then oxidation can cause magnetite formation and a ferromagnetic signature.
Mansor et al. (p. 703) investigated metal sulfide nanoparticles (NPs) because they are present in the environment and are important in controlling the availability of bio-essential and toxic metals in environmental remediation and in resource recovery. Characterizing the basic attributes of these NPs is the first step in understanding their behaviors in various processes. Experiments were performed in the presence and absence of the sulfate-reducer Desulfovibrio vulgaris to elucidate biological controls on NP formation. First, the single-metal end-member NPs were determined by precipitation in a solution containing either aqueous Fe(II) or Cu(II). Limited differences are observed between biogenic and abiogenic precipitates aged for up to one month; the Fe-only experiments resulted in 4-10 nm mackinawite (FeS) NPs that aggregate to form nanosheets up to ~1,000 nm in size, while the Cu-only experiments resulted in mixtures of covellite (CuS) NPs comprised of < 10 nm fine nanocrystals, 20-40 x 6-9 nm nanorods and ~ 30 nm nanoplates. The crystal sizes of biogenic mackinawite and covellite are respectively larger and smaller than their abiogenic counterparts, indicating a mineral-specific response to biological presence. Structural defects are observable in the fine nanocrystals and nanorods of covellite in both biogenic and abiogenic experiments, indicative of intrinsic NP instability and a formation mechanism via particle attachment. In contrast, covellite nanoplates are defect-free, indicating high stability and potentially rapid recrystallization following particle attachment. Mixed-metal sulfide NPs were precipitated at variable initial aqueous Fe-to-Cu ratios (2:1, 1:1 and 1:5). With increasing ratios of Fe-to-Cu, Fe-rich covellite, nukundamite (Cu5.5FeS6.5), chalcopyrite (CuFeS2), and Cu-rich mackinawite are formed. The Fe-rich covellite NPs are larger (100-200 nm) than covellite precipitated in the absence of Fe, indicating a role for Fe in promoting crystal growth. Chalcopyrite and nukundamite are formed through incorporation of Fe into precursor covellite NPs while retaining the original crystal morphology, as confirmed by doping a covellite suspension with aqueous Fe(II), resulting in the formation of chalcopyrite and nukundamite within days. Additionally, in the biological systems we observe the recrystallization of mackinawite to greigite (Fe3S4) after six months of incubation in the absence of Cu, and the selective formation of chalcopyrite and nukundamite at lower initial Fe-to-Cu ratios compared to abiotic systems. These observations are consistent with NP precipitation that ise influenced by the distinct (sub)micro-environments around bacterial cells. Comparative TEM analyses indicate that the synthetic NPs are morphologically similar to NPs identified in natural environments, opening ways to studying behaviors of natural NPs using experimental approaches.
Tateno et al. (p. 718) determined the pressure-volume-temperature (P-V-T) relationship of the B2 (CsCl-type) phase of KCl at 9-61 GPa at 1500-2600 K and up to 229 GPa at room temperature using synchrotron X-ray diffraction measurements in a laser-heated diamond-anvil cell (DAC). The nonhydrostatic stress conditions inside the sample chamber were critically evaluated based on the platinum pressure marker. With thermal annealing by laser after each pressure increment, the deviatoric stress was reduced to less than 1% of the sample pressure even at the multi-Mbar pressure range. The obtained P-V-T data were fitted to the Vinet equation of state with the Mie-Grüneisen-Debye model for thermal pressure. The thermal pressure of KCl was found to be as small as ~10 GPa even at 3000 K at any given volume, which is only half of that of common pressure markers (i.e. Pt, Au, or MgO). Such a low thermal pressure validates the use of a KCl pressure medium as a pressure marker at high temperatures.
Wang et al. (p. 724) studied the petrology, mineralogy, and geochemistry of lawsonite blueschists from the Tavşanli zone in NW Turkey - one of the best-preserved blueschist terranes in the world. The blueschist samples contain lawsonite + sodic amphibole + phengite + chlorite + titanite + apatite ± aragonite ± quartz ± relict igneous pyroxene ± Mn-rich garnet and opaque phases. Lawsonite is a significant repository for Sr, Pb, Th, U, and REE, whereas phengite carries the most LILE; titanite hosts the highest Nb and Ta as well as considerable amounts of HFSE, and apatite strongly controls Sr. Two groups of blueschists with different origins were identified: one from enriched continent-derived terrigenous sediments and another from MORB-like submarine basalts. Lawsonite in blueschists with the enriched origin exhibits strong Th/La fractionation, raising the possibility of the involvement of blueschist facies mélange to explain the origin of Mediterranean potassium-rich magmatism, because similarly high Th/La ratios are also observed in the Mediterranean potassium-rich lavas. We propose that subduction-induced tectonic imbrication took place entirely at shallow depths (less than 80 km), giving rise to newly formed lithosphere where oceanic and continental crustal materials, sediments, strongly depleted peridotite blocks and metamorphic rocks are all imbricated together, and in which many of the compositional characteristics of the lawsonite blueschist are sequestered. Subsequent melting of the fertile and enriched components in this new lithosphere would result in the generation of potassium-rich post-collisional mafic magmas with diagnostic geochemical affinities.
Dey et al. (p. 744) studied a calc-silicate rock from part of the Chotanagpur Granite Gneiss Complex (CGGC), East India, that contains veins and patches of vesuvianite (F: 2.3-3.9 apfu, Fe3+: 1.7-2.1 apfu) and garnet (Gr71-80Alm12-17Adr1-9) proximal to amphibole-bearing quartzo-feldspathic pegmatitic veins. The vesuvianite-garnet veins are both parallel to, and cross-cutting, the gneissic banding of the host calc-silicate rock. Two contrasting mineralogical domains that are rich in garnet and vesuvianite respectively develop within the vesuvianite-garnet veins. Textural studies support the view that the garnet- and vesuvianite-rich domains preferentially develop in the clinopyroxene- and plagioclase-rich layers of the host calc-silicate rocks respectively. Some of the vesuvianite-rich domains of the veins develop the assemblage vesuvianite + quartz + calcite +anorthite (as a result of the reaction diopside + quartz + calcite + anorthite = vesuvianite) which was deemed metastable in the commonly used qualitative isobaric T-XCO2 topology in the system CaO-MgO-Al2O3-SiO2-H2O-CO2 (CMASV). Using an internally consistent thermodynamic database, quantitative petrogenetic grids in the P-T and isobaric T-XCO2 spaces were computed in the CMASV system. The influence of the non-CMASV components (e.g., Na, Fe3+, F) on the CMASV topologies are discussed using the published a-X relations of the minerals. This study shows topological inversion in the isobaric T-XCO2 space, which is primarily dependent upon the composition of the vesuvianite. The quantitative CMASV topologies presented in this study successfully explain the stabilities of the natural vesuvianite-bearing assemblages, including the paradoxical assemblage vesuvianite + quartz + calcite + anorthite. Application of the activity-corrected CMASV topology suggests that infiltration of F-bearing oxidizing aqueous fluids into the calc-silicate rocks developed the vesuvianite-garnet veins in the studied area. A genetic link between quartzo-feldspathic pegmatites and the vesuvianite-garnet veins seems plausible. This study demonstrates controls of topological inversion in the complex natural system, owing to which, certain mineral assemblages that are deemed metastable in one set of reaction geometries can develop in nature.
Rezvukhin et al. (p. 761) recognized LILE-enriched chromium titanates of the magnetoplumbite (AM12O19) and crichtonite (ABC18T2O38) groups as abundant inclusions in orthopyroxene grains in a mantle-derived xenolith from the Udachnaya-East kimberlite pipe, Daldyn field, Siberian craton. The studied xenolith consists of three parts: an orthopyroxenite, a garnet clinopyroxenite, and a garnet-orthopyroxene intermediate domain between the two. Within the host enstatite (Mg# 92.6) in the orthopyroxenitic part of the sample, titanate inclusions are associated with Cr-spinel, diopside, rutile, Mg-Cr-ilmenite, and pentlandite. Crichtonite-group minerals also occur as lamellae inclusions in pyrope grains of the intermediate domain adjacent to the orthopyroxenite, as well as in interstitial-to-enstatite oxide intergrowths together with Cr-spinel, rutile, and ilmenite. Yimengite-hawthorneite inclusions in enstatite contain (wt%) 3.72-8.04 BaO, 2.05-3.43 K2O, and 0.06-0.48 CaO. Their composition is transitional between yimengite and hawthorneite end-members with most grains exhibiting K-dominant chemistry. A distinct feature of the studied yimengite-hawthorneite minerals is a high content of Al2O3 (5.74-7.69 wt%). Crichtonite-group minerals vary in composition depending on the occurrence in the xenolith: inclusions in enstatite are moderate-to-high in TiO2 (62.9-67.1 wt%), moderately Cr-rich (12.6-14.0 wt% Cr2O3), Ba- or K-specific in the A site, and contain low ZrO2 (0.05-1.72 wt%), whereas inclusions in pyrope are moderate in TiO2 (61.7-63.3 wt% TiO2), relatively low in Cr (8.98-9.62 wt% Cr2O3), K-dominant in the A site, and are Zr-enriched (4.64-4.71 wt% ZrO2). Crichtonite-group minerals in polymineralic oxide intergrowths show highly diverse compositions even within individual aggregates, where they are chemically dominated by Ba, Ca, and Sr. P-T estimates indicate the orthopyroxenite equilibrated at ~800 °C and 35 kbar. Preferentially oriented lamellae of enstatite-hosted Cr-spinel and diopside, as well as pyrope, diopside, and Cr-spinel grains developed around enstatite crystals, are interpreted to have been exsolved from the high-T, Ca-Al-Cr-enriched orthopyroxene precursor. The observed textural relationships between inclusions in enstatite and exotic titanate compositions imply that the studied orthopyroxenite has undergone metasomatic processing by a mobile percolating agent; this highly evolved melt/fluid was enriched in Ba, K, HFSE, and other incompatible elements. The prominent textural and chemical inhomogeneity of the interstitial oxide intergrowths is either a consequence of the metasomatic oxide crystallization shortly prior the kimberlite magma eruption, or arose from the intensive modification of pre-existing oxide clusters by the kimberlite melt during the Udachnaya emplacement. Our new data provide implications for the metasomatic treatment of orthopyroxenites in the subcontinental lithospheric mantle from the view of exotic titanate occurrences.
Bindi et al. (p. 775) report the first natural occurrence and single-crystal X-ray diffraction study of the Fe-analogue of wadsleyite [a = 5.7485(4), b = 11.5761(9), c = 8.3630(7) Å;, V = 556.52(7) Å3; space group Imma], spinelloid-structured Fe2SiO4, a missing phase among the predicted high-pressure polymorphs of ferroan olivine, with the composition (Fe2+1.10Mg0.80Cr3+0.04Mn2+0.02Ca0.02Al0.02Na0.01)Σ2.01(Si0.97Al0.03)Σ1.00O4. The new mineral was approved by the International Mineralogical Association (No. 2018-102) and named asimowite in honor of Paul D. Asimow, the Eleanor and John R. McMillan Professor of Geology and Geochemistry at the California Institute of Technology. It was discovered in rare shock-melted silicate droplets embedded in Fe,Ni-metal in both the Suizhou L6 chondrite and the Quebrada Chimborazo (QC) 001 CB3.0 chondrite. Asimowite is rare, but the shock-melted silicate droplets are very frequent in both meteorites and most of them contain Fe-rich wadsleyite (Fa30-45). Although the existence of such Fe-rich wadsleyite in shock veins may be due to the kinetic reasons, new theoretical and experimental studies of the stability of (Fe,Mg)2SiO4 at high temperature (> 1800 K) and pressure are clearly needed. This may also have a significant impact on the temperature and chemical estimates of the mantle's transition zone in Earth.
Belakovskiy et al. (p. 779) present names for 11 new minerals, including ammoniovoltaite, belousovite, chlorellestadite, clino-suenoite, marcobaldiite, markeyite, martinandresite, parisite-(La), plumbopharmacosiderite, somersetite, and ziminaite.
April 2019
This issue of American Mineralogist starts with an article to introduce the special collection “Earth in Five Reactions: A Deep Carbon Perspective” by Li et al. (page 465). This special collection features review papers on the role of carbon-related reactions in Earth’s dynamics and evolution, and includes original studies on carbon-bearing phases and the impact of chemical and polymorphic reactions on Earth’s deep carbon cycle.
Hazen (page 468) then describes the five reactions that influence the Earth’s history, which were identified at the Earth in Five Reactions Workshop held at the Carnegie Institution for Science, Washington, D.C., March 22–23, 2018. The workshop posed two challenges: (1) the formulation of a conceptual definition of “reaction” and (2) the identification and ranking of the “most important reactions” in the context of planetary evolution.
Knipping et al. (page 471) determined grain-to-grain and intra-grain Fe isotope variations in magnetite grains from the Los Colorados Kiruna-type iron oxide-apatite (IOA) deposit, Chile, using in situ femtosecond laser ablation MC-ICP-MS. The results reveal an igneous and magmatic-hydrothermal growth mechanism of magnetite, which is consistent with the formation model of this IOA deposit proposed based on trace element zonation in its magnetite grains. This is a contribution to the special collection “From Magmas to Ore Deposits”.
Wu et al. (page 485) report the discovery of ophiolite-hosted diamonds in the podiform chromitites of the Skenderbeu massif of the Mirdita ophiolite in the western part of Neo-Tethys. Carbon and nitrogen isotopes and mineral inclusions in diamonds demonstrate recycling of oceanic crust into the mantle. This discovery not only provides new evidence of diamonds in these settings but also sheds light on deep cycling of subducted oceanic crust and mantle composition.
Pe-Piper et al. (page 501) investigated the geochronology and trace element mobility in rutile from a Carboniferous syenite pegmatite using SEM, Raman spectroscopy, LA-ICP-MS and in situ U-Pb analysis. In particular, they determined the role of halogens in the mobility of Ti and associated Zr, U and REE, which led to hydrothermal alteration of magmatic rutile in syenite. The complexities of rutile chemistry in this hydrothermal setting could be reproduced in deeper subduction settings as a result of variations in halogen content of fluids released by prograde metamorphism.
Jenkins (page 514) synthesized calcium amphiboles from ferro-pargasite and hastingsite bulk compositions at 600–950 °C, 0.1–0.45 GPa and logfH2 of 1.4 to 2.4 for durations of 111–672 h, and determined how variations in formation conditions (temperature, pressure, hydrogen fugacity), bulk composition (Na and K ratio), and choice of starting material salts affect the Cl contents of synthesized calcium amphiboles. The results imply that the crystal-chemical controls for Cl incorporation in calcium amphiboles are dominated by substitution of Fe2+ for Mg, TAl for Si, and K for Na into the crystallographic A site with a linear dependence at the rate of 0.45 Cl per FeAlK index above a minimum value of about 0.34.
Zhang et al. (page 525) performed three series of amphibole crystallization experiments from hydrous basaltic melt at 0.6–2.6 GPa and 860–970 °C and measured the amphibole crystal size distribution (CSD). The results show that the amphibole growth rate increases with increasing temperature in the isobaric series and with increasing pressure at constant temperature. By contrast, the growth rate is negatively correlated with crystallization time at constant temperature and pressure. The authors developed a functional form for evaluating growth rate at known pressure and temperature from an observed amphibole CSD and applied to a diorite from the eastern Tianshan Mountains, NW China.
Yuguchi et al. (page 536) investigated the role of micropores, mass transfer, and reaction rate in the hydrothermal alteration process of plagioclase from the Toki granitic pluton in central Japan. Important observations include: 1) Micropores form during the incipient stage of plagioclase alteration by dissolution of the anorthite component, and then contribute to the infiltration of hydrothermal fluid into the plagioclase. 2) The mass transfer of the components released from biotite by chloritization involves the inflow of H4SiO4, Al3+, Fe2+, Mn2+, Mg2+, K+, CO2, and F-, and the outflow of H2O, H+, and Ca2+. 3) The infiltration rate of the hydrothermal fluid and the potassium transfer rate through the micropores into the plagioclase represent the mass transfer rate of the alteration.
Beyer et al. (page 557) measured the diffusivity of Pb in CaTiO3 perovskite (commonly used for dating kimberlites and carbonatites) using Rutherford backscattering and TOF-SIMS in the depth-profiling mode. Experiments were performed on oriented CaTiO3 single crystals with (Ca0.83Pb0.07)Ti1.05O3 thin film or (Ca0.9Pb0.1)TiO3 powder as the Pb-source, which were annealed at 736—1135 °C for 2—283 h. The measured Pb profiles show two regions — a steep gradient at the diffusion interface that transitions sharply to a low concentration tail that penetrates deeper into the crystal. Moreover, Pb is trapped in the planar defects formed due to the CaTiO3/Pb-bearing perovskite lattice mismatch, and the closure temperature for Pb in CaTiO3 is found to be between 300 and 400 °C for a range of different cooling scenarios if diffusive resetting of Pb in CaTiO3 occurs. At typical cooling rates of hours to days for ascending kimberlite, the age of crystal growth is preserved, with closure temperatures similar to the magma temperature.
Jonckheere et al. (page 569) compare the traditional bulk etch rate (vB) and an alternative radial etch rate (vR) model for fission-track etching in apatite. A skeletal vR-model, based on the inferred orientations of the vR minima and maxima, accounts for the main geometrical features of etched fission tracks, unifies the diverse appearances of etched tracks, and embeds fission-track etching in the mainstream theories of crystal growth and dissolution. The authors suggest that the anisotropic-vB-model may be replaced with an anisotropic-vR-model based on the radial etch rate.
Zhang et al. (page 580) investigated the compressional behavior of a synthetic liebenbergite, Ni2SiO4, that has the olivine structure, up to 42.6 GPa using single-crystal synchrotron X-ray diffraction. Over this pressure range, liebenbergite retains the orthorhombic Pbnm structure. Fitting the pressure-dependent variation in its unit-cell volume to a third-order Birch-Murnaghan equation of state yielded a bulk modulus of 163(3) GPa — the most incompressible olivine-structured silicate.
Tsujino et al. (page 588) determined the wadsleyite-ringwoodite phase transition loop in the Mg2SiO4-Fe2SiO4 system under dry conditions from 1473–1873 K using in situ high P-T synchrotron X-ray diffraction. Assuming an equilibrium composition of wadsleyite and ringwoodite coexisting with garnet in a pyrolite model and an adiabatic temperature gradient with a potential temperature of 1550–1650 K, the phase transition depth and effective width of the seismic discontinuity were found to be 500–514 and 20–22 km, respectively. Considering wet and oxidized conditions, the depth of the wadsleyite-ringwoodite phase boundary could be >520 km. Variation in the depth of seismic anomaly may be attributed to water content or oxygen fugacity of the transition zone.
Stachowicz et al. (page 595) investigated the cation ordering, valence states, and symmetry breaking in chevkinite-(Ce) from the Biraya rare-metal deposit, Russia, using single-crystal X-ray diffraction and X-ray photoelectron spectroscopy (XPS). Nb-rich chevkinite-(Ce) typically possesses a space group of C2/m, though a specimen of lower, P21/α, symmetry has also been recognized. While XPS shows that both C2/m and P21/α structures contain Ti4+ and Ti3+, it also indicates that Ti2+ may occur in the P21/α phases. In addition to the substitution CFe3+ + DTi4+ ↔ CFe2+ + DNb5+, the authors propose that another substitution, 2DTi4+ ↔ DNb5+ + DTi3+, can occur, leading to substantial Nb-enrichment.
Kampf et al. (page 603) describe the crystal structure, chemical composition and physical properties of meyrowitzite, Ca(UO2)(CO3)2·5H2O, a new mineral from the Markey mine, Red Canyon, San Juan County, Utah, U.S.A. The structure is monoclinic, P21/n, and contains a novel uranyl-carbonate sheet. Meyrowitzite is a secondary phase found on calcite-veined asphaltum in association with gypsum, markeyite and rozenite.
Bindi et al. (page 611) describe the discovery of the first natural metal hydride, gamma-VH2. .It was discovered in xenoliths within volcanic rock on Mount Carmel, Israel. The hydride coexists with metallic V, which requires oxygen fugacities of ΔIW -9 or lower. The presence of VH2 is interpreted as a signature of deep-seated basaltic magmas with mantle-derived CH4+H2 at high fluid/melt ratios.
Lindsley et al. (page 615) describe the synthesis of pigeonite samples and offer them for non-destructive study. As pigeonite does not survive as a discrete phase in coarse plutonic rocks, natural samples suitable for study are difficult to come by. Eight samples of differing composition, the results from more than 125 trials, in sizes 5-50 micrometers are available for bulk analysis.
This issue contains four 2018 MSA award presentations: (1) page 619–620: “Presentation of the 2018 Roebling Medal of the Mineralogical Society of America to E. Bruce Watson” by Frank Richter; (2) page 621–622: “Acceptance of the 2018 Roebling Medal of the Mineralogical Society of America” by E. Bruce Watson; (3) page 623: “Presentation of the Dana Medal of the Mineralogical Society of America for 2018 to Jörg Hermann” by Bradley R. Hacker; and (4) page 624: “Acceptance of the Dana Medal of the Mineralogical Society of America for 2018” by Jörg Hermann.
Belakovskiy and Cámara (page 625) introduce eight new minerals, including fengchengite, ferriperbøeite-(Ce), genplesite, heyerdahlite, millsite, saranchinaite, siudaite and vymazalováite, and summarize new data on lavinskyite-1M.
Gysi (page 630 ) reviews the book: “Thermodynamics of Natural Systems: Theory and Applications in Geochemistry and Environmental Science”, 3rd ed. (2017) by Greg Anderson, Cambridge University Press, 428 p. Compared to the two earlier editions, this edition is shorter and more concise and is suitable as an introduction to thermodynamics book.
March 2019
Rodeghero et al. (p. 317) study the ability of zeolite ZSM-12 to remove the groundwater contaminants chloroacetanilides and their degradation products. They measured the removal of 2-ethyl-6-methyl-aniline [C2H5C6H3(CH3)NH2, labeled EMA] from water by combining chromatographic, thermogravimetric, and synchrotron X-ray powder diffractometric techniques and demonstrate that ZSM-12 can rapidly incorporate about 4 EMA molecules per unit cell. The authors document the strong interaction with framework O atoms in ZSM-12 that confers stability to the pollutants in the zeolite cages. The rapid kinetics combined with the good adsorption capacity makes ZSM-12 a promising material to control and minimize water pollution from acetanilide compounds as well as other agro-chemical contaminants.
Zhu et al. (p. 325) report chemical and structural analyses of synthetic Fe7C3, a potential host of reduced carbon in Earth’s mantle and a candidate component of the inner core. They synthesized Fe7C3 utilizing a diffusive reaction between iron and graphite that contained 31 to 35 at% carbon. They found that more carbon-rich Fe7C3 has smaller unit-cell volumes, suggesting that excess carbon atoms substituted for iron atoms instead of entering the interstitial sites of the closed-packed iron lattice as in FeCx steel. This substitution leads to a larger reduction in the unit-cell mass than the volume so that the carbon-rich end-member may be as much as 5% less dense than stoichiometric Fe7C3. If Fe7C3 solidifies from Earth’s iron-rich liquid core, it is expected to have a nearly stoichiometric composition with a compositional expansion coefficient of ~1.0. However, laboratory experiments using carbon-rich synthetic Fe7C3 to model the inner core may overestimate the amount of carbon that is needed to account for the core density deficit.
Hunt and Lamb (p. 333) applied mineral equilibria to estimate values of aH2O in rocks that originated below the Moho. The chemical compositions of olivine + orthopyroxene + clinopyroxene + amphibole + spinel ± garnet were used to estimate values of temperature (T), pressure (P), aH2O, hydrogen fugacity (fH2), and oxygen fugacity (fO2) in 11 amphibole-bearing mantle xenoliths from the southwestern U.S.A. The activity of water was first calculated by amphibole dehydration equilibria, and then oxygen fugacity calculated from coexisting olivine, spinel, and orthopyroxene was combined with hydrogen fugacities calculated from amphibole dehydrogenation equilibria to construct a separate estimate of the water activity. The two separate estimates of aH2Ogenerally agree to within 0.05. This agreement indicates that the amphibole in these samples has experienced little or no retrograde H-loss and that amphibole equilibria yields robust estimates of aH2Othat, in these xenoliths, are generally <0.3, and are often 0.1 or less.
Macdonald et al. (p. 348) review chevkinite-group minerals (CGM), minerals that are dominantly monoclinic REE-Ti-Fe sorosilicates [(REE, Ca)4Fe2+(Fe2+,Fe3+,Ti)2Ti2(Si2O7)2O8)], with REE2O3 contents up to ~50 wt%, although some members with predominant Mg, Al, Mn, Cr, Sr, or Zr in one of the cation sites are also known. They show that these minerals can be found in igneous and metamorphic rocks on Earth as well as on the Moon and Mars; these minerals may form over the pressure range 50 to <10 kbar, and over a wide temperature range. In common with other REE-bearing accessory minerals, CGM are prone to alteration by hydrothermal fluids. The nature and extent of the alteration are primarily determined by the composition of the fluids. Fluids poor in ligands tend to generate a Ti-enriched phase whose nature is unknown but is probably amorphous. With increasing F + CO2 levels, complex replacement assemblages are formed. This review also discusses some of our ignorance. The stability of CGM vis-à-vis other REE-Ti-bearing accessories is poorly understood. They are often the major carriers of REE and actinides, and they have a high potential for fractionating the light lanthanides and Th from U, but very little systematic work has been done in determining CGM-melt partition coefficients, yet such data are critical in, inter alia, geochemical modeling. And although observational evidence of the effects of alteration and element mobility is accumulating and chemical equations can be constructed to approximate the reactions, there is still no firm geochemical basis for understanding element redistribution during these processes.
Treiman et al. (p. 370) investigate the origin of magnesium aluminate spinel, (Mg,Fe)Al2O4, in lunar anorthositic rocks Although uncommon, recent near-infrared spectra of the Moon have delineated regions where spinel is the only ferromagnesian mineral, and the rock is inferred to be spinel anorthosite. The authors consider multiple alternative hypotheses for the origin of spinel anorthosites: formation at high pressure, low-pressure assimilation of anorthosite by picritic magmas, and crystallization of superliquidus anorthite-rich melts created by impacts. The authors conclude that near the lunar surface, the most likely process of spinel formation is rapid crystallization of impact melts of anorthosite + picrite or peridotite compositions. The presence of spinel anorthosite on the walls and central peaks of impact craters results from rapid cooling and partial crystallization of superliquidus melts produced in the impacts, and not from the uplift of deep material to the Moon's surface.
Kuroda et al. (p. 385) investigated the diffusion of deuterated water into silica glass at 900–750 °C and a water vapor pressure of 50 bar and found it to be an order of magnitude greater than previously measured. Their analysis indicates that the species responsible for the fast diffusion is not molecular hydrogen, but molecular water, and hypothesize that water diffuses through the free volume of the glass in a manner similar to noble gases. The abundance of free volume in the silica glass structure estimated previously is higher than that of 2H observed in the fast diffusion of this study, suggesting that the free volume was not fully occupied by 2H2O under the present experimental conditions. This implies that the contribution of the fast water diffusion to the total water transport in volcanic glass becomes larger at higher water vapor pressure conditions.
Cao et al. (p. 391) characterized textural and compositional microscale (10–100 μm) and nanoscale (10–100 nm) zoning in a plagioclase phenocryst from a fresh, syn-mineralization diorite porphyry (Black Mountain porphyry Cu-Au deposit, Philippines) by electron microprobe, laser ablation-inductively coupled plasma-mass spectrometry, and atom probe tomography. The complex plagioclase crystal (3.0 × 5.4 mm) has a patchy andesine core (An41–48 mol%), eroded bytownite mantle (An71-80 mol%), and oscillatory andesine rim (An39–51 mol%). Microscale variations with a periodic width of 50 to 200 μm were noted for most major and trace elements (Si, Ca, Al, Na, K, Fe, Mg, Ti, Sr, Ba, Pb, La, Ce, and Pr) with a ΔAn amplitude of 4–12 mol% in both the core and rim. The mantle has a distinct elemental composition, indicating the addition of hotter mafic magma to the andesitic magma. Atom probe tomography shows an absence of nanoscale variations in the andesine rim but alternating nanoscale (25–30 nm) Al-rich, Ca-rich, and Si-rich, Na-rich zones with a Ca/(Ca+Na)at% amplitude of ~10 in the bytownite mantle. The estimated physiochemical parameters for crystallization suggest that microscale oscillatory zoning was likely controlled by internal crystal growth mechanisms, not by periodic variations in physiochemical conditions. The micro-scale zoning in plagioclase indicates a minimum cooling rate of 0.0005 °C/yr during crystallization, but the retention of nanoscale zoning (~28 nm) requires a minimum cooling rate of 0.26 °C/yr. Given that this is significantly faster cooling than would occur in a magma chamber, this texture likely records the post-crystallization emplacement history.
Masci et al. (p. 403) investigated Fe3+ in chlorite using the electron microprobe and XANES to assess the importance of oxychlorite and how ferric iron influenced cation site distributions and thermobarymetric calculations. Their analyses show iron oxidation states varying from ferrous to ferric; iron is in octahedral coordination in all ferromagnesian chlorites but to ~25% tetrahedral in the lithian chlorite cookeite (1.0 wt% Fe2O3(total)). Absolute amounts of ferric iron cover an unprecedented range (0 to ~30 wt% Fe2O3). For highly magnesian, ferric chlorite, Fe concentrations are low and can be accounted for by Al = Fe3+ substitution. In Fe-rich samples, Fe3+ may exceed 2 atoms per formula unit (pfu, 18 oxygen basis). When structural formulas are normalized to 28 charges corresponding to the standard O10(OH)8 anionic basis, these measurements define the exchange vector of a di-trioctahedral-type substitution: 3 VI(Mg, Fe2+) = VI☐ + 2 VIFe3+, as described in earlier studies. However, structural formulas calculated on the basis of the oxygen contents actually measured by EPMA show that this trend is an artifact, due to the neglect of variations in the number of protons in the structure. Our measurements indicate increasing hydrogen deficiency with increasing Fe3+ content, up to ~2 H+ pfu in the Fe3+-rich chlorite samples, corresponding to a net exchange vector of the type R2+ + H+ = Fe3+. These results highlight the need to consider substitution toward an “oxychlorite” (i.e., H-deficient) ferric component, close to tri-trioctahedral, with an O12(OH)6 anionic basis, even in green, pristine-looking chlorite. The effects of iron oxidation and H deficiency on chlorite geothermometers were explored and it was found that, given the sensitivity of most thermometers to octahedral vacancy, the assumption FeTotal = Fe2+ is still safer than using high measured Fe3+ contents and the standard 28 charge basis, which artificially increases vacancies. With the help of constraints from thermodynamic models, charge balance, crystal symmetry, and proton loss, a new cation site distribution is proposed for di-tri- to tri-trioctahedral chlorites in the Fe2+-Fe3+-Mg-Al-Si-O-H system, allowing a more realistic thermodynamic handling of their solid solutions.
Zhao et al. (p. 418) used Brillouin scattering spectroscopy to study variations in sound velocity across calcite phase transitions at pressures to 10.3 GPa. Dramatic decreases in the velocities of the compressional wave (Vp) and shear wave (Vs) and abrupt increases in the Vp anisotropy (Ap) and maximum Vs anisotropy (Asmax) were detected across the phase transition from CaCO3-I to CaCO3-II. Dramatic increases in the Vp and Vs and an abrupt decrease in Ap were observed across the phase transition from CaCO3-II to CaCO3-III. The phase transition from CaCO3-I to CaCO3-II may potentially explain the Gutenberg discontinuity at 51 km in the Izu-Bonin region. The Vp and Vs values of calcite were low. The new results combined with literature data suggest that the low velocities of CaCO3 could potentially explain the low-velocity zone occurring in northeastern (NE) Japan.
Wei et al. (p. 425) studied the distribution of trace elements in sulfosalts (bournonite, jamesonite, tetrahedrite, boulangerite, semseyite, heteromorphite, robinsonite and (Cu)-Pb-Bi-Sb sulfosalts) and coexisting base-metal sulfides in auriferous veins from the Gutaishan Au-Sb deposit, China, by electron probe microanalysis and by laser ablation-inductively coupled plasma-mass spectrometry (LA-ICP-MS) trace-element analysis. Two generations of native gold are documented; the first is coarse-grained, Ag- and Bi-bearing, and is associated with the main (Cu)-Pb-Sb sulfosalts (bournonite, jamesonite, tetrahedrite, and boulangerite). The second generation is fine-grained and has the highest fineness. Increase in the complexity of sulfosalt assemblages, re-distribution of Ag within coarse native gold and dissolution-reprecipitation reactions among the sulfosalt-gold association increase the gold fineness. All (Cu)-Pb-Sb sulfosalts analyzed were found to be remarkably poor hosts for gold. Trace, yet measurable, concentrations of Au are, however, noted in the (Cu)-Pb-Bi-Sb sulfosalts, in agreement with published data indicating that (Cu)-Pb-Bi-Sb sulfosalts may be minor Au-hosts in some ore systems. Silver is preferentially partitioned into tetrahedrite at the expense of other sulfosalt phases, and tetrahedrite is thus the major host for Ag in the Gutaishan deposit. Cd and Co are typically enriched in sphalerite relative to any sulfosalt, and when present, pyrite is always enriched in Au and Co relative to all other phases. The present study shows that linking petrographic aspects at the micrometer-scale with minor/trace element distributions in complex sulfidesulfosalt assemblages can track a complex history of Au deposition and enrichment.
Bollmeyer et al. (p. 438) report their investigation of carbonate substitution into apatite. The substitution of carbonate is particularly important because of the presence of carbonate in bone mineral and the recent suggestion that most of the substituted carbonate resides in the apatite channels (A-type substitution) rather than in place of phosphate (B-type substitution). To better understand the A-type substitution, a Sr homolog of Ca hydroxylapatite was studied because of its larger channel volume and a greater unit-cell a-axial length than its unsubstituted parent. Strontium hydroxyl-, chlor-, and fluorapatites, containing incorporated 13C-carbonate up to 7 wt%, were synthesized by aqueous precipitation reactions in the presence of Na, K, and ammonium counter cations. These samples were studied by infrared spectroscopy (IR) and 13C MAS NMR. These IR and NMR spectra were interpreted as representing three channel environments (A-type substitution: A, A′, A″) and one B-type substitution. Heating samples to 600 °C resulted in the loss of carbonate and conversion to A-type carbonate demonstrating the stability of A-type carbonate at higher temperatures. Analysis of the populations of A-, A′-, and A″-, and B-sites for the hydroxyl-, chlor-, and fluorapatites prepared under both low Na and high Na conditions revealed that high Na/carbonate ratios produce a larger amount of channel substitution, contrary to observations for Ca homologs. It is speculated that multiple A-environments also exist for Ca hydroxylapatite prepared by aqueous precipitation, which is consistent with previous analysis of apatite prepared at high temperature and high pressure.
Ma and Liu (p. 447) report the first discovery of a Zn-rich mineral on the pristine surface of orange pyroclastic beads from Apollo sample 74220. This Zn-rich mineral is widely occurring, trigonal or hexagonal in shape, with a normalized composition of ~59 wt% Zn, ~26 wt% O (calculated), ~6 wt% S, ~5 wt% Na, and ~4 wt% Cl. The crystal morphology, homogeneity, and chemistry of individual grains are most consistent with gordaite, a zinc chlorohydroxosulfate mineral, showing an empirical formula of Na1.02Zn3.98[(SO4)0.84(OH)0.30](OH)6[Cl0.50(OH)0.50]·nH2O, albeit the exact amounts of OH and H2O are uncertain. The authors concluded that this zinc-rich mineral likely formed through rapid alteration (oxidation and hydration) by terrestrial air of the original vapor-deposited Zn, Cl, S, and Na-bearing solids. The composition of the zinc-rich mineral indicates that the vapor condensates consist of metallic Zn and metallic Na with either ZnS or native S, and either ZnCl2 or NaCl. This is the first direct evidence that metallic Zn and Na are key components in the vapor condensates of lunar volcanic gas, which implies lunar volcanic gas may be under higher pressure than previously thought, and the gas composition may be different than previously inferred. Additionally, the formation of this mineral indicates that detailed protocols for the handling of extra-terrestrial samples must be constructed to minimize sample modifications (e.g., destruction of previous minerals or growth of new minerals) during collection, handling, curation, and sample preparation.
McCanta et al. (p. 453) investigated ferric iron variations in lunar glasses by X-ray absorption spectroscopy mapping. Multivariate analysis (MVA) allows selection of specific channels in a spectrum to inform predictions of spectral characteristics. Here, the sparse model of the least absolute shrinkage and selection operator (Lasso) is used to select key channels in XAS channels that can be used to predict accurate in-situ Fe3+ analyses of silicate glasses. By tuning the model to use only six channels, analytical time is decreased enough to allow mapping of Fe3+ variations in samples by making gridded point analyses at the scale of the XAS beam (1–2 μm). Maps of Fe3+ concentration can then be constructed using freely available, open source software (http://cars.uchicago.edu/xraylarch/). This result shows the enormous potential of using MVA to select indicative spectral regions for predicting variables of interest across a wide variety of spectroscopic applications. Redox gradients in lunar picritic glass beads first observed with point analyses are confirmed through this XAS mapping and suggest degassing processes during ascent and eruption are responsible for the range of Fe3+ values measured in these samples.
The Letter by Li et al. (p. 459) reports their success in using high-angle annular dark-field scanning transmission electron microscopy to depict the structural motifs in Pb-(Bi-Sb)-sulfosalts. Using two homologs from the kobellite homologous series, a group of “chessboard derivative structures,” represented by Bi-, and Sb-rich pairs of natural phases (the kobellite-tintinaite isotypic series and giessenite-izoklakeite homeotypic series), we visualize the slabs underpinning crystal structural modularity for the N = 2 homolog kobellite and the N = 4 homolog, in this case, a Bi-rich izoklakeite [Sb/(Sb+Bi) = 0.35]. The homolog number, N, can be readily calculated as N = n1/6 – 1 and N = n2/4, where n1 and n2 are the numbers of atoms in the PbS- and SnS-motifs, respectively. Atom-scale imaging of thinned foils extracted in situ from samples for which compositional data are available also reveals syntactic unit-cell scale intergrowths on [001] zone axis with akobellite || bizoklakeite. These are as small as half-unit cells of bizoklakeite and one-unit cell akobellite. Replacement relationships are also observed as irregular slabs of kobellite “intruding” into izoklakeite. Both banded and irregular intergrowths account for the compositional fields measured at the micrometer scale.
Johnson (p. 463) Reviews the new book entitled Mineralogy of Uranium and Thorium. The author concludes that Mineralogy of Uranium and Thorium is an accessible and engaging book for anyone with an interest in the mineralogy and crystallography of U- and Th-bearing minerals and the ore deposits from which they are mined.
February 2019
This issue of American Mineralogist starts with an extensive review by Luguet and Pearson (pages 165-189) on Re-Os isotopic dating of mantle peridotites using the main Re-Os host minerals—base metal sulfides (BMS) and platinum group minerals (PGM)—versus whole-rock peridotites. Comparison of the results obtained at the two scales indicates that (1) BMS may provide a record of much older partial melting event, pushing back in time the age of the lithospheric mantle stabilization (BMS±PGM are considered as the mantle equivalents of crustal zircons), (2) if only whole-rock peridotite Re-Os analyses are possible, the best targets for constraining the timing of lithospheric stabilization are BMS-free/poor ultra-refractory spinel-bearing peridotites with very minimal metasomatic overprint, (3) while lherzolites are “fertile” in terms of their geochemical composition, they do not have a “primitive,” unmodified composition, and (4) the combined Re-Os isotopic investigations of BMS and whole-rock in BMS-rich mantle peridotites would provide a complementary view on the timing and nature of the petrological events responsible for the chemical and isotopic evolution and destruction of the lithospheric mantle. In addition, the 187Os/188Os composition of the BMS±PGM within any single peridotite may define several age clusters—in contrast to the single whole-rock value—and thus provide more accurate information on the formation and evolution of the lithospheric mantle.
D’Errico et al. (pages 190-206) conducted in situ measurements of Pb and other trace elements in 150 abyssal peridotite sulfide grains (predominantly pentlandite) from the Gakkel and Southwest Indian ridges using the Sensitive High-Resolution Ion Microprobe with Reverse Geometry (SHRIMP-RG). The goal was to provide constraints on the storage of Pb and associated elements in the mantle. The authors developed a model for sulfide petrogenesis to explore the implication of assuming that all mantle Pb is hosted in mantle sulfides prior to melting. The results indicate that the measured average Pb concentration of 4 ppm (varying from 0.1 to 36 ppm) can be reproduced by >90% fractional crystallization from a sulfide melt. The remaining sulfide melt, which is modeled to contain 800 ppm Pb, will dissolve into silicate melt, as it rises through the mantle due to the increasing solubility of sulfur in silicate melt as pressure decreases. However, the amount of sulfide melt that remains after fractional crystallization is too low to contribute a significant amount of Pb to mid-ocean ridge basalts. Therefore, sulfides are not the main host for mantle Pb, even prior to the onset of any melting, and that the majority of mantle Pb is stored in silicate phases.
Griffin et al. (pages 207-219) reported a unique occurrence of magmatic hibonite-grossite-spinel assemblages, crystallized from highly reduced Ca-Al-rich silicate melts trapped within aggregates of hopper/skeletal corundum, found as ejecta from Cretaceous pyroclastic deposits on Mt Carmel, N. Israel. The crystallization conditions were comparable to those of their meteoritic counterparts. Coarse-grained aggregates of hibonite + grossite + spinel +fluorite ± krotite ± perovskite ± Ca4Al6O12F2 represent a further evolution of the silicate melts. Native vanadium occurs as rounded inclusions in the hibonite, grossite, and spinel of the coarse aggregates. The presence of V0 requires fO2 ≤ ΔIW-9, suggesting a decline in fO2 by ~3 log units during the crystallization of this assemblage. The late crystallization of Ca4Al6O12F2 together with fluorite in the hibonite-grossite-spinel aggregates suggests that crystallization of the aggregates began at T >1400 °C, cooled to the pseudo-eutectic grossite + fluorite + Ca4Al6O12F2 + liquid at ca. 1375 °C, and remained at T >1150 °C until crystallization was terminated by the volcanic eruption. This study reported the first terrestrial example of the crystallization of hibonite and grossite from high-T silicate melts, the first terrestrial occurrence of krotite, and the first occurrence of native vanadium melts.
Bollinger et al. (pages 220-231) performed deformation experiments of polycrystalline forsterite at pressures of 3.5–5.0 GPa, temperatures of 1000–1200 °C, and a strain rate of ˜2 × 10–5s–1 at various applied strains in a 6-axis Mavo press to determine the contribution of individual mechanism (dislocation creep, diffusion creep, grain boundary sliding) to olivine deformation. They developed a methodology that allows the usage of the split-cylinder technique to extract information of the deformation mechanisms from an internal surface (with engraved strain markers) in the sample before and after the deformation experiments. The results suggest the dominance of intragranular deformation, in agreement with the fact that the samples have been deformed in the dislocation creep regime. Moreover, from strain markers and out-of-plane displacements of grains, the authors obtained the first microstructural evidence for a contribution of grain boundary sliding to plastic deformation at upper mantle pressure. Electron backscatter diffraction data indicate that grain boundary processes become increasingly relevant at temperatures above 1100 °C and ensure homogenous plastic strain distribution in the aggregate. Since olivine is the major component of the upper mantle, this study sheds important light on the plasticity and rheological behavior of the mantle.
Aja (pages 232-243) reported the thermodynamic properties of two natural chlorites—a magnesian chamosite and a ferroan clinochlore )—derived from calorimetric and low-temperature hydrothermal measurements. Moreover, the author developed a molecular modeling approach to calculate the excess thermodynamic properties of chlorite solid solutions. The obtained excess entropy of mixing in the ternary Al-rich and Si-rich system exhibits a curvilinear dependence on composition, and at 25 °C, the excess Gibbs energy of mixing varies from about –72 to 413 kJ/mol, implying a significant deviation from ideality. The molecular solid solution model further reveals significant deficiencies in the available database of standard state thermodynamic properties of chlorites. Application of the results to examine the neoformation of authigenic iron chlorites in green rusts suggests that green rusts will readily transform to berthierine and Fe-chlorites except under oxidizing conditions atypical of aquatic environments and ferrugineous sediments.
Gao et al. (pages 244-261) investigated the pyroxene lamellar exsolutions and associated Fe–Ti oxides and spinels in clinopyroxne of an olivine gabbro sample from the Panzhihua intrusion, Southwest China, using high-angle annular dark-field scanning transmission electron microscopy, electron diffraction, and energy dispersive spectroscopy. The results indicate a sequence of nanoscale processes: from higher-T (~1030–1100 °C): (1) (clino)enstatite exsolutions in low-Ca diopside, followed by (2) slightly Ca-richer diopside overgrowths and high-T titanomagnetite exsolution in diopside; to lower-T (<450 °C) (3) titanomagnetite exsolutions into ulvöspinel + magnetite; followed by (4) sub-solidus re-equilibration in clinopyroxenes and among Fe–Ti oxides + hercynite. Using the exact phase boundary theory, the authors estimated the pressures of lamellar exsolution within the host diopside to be ~2 GPa. This study demonstrates that a nanoscale approach can help constrain the petrogenetic evolution during formation of layered intrusions.
Fan et al. (pages 262-275) measured the acoustic wave velocities and density of a periclase single-crystal by Brillouin light scattering combined with in situ synchrotron X ray diffraction up to ~30 GPa and 900 K in an externally heated diamond-anvil cell. Based on a comparison of the obtained elastic moduli of periclase with those of ferropericlase reported in the literature, the authors developed a comprehensive thermoelastic model for ferropericlase with up to ˜20 mol% FeO to evaluate the effect of Fe-Mg substitution on the elasticity and seismic parameters of ferropericlase at the lower mantle P-T conditions. The modeling results indicate that both the increase of the Fe content in ferropericlase and the increasing depth could change the compres¬sional wave anisotropy and shear wave splitting anisotropy of ferropericlase in the upper parts of the lower mantle. Furthermore, the authors conclude that Fe-induced lateral heterogeneities can significantly contribute to the observed seismic lateral heterogeneities in the lower mantle.
Burnley and Kaboli (pages 276-281) conducted a suite of low strain deformation experiments on polycrystalline San Carlos olivine using a deformation DIA apparatus combined with in situ synchrotron X-ray diffraction at temperatures of 440–1106 °C and pressures of 3.8–4.6 GPa. The obtained data were fitted using elastic plastic self-consistent (EPSC) models, which incorporate an isotropic deformation mechanism that permits a small amount of non-elastic defor¬mation during the initial elastic portion of the experiment. This deformation mechanism mimics the observed reduction in the elastic modulus as a function of temperature and allows for better modeling of the remainder of the stress-strain curve. The critical resolved shear stresses (CRSS) for slip obtained from these models are in good agreement with those measured in single-crystal deformation experiments. Hence, polycrystalline deformation experiments analyzed with an EPSC model may be a viable approach to measure CRSS under conditions where single-crystal deformation experiments are more challenging.
Liu et al. (pages 282-290) studied the dehydration kinetics of antigorite by thermogravimetric analysis using different heating rates of 10, 15, 20, and 25 K/min at temperatures up to 1260 K. The data were best fitted with the double-Gaussian distribution activation energy model (2-DAEM), in which a compensation effect exists between the pre-exponential factor and the average activation energy. The determined activation energy of the first step of antigorite dehydration stretches over a wide interval, whereas the second step has a significantly higher activation energy, distributed over a narrower interval. The release rate of water is 8.0×10–5 and 2.1×10–3 m3fluidm3rocks–1 at 893 and 973 K, respectively, which are near the onset temperature for the isothermal dehydration reaction. The results indicate that antigorite dehydration is fast enough to induce mechanical instabilities that may trigger seismicity in the lower plane of the double seismic zone.
Deng et al. (pages 291-299) measured the sound wave velocities and density (ρ) of the Fe5Si (9 wt% Si) alloy that possesses a body-centered cubic (bcc) structure using ultrasonic technique and synchrotron X ray radiography combined with a Paris-Edinburgh press at pressures of 2.6–7.5 GPa and temperatures of 300–1173 K. The results show that at room temperature, the addition of Si to bcc-Fe increases the compressional wave velocity (vP) but decreases the shear wave velocity (vS). At high temperatures, a pronounced effect of pressure on the vS-T relations is observed. In the studied P-T range, the vP-ρ relationship follows the Birch’s law, whereas the vS-ρ relation exhibits complex behavior. Combined with planetary/spacecraft observations, these results have important implications in constraining the compositions of the lunar and Mercurian cores.
Merkulova et al. (pages 300-306) reported the first observation of the incorporation of As3+ in goldfieldite [Cu12(As,Sb,Bi)2Te2S13] and As5+ in colusite [Cu26V2(As,Sb)4Sn2S32] inclusions in pyrite (which contains up to 50 ppm As1-) from high-sulfidation deposits in Peru, using electron probe microanalysis, synchrotron X-ray fluorescence and absorption spectroscopy. The two Cu sulfide inclusions range from several to one hundred micrometers in size, and the As3+/As5+ concentration varies from a few ppm to 17.33 wt%. The results indicate that oxidizing hydrothermal conditions prevailed during the late stage of the mineralization process in the ore deposits, and provide new insights into the substitutional mechanisms of As3+ and As5+ in copper sulfosalts. From an environmental perspective, high concentrations of potentially toxic As contained in pyrite may pose a heretofore unrecognized threat to ecosystems in acid mine drainage settings.
Brounce et al. (pages 307-312) performed measurements of the oxidation state of S in lunar apatites and associated mesostasis glass using synchrotron X‑ray absorption near edge structure spectroscopy. The results show that lunar apatites and glass contain dominantly S2–, whereas Earth apatites are only known to contain S6+. It is likely that many terrestrial and martian igneous rocks contain apatites with mixed sulfur oxidation states. The S6+/S2– ratios of such apatites could be used to quantify the fO2 values at which they crystallized, given information on the portioning of S6+ and S2– between apatite and melt and on the S6+/S2– ratios of melts as functions of fO2 and melt composition. Such a S-in-apatite oxybarometer could be developed and applied to igneous rocks from various planetary bodies in our solar system.
The issue ends with a list of the 2018 reviewers for American Mineralogist, who are thanked for their invaluable services to the journal.
January 2019
The January issue of American Mineralogist starts with a “Highlights and Breakthroughs” by Su and Liu (page 1). In their short contribution, they discuss the importance of the study by Lai et al. (published in last October's issue of American Mineralogist) on the thermoelastic properties of Fe7C3, a candidate component for the Earth's inner core.
On page 2, Cambell et al. demonstrate that the combination of zeolitized proxy-glass signatures in alkaline-mafic pyroclastic deposits and Rhyolite-MELTS can provide new insights into the magmatic evolution of mafic alkaline systems. The predictive capability of the novel procedure is demonstrated in the case of a major caldera-forming eruption, the 355 ka Villa Senni event of the quiescent Colli Albani volcano, Rome, Italy, and its pervasively zeolitized Tufo Lionato deposit (>50 km3). The key finding is that a more-evolved residual melt fraction has been revealed, based on a reconstructed SiO2/Al2O3 ratio of 2.05 relative to that of the parent magma at 2.68, with implications for a reappraisal of pre-eruptive conditions and eruption mechanisms, and potentially for similar patterns across the volcanic stratigraphy and for other alkaline volcanoes.
Chapman et al. (page 17) used large-scale large-scale electron backscatter diffraction (EBSD) and microbeam analysis to investigate crystallographic orientation and mineral chemistry data and quantify the proportion of relict igneous and neoblastic minerals forming a variably deformed, Cretaceous orthogneiss from Fiordland, New Zealand. Distinct metamorphic stages can be identified by texture and chemistry and were at least partially controlled by strain magnitude. At the grain-scale, the coupling of metamorphism and crystal plastic deformation appears to have permitted efficient transformation of an originally igneous assemblage. The effective distinction between igneous and metamorphic paragenesis and their links to deformation history enables greater clarity in interpretations of the makeup of the crust and their causal influence on lithospheric scale processes.
Mosefelder et al. (page 31) investigate nitrogen incorporation in Earth materials by a combination of chemical (SIMS, EPMA, and laser-extraction mass spectrometry) and spectroscopic (FTIR) observations to study nitrogen contents and speciation mechanisms in silicate glasses, metal alloys, and an N-bearing silicate mineral (hyalophane). They demonstrate the general veracity of EPMA analysis of N in these samples and using SIMS show that the N content determined by EPMA (or laser extraction) are best fit with exponential functions rather than the linear regressions that are most commonly applied to SIMS data. They infer that under reducing conditions at high pressure and temperature N is dissolved in basaltic melts chiefly as NH−2 and NH2–, with N2 and/or nitride (X-N3–) complexes becoming increasingly important at low fO2, increasing N content, and decreasing H content. Our results have implications for future studies seeking to accurately measure N by SIMS and for studies of N partitioning at high pressure relevant to planetary accretion and differentiation.
Wang et al. (page 47) measured deformation mechanisms in anhydrous and hydrated (4-60 ppm H2O) olivine. The hydrated and dehydrated olivines were sheared in the [100] direction on the (001) plane at pressures of 2 to 5 GPa and temperatures of 1473 or 1573 K then observed by transmission electron microscopy on the (001) plane to determine whether the [100](001) slip system was activated or not. Only c-elongated [100] dislocations were observed for the anhydrous samples, while [100](001) dislocations dominated in the hydrous samples. These results support the idea that E-type fabrics can exist under hydrous conditions and that a transition to this fabric may be the cause of seismic anisotropy decrease with depth in the asthenosphere.
Liu et al. (page 53) collected in-situ high-temperature Raman and Fourier transform infrared (FTIR) spectra for both a synthetic [Mg9Si4O16(OH)2] and a natural, F-bearing, [Mg7.84Fe0.58Mn0.01Ti0.25(SiO4)4O0.5(OH)1.30F0.20], clinohumite sample up to 1243 K. Three OH bands above 3450 cm–1 are detected for both the natural and synthetic samples with negative temperature dependence, due to neighboring H-H repulsion in the crystal structure. Additional OH peaks are detected for the natural sample below 3450 cm–1 with positive temperature dependence, indicating that F- substitution significantly changes the high-temperature behavior of hydrogen bonds in the humite-group minerals. The mode Grüneisen parameters (γiP, γiT), as well as the intrinsic anharmonic parameters (ai) for clinohumite, chondrodite, and phase A, the dense hydrous magnesium silicate (DHMS) phases along the brucite–forsterite join were also evaluated. The averaged anharmonic parameters for the DHMS phases are systematically smaller (no more than 2% at 2000 K) than those of olivine and suggest that quasi-harmonic approximations are valid for clinohumite at subduction zone temperatures. Hence, the classic Debye model can reasonably simulate the thermodynamic properties (e.g., heat capacity) of these DHMS phases in subduction zones.
Liu et al. (page 64) experimentally investigated the stability of hydrous phases in mafic oceanic crust under deep subduction conditions by high-pressure and high-temperature experiments at 17–26 GPa and 800–1200 °C. In contrast to previous studies, three hydrous phases, including Fe-Ti oxyhydroxide, Al-rich phase D and Al-rich phase H, were present at the investigated P-T conditions. These results, in combination with published data on the stability of hydrous phases at lower pressures, suggest that a continuous chain of hydrous phases may exist in subducting, cold, oceanic crust (≤1000 °C): lawsonite (0–8 GPa), Fe-Ti oxyhydroxide (8–17 GPa), Al-rich phase D (18–23 GPa), and Al-rich phase H (>23 GPa). Therefore, in cold subduction zones, mafic oceanic crust, in addition to peridotite, may also carry a substantial amount of water into the mantle transition zone and the lower mantle.
Diego Gatta et al. (page 73) investigated ettringite, (Ca6Al2(SO4)3(OH)12·26H2O), a secondary-alteration mineral with more than 40 wt.% H2O and an important crystalline constituent of Portland cements. The crystal structure and crystal chemistry of ettringite were investigated by electron microprobe analysis, infrared spectroscopy, and single-crystal neutron diffraction at 20 K. Anisotropic neutron structure refinement allowed the location of (22+2) independent H sites, the description of their anisotropic vibrational regime and the complex hydrogen-bonding schemes. Analysis of the difference-Fourier maps of the nuclear density showed a disordered distribution of the inter-column (“free”) H2O molecules of the ettringite structure. Because disorder is still preserved down to 20 K, the authors are inclined to consider that as a “static disorder.” The structure of ettringite is largely held together by hydrogen bonding: the building units (i.e., SO4 tetrahedra, Al(OH)6 octahedra, and Ca(OH)4(H2O)4 polyhedra) are interconnected through an extensive network of hydrogen bonds. The effect of the low-temperature stability of ettringite and thaumasite on the pronounced “Sulfate Attack” of Portland cements, observed in cold regions, is discussed.
Yang et al. (page 79) Investigated the behavior of hydrogen defects in 10 natural clinopyroxene crystals at temperatures up to 1000 °C using in situ and quenched experiments. The in situ high-T Fourier transform infrared (FTIR) spectra indicate no proton transfer between point defects, but the local environments of hydrogen defects vary. Dehydration rates at 1000 °C of the six samples are not only slightly site-specific but also increase with Fe and tetrahedrally coordinated Al contents. Near-FTIR spectra suggest that the dehydration of the studied samples involves oxidation of Fe2+. For two diopsides with a mantle affinity, the diffusivity is about 10–12 m2/s at 1000 °C. The results imply that the different local environments of hydrogen defects between high T and low T may be responsible for the different mechanism of water impact on electrical conductivity between high and low T experiments; and because hydrogen diffusivities are positively related to Fe and IVAl contents, more care is required for interpretation of measured water concentrations in clinopyroxenes with high Fe and IVAl contents. Based upon the hydrogen diffusivities of olivine, orthopyroxene, and clinopyroxene in mantle peridotite, clinopyroxene should be the most reliable recorder of water from a given depth.
Komabayashi et al. (page 94) examined the phase transition between a face-centered cubic (fcc) and hexagonal close-packed (hcp) structures in Fe-4wt% Si and Fe-6.5wt% Si alloys to 71 GPa and 2000 K by in situ synchrotron X-ray diffraction. The fcc-hcp phase boundaries in the Fe-Si alloys are located at higher temperatures than that in pure Fe, indicating that the addition of Si expands the hcp stability field. The dP/dT slope of the boundary of the fcc phase in Fe-4wt% Si is similar to that of pure Fe, but the two-phase region is observed over a temperature range that widens with increasing pressure, from 50 K at 15 GPa to 150 K at 40 GPa. The triple point, where the fcc, hcp, and liquid phases coexist in Fe-4wt% Si, is placed at 90–105 GPa and 3300–3600 K with the assumption that the melting curve is same as Fe. These results support the hypothesis that the hcp phase is stable at Earth's inner core conditions. The core of Mercury (well below the triple point), containing an Fe-Si alloy with a Si content up to 6.5 wt% would likely crystallize an inner core with an fcc structure. Both cores from Venus and Mars are currently believed to be totally molten. Upon secular cooling, Venus is expected to crystallize an inner core with an hcp structure, as the pressures are similar to those of the Earth's core (far higher than the triple point), whereas the Martian inner core will take an hcp or fcc structure depending on the actual Si content and temperature.
Hong et al. (page 100) studied distinctive quartz-rich unidirectional solidification textures (USTs) in apical carapaces of the Sn-mineralized Heemskirk Granite in western Tasmania (SE Australia). Individual UST layers consist dominantly of hexagonal quartz (>95%) with minor K-feldspar, plagioclase, biotite, muscovite, and magnetite. Multiple UST-quartz layers are intercalated with aplitic layers, and can locally extend for hundreds of meters. The Ti-in-quartz geothermometer yields temperatures of 545 ± 40 and 580 ± 20 °C (at 130 MPa) for the UST and aplitic quartz, respectively. The UST-quartz have higher Al/Ti values and Ge/Ti values than quartz phenocrysts in aplite layers, which is consistent with crystallization from a highly evolved fluid. LA-ICP-MS analyses show that UST-quartz has lower Ti, Li, and Sn than aplitic quartz, but higher Al, Li, Na, K, Mn, Fe, Ge, Rb, and Cs concentrations. The O-isotopic compositions (+5.1 to +10.2‰) of UST and aplitic quartz are consistent with magmatic source circulated by minor meteoric and/or formation waters. Scanning electron microscope-cathodoluminescence (SEM-CL) reveals that aplitic quartz is homogeneous and CL-bright with minor CL-dark patches. The bases of the UST quartz crystals are homogeneous and CL-bright with minor thin CL-dark fractures, whereas the trigonal apexes of the UST-quartz display CL-oscillatory growth zones. The results show that the UST layers in the Heemskirk Granite precipitated from magmatic-hydrothermal aqueous fluid exsolved from granitic melt during emplacement into the shallow crust (6–10 km). Such UST layers are characteristics of mineralized intrusions, and therefore provide significant indications for mineral exploration.
Cheng et al. (page 118) evaluate controls on cassiterite crystallization under hydrothermal conditions based upon the texture and geochemistry of cassiterite from a traverse from close to the host granitic pluton out into the mineralized country rock (Gejiu tin district, southwest China). The cassiterite samples feature diverse internal textures, as revealed by cathodoluminescence (CL) imaging, and contain a range of trivalent (Ga, Sc, Fe, Sb), quadrivalent (W, U, Ti, Zr, Hf), and pentavalent (Nb, Ta, V) trace elements, with Fe, Ti, and W being the most abundant trace elements. Cassiterite Ti/Zr ratios tend to decrease with distance away from the granite intrusion, and potentially can be used as a tool for vectoring toward a mineralized intrusive system. Elemental mapping of cassiterite grains reveals that trace-element concentration variations correspond closely to CL zoning patterns. The exceptions are distinct irregular domains that sharply cut across the primary oscillatory zoning, as defined by the concentrations of W, U, Sb, and Fe. Zones with low W and U (and Sb) and high Fe are interpreted to have formed during interaction with relatively oxidized fluids in which W and U are stripped from cassiterite due to cation exchange with Fe3+. Systematics of W, U, Sb, and Fe partitioning into cassiterite can, therefore, be used as a monitor of the relative oxidation state of the hydrothermal fluid from which cassiterite precipitates. Cassiterite U-Pb ages determined on zones of dissolution-reprecipitation are similar to ages for primary cassiterite growth and demonstrate a short (<3 m.y.) timespan of hydrothermal activity, indicating the potential of U-Pb dating of cassiterite for constraining the timing of Sn deposition.
Mookherjee et al. (page 130) performed high-pressure, high-temperature experiments on lithological compositions resembling hydrated sedimentary layers in subducting slabs and found that the phase egg, AlSiO3(OH), is stable to pressures of 20–30 GPa or depths equivalent to the transition zone to lower mantle. Thus, phase egg is a potential candidate for transporting water into the Earth's mantle transition zone. First-principles simulations based on density functional theory explored the pressure dependence of crystal structure and its influence on energetics and elasticity. The phase egg exhibits anomalous behavior of the pressure dependence of the elasticity at mantle transition zone depths (~15 GPa). The anomalous behavior is related to changes in the hydrogen bonding O-H···O configurations, which were delineated as a transition from a low-pressure to a high-pressure structure of phase egg. Full elastic constant tensors indicate that phase egg is anisotropic, resulting in a maximum anisotropy of compressional wave velocity, AvP ≈ 30% and of shear wave velocity, AvS ≈ 17% at zero pressure. Results indicate that the phase egg has one of the fastest bulk sound velocities (vP and vS) compared to other hydrous aluminous phases in the Al2O3-SiO2-H2O ternary, which include topaz-OH, phase Pi, and δ-AlOOH. At depths corresponding to the base of the mantle transition zone, phase egg decomposes to a mixture of δ-AlOOH and stishovite. The changes in compressional ΔvP and shear ΔvS velocity associated with the decomposition is ~0.42% and –1.23%, respectively. Although phase egg may be limited to subducted sediments, it could hold several weight percentages of water along a normal mantle geotherm.
Kaminsky et al. (page 140) found polycrystalline diamond grains within the Valizhgen Peninsula in Koryakia, northern Kamchatka, Russia. One grain from the Aynyn River area studied by TEM contained diamond crystallites, 2–40 μm in size, that are twinned and have a high dislocation density. The crystallites are cemented by tilleyite Ca5(Si2O7)(CO3)2, SiC, Fe-Ni-Mn-Cr silicides, native silicon, graphite, calcite, and amorphous material. Three polymorphs of SiC were discriminated: hexagonal 4H and 6H and cubic C3 (β-SiC). Silicides have variable stoichiometry with (Fe,Ni,Mn,Cr)/Si = 0.505–1.925. Native silicon is an open-framework allotrope of silicon S24, which appears to be a new natural mineral phase. Three types of amorphous material were distinguished: a Ca-Si-C-O material, similar in composition to tilleyite; amorphous carbon and amorphous SiO2. Diamond crystallites and moissanite are intensively twinned, which is characteristic when these minerals formed by gas phase condensation or chemical vapor deposition (CVD) processes. The synthetic analogs of all other cementing compounds (β-SiC, silicides, and native silicon) are typical products of CVD processes. This confirms the earlier suggested CVD mechanism for the formation of Avacha diamond aggregates. Both Avacha and Aynyn diamond aggregates are not related to “classic” diamond locations within stable cratons, but to areas of active and Holocene volcanic belts. The studied diamond aggregates from Aynyn and Avacha, by their mineralogical features and by their origin during the course of volcanic eruptions via a gas phase condensation or CVD mechanism, may be considered a new variety of polycrystalline diamond and may be called “kamchatite.” Kamchatite extends the number of unusual diamond localities. It increases the potential sources of diamond and indicates the polygenetic character of diamond.
Zhang et al. (page 150) describe spherical (Mg,Fe)-oxides with a protrusion surface in a shock-induced melt pocket from the Martian meteorite Northwest Africa 7755. Transmission electron microscopic observations demonstrate that the (Mg,Fe)-oxides are structure-coherent intergrowth of ferropericlase and magnesioferrite. The magnesioferrite is mainly present adjacent to the interface between (Mg,Fe)-oxides spherules and surrounding silicate glass, but not in direct contact with the silicate glass. Thermodynamic and kinetic considerations suggest that development of the spherical (Mg,Fe)-oxides can be best interpreted with crystallization by particle attachment and subsequent Ostwald ripening. This indicates that crystallization by particle attachment (previously hypothesized to occur in low-temperature aqueous natural and synthetic systems) can take place in high-temperature melts and has potential implications for understanding the nucleation and growth of early-stage crystals in high-temperature melts, such as chondrules in the solar nebula, erupted volcanic melts, and probably even intrusive magmas.
Etschmann et al. (page 158) provide an experimental confirmation of the suggestion, based on thermodynamic simulations and extrapolations (Zhong et al. 2015), that Zn is transported in the form of chloride complexes in most acidic, shallow hydrothermal systems; while bisulfide complexes become increasingly important in deep, pH neutral to basic hydrothermal systems. We used in situ X-ray absorption spectroscopy (XAS) diamond-anvil cell experiments to determine Zn(II) speciation in a 1 m NaHS + 0.2 m HCl solution in contact with sphalerite. XANES data indicate that Zn coordinates to oxy/hydroxyl/chloride ligands from room temperature up to and including 200 °C, and then at higher temperatures (≥300 °C) and pressures (>200 MPa) it changes to complexing with sulfur. Our data confirm that bisulfide complexes become increasingly important in neutral-alkaline solutions at high pressure and temperature, due to an increase in sulfur solubility and to favorable entropy contributions for bisulfide vs. chloride complexes.
Elimi (page 162) reviews the book: Infrared and Raman Spectroscopies of Clay Minerals, Volume 8, Developments in Clay Science, 1st Edition, by Will Gates, J. Theo Kloprogge, Jana Madejova, and Faïza Bergaya. (2017) Elsevier, pp. 620.