

Mineralogical Society of America, Founded December 30, 1919
Subscriber Access MinPubs.org Pay-Per-View
Order Publications Online (25% discount for MSA, CMS and GS members, except shipping)


2017, i-xvi + 885 pages. ISBN 978-0-939950-98-0
Traditional stable isotope geochemistry involves isotopes of light elements such as H, C, N, O, and S, which are measured predominantly by gas-source mass spectrometry. Even though Li isotope geochemistry was developed in 1980's based on thermal ionization mass spectrometry (TIMS), the real flourish of so-called non-traditional stable isotope geochemistry was made possible by the development of multi-collector inductively coupled plasma mass spectrometry (MC-ICPMS). Since then, isotopes of both light (e.g., Li, Mg) and heavy (e.g., Tl, U) elements have been routinely measured at a precision that is high enough to resolve natural variations. The publication of RIMG volume 55 (Geochemistry of Non-Traditional Stable Isotopes) in 2004 was the first extensive review of Non-Traditional Stable Isotopes summarizing the advances in the field up to 2003. When compared to traditional stable isotopes, the non-traditional stable isotopes have several distinctive geochemical features:
These features make the different elements susceptible to different fractionation mechanisms, and by extension, make them unique tracers of different cosmochemical, geological and biological processes, as highlighted throughout this volume.
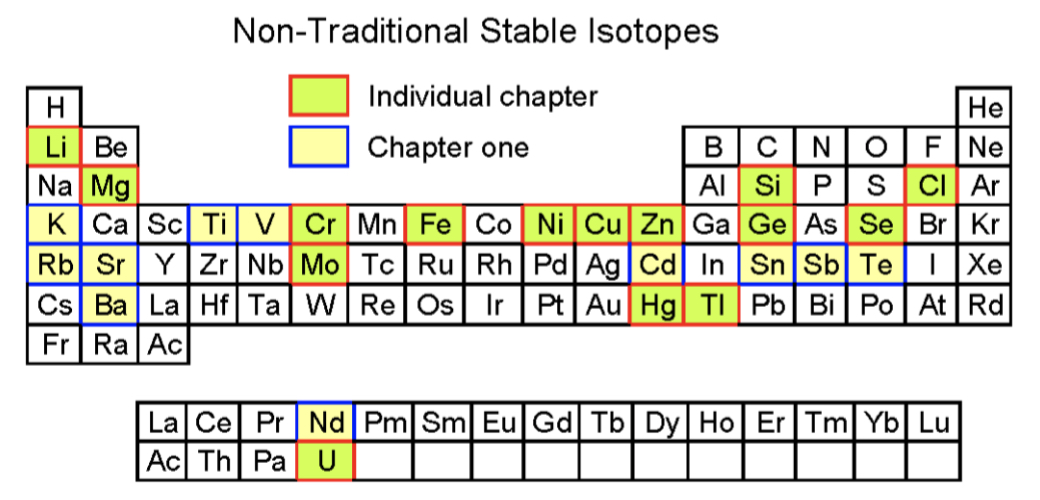
Large variations have been documented in both natural samples and laboratory experiments for non-traditional stable isotopes. These studies suggest that the following factors control the degree of isotope fractionation in non-traditional stable isotopes during various processes: the relative mass difference between isotopes, change of the oxidation state, biological sensitivity, and volatility. Among these elements, Li displays the largest isotopic variation in terrestrial samples, and since Li is not volatile during geological processes and is not sensitive to redox reactions and biological processes, the large isotope fractionation is controlled mainly by the large relative mass difference. For many of the other elements, other factors may be equally, if not more, important. For example, Chlorine exhibits the second largest isotopic variation, which is due to kinetic isotope fractionation during volcanic degassing. Selenium isotopes also show large isotopic variations, but this reflects redox-controlled Se isotope fractionation, while the large mercury isotopic variations are mainly associated with biological processes.
From Chapter 1.
Fang-Zhen Teng, Isotope Laboratory, Department of Earth and Space Sciences University of Washington, Seattle WA, USA
Nicolas Dauphas, Origins Lab, Department of the Geophysical Sciences and Enrico Fermi Institute, The University of Chicago, Chicago IL, USA
James M. Watkins, Department of Earth Sciences, University of Oregon, Eugene OR, USA
January 2017
Title Page
p. i
Copyright
p. ii
From the Series Editor
p. iii
Preface
p. iii
Table of Contents
p. iv-xiv
Chapter 1. Non-Traditional Stable Isotopes: Retrospective and Prospective
by Fang-Zhen Teng, Nicolas Dauphas, James M. Watkins, p. 1-26.
Errata for the print version of Chapter 1 as a pdf (Download (98 KB))
Chapter 2. Equilibrium Fractionation of Non-traditional Isotopes: a Molecular Modeling Perspective
by Marc Blanchard, Etienne Balan, Edwin A. Schauble, p. 27-64.
Chapter 3. Equilibrium Fractionation of Non-Traditional Stable Isotopes: an Experimental Perspective
by Anat Shahar, Stephen M. Elardo, Catherine A. Macris, p. 65-84.
Chapter 4. Kinetic Fractionation of Non-Traditional Stable Isotopes by Diffusion and Crystal Growth Reactions
by James M. Watkins, Donald J. DePaolo, E. Bruce Watson, p. 85-126.
Chapter 5. In Situ Analysis of Non-Traditional Isotopes by SIMS and LA–MC–ICP–MS: Key Aspects and the Example of Mg Isotopes in Olivines and Silicate Glasses
by Marc Chaussidon, Zhengbin Deng, Johan Villeneuve, Julien Moureau, Bruce Watson, Frank Richter, Frédéric Moynier, p. 127-164.
Supplementary Tables 1 through 7 (as a MicroSoft Excel file) Download (119 KB)
Chapter 6. Lithium Isotope Geochemistry
by Sarah Penniston-Dorland, Xiao-Ming Liu, Roberta L. Rudnick, p. 165-218.
Supplement Appendix on Li flux contamination (as a pdf file) Download (18 KB)
Errata for both the online and print versions of Chapter 6 as a pdf (Download (98 KB))
Chapter 7. Magnesium Isotope Geochemistry
by Fang-Zhen Teng, p. 219-288.
Errata for the print version of Chapter 7 as a pdf (Download (98 KB))
Chapter 8. Silicon Isotope Geochemistry
by Franck Poitrasson, p. 28-344.
Chapter 9. Chlorine Isotope Geochemistry
by Jaime D. Barnes, Zachary D. Sharp, p. 345-378.
Chapter 10. Chromium Isotope Geochemistry
by Liping Qin, Xiangli Wang, p. 379-414.
Chapter 11. Iron Isotope Systematics
by Nicolas Dauphas, Seth G. John, Olivier Rouxel, p. 415-510
Chapter 12. The Isotope Geochemistry of Ni
by Tim Elliott, Robert C. J. Steele, p. 511-542.
Chapter 13. The Isotope Geochemistry of Zinc and Copper
by Frédéric Moynier, Derek Vance, Toshiyuki Fujii, Paul Savage, p. 543-600.
Chapter 14. Germanium Isotope Geochemistry
by Eva E. Stüeken, p. 657-682.
Chapter 15. Selenium Isotopes as a Biogeochemical Proxy in Deep Time
by Tim Elliott, Robert C. J. Steele, p. 511-542.
Chapter 16. Good Golly, Why Moly? The Stable Isotope Geochemistry of Molybdenum
by Brian Kendall, Tais W. Dahl, Ariel D. Anbar, p. 683-732.
Chapter 17. Recent Developments in Mercury Stable Isotope Analysis
by Joel D. Blum, Marcus W. Johnson, p. 733-758.
Chapter 18. Investigation and Application of Thallium Isotope Fractionation
by Sune G. Nielsen, Mark Rehkämper, Julie Prytulak, p. 759-798.
Chapter 19. Uranium Isotope Fractionation
by Morten B. Andersen, Claudine H. Stirling, Stefan Weyer, p. 799-850.
Chapter 20. Medical Applications of Isotope Metallomics
by Tim Elliott, Robert C. J. Steele, p. 851-885.