| Home | AmMin | GMR | RiMG | Collectors Corner | Directory | Short Courses | |
|
|
|||||||

|
|
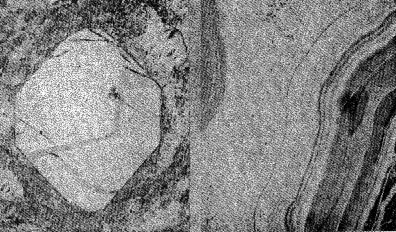
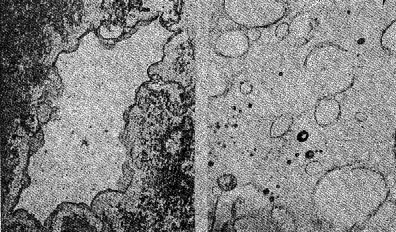
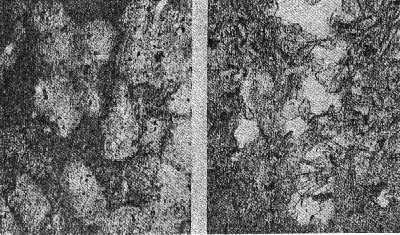
Volume 13, pages 73-92, 1928 NATURAL HISTORY OF THE SILICA MINERALS* AUSTIN F. ROGERS, Stanford University. For any mineral or group of minerals there are two general classes of facts to be ascertained: (1) the geometrical, physical, and chemical properties; (2) the mode of occurrence, association, and origin, or briefly what may be called the natural history of the mineral. To the geologist, of course, the natural history of a mineral is its most attractive feature, but until the mineral is accurately defined and determined, the significance of its occurrence and origin may be lost sight of. It is my purpose to discuss the various silica minerals. The silica minerals are with a single exception accurately defined; they have been produced in the laboratory; they constitute the most complete example of polymorphism known. They are, moreover, of considerable geological interest, and altogether we have a fairly good idea of the role that the silica minerals play in Nature. Let us first consider briefly the silica minerals and their properties as a background for the discussion of their natural history. LOW QUARTZ Though by no means the most common, quartz is probably the best known of all minerals. The ancient Greeks called it crystallus, in the belief that it was permanently congealed water, and thus was derived our word crystal. Quartz has been investigated from almost every standpoint, and its physical constants have been very accurately determined. The symmetry of low quartz is trigonal trapezohedral (A3 · 3A2). Twinned crystals of quartz are not uncommon. The twinning may be reflection-twinning (Japan law), rotation-twinning (Dauphiné law) or inversion-twinning (Brazil law). The symmetry is made evident by the etching of crystals or polished spheres by hydrofluoric acid. HIGH QUARTZ On heating quartz, Le Chatelier, the French chemist, noticed a rapid change in some of its properties at a temperature of about 570°C. This has been confirmed by subsequent investigators, and Wright and Larsen1 place the inversion point at 575°±2°C. At this temperature there is a sudden change in the properties, which means that a new substance is produced. The new substance is hexagonal trapezohedral,2 with the symmetry A6·6A2. It has been called high quartz to distinguish it from the ordinary form which is low quartz. The difference in symmetry of the two forms is shown by Laue X-ray photographs, which means that there is a difference in the arrangement of the atoms or molecules in the two cases. The crystal habit of high quartz is usually short and stumpy with narrow prism faces (Plate 1, Fig. 1) in contrast with the prismatic habit of low quartz. CHALCEDONY The microcrystalline form of silica known as chalcedony is very much like low quartz in its properties, but seems to be somewhat different from low quartz. It does not appear to grade into quartz, and may easily be distinguished from it in practically all cases. Plate I, Fig. 2 shows the appearance of interbanded chalcedony and quartz. Chalcedony shows evidence of rhythmic banding, which reaches a remarkable development in agate. In polarized light between crossed nicols, it shows an aggregate structure which is very characteristic. Whether chalcedony is a distinct form of silica or simply a variety of quartz is uncertain. A reasonable explanation is that chalcedony is an aggregate of silica fibers which are oriented in one direction only. In quartz the fibers are oriented in all directions. This assumes the thread structure advocated by Sosman.3 OPAL Opal is a hydrogel mineral of colloidal origin. In some specimens we have evidence of its origin by dessication of a gelatinous mass of silica. Opal is probably a solid solution of water in silica. When first formed it consists of two phases, silica and water, but in time the water gradually diffuses into the silica and a solid solution is formed. Opal is not, then strictly speaking, a colloid, but only colloidal in origin. When formed in open spaces hyalite opal shows a colloform 3a structure (Plate I, Fig. 3), often with double refraction due to strain set up in the hardening of the gel.Opal is apparently one of the few strictly amorphous minerals, since it gives no X-ray diffraction pattern. 4TRIDYMITE In 1868, vom Rath described a new form of silica from an augite andesite near Pachuca, Mexico. This he named tridymite from the fact that the crystals are often cyclic twins made up of three individuals. Since then it has proved to be a widely distributed mineral in volcanic rocks. Perhaps the best known locality in this country is Obsidian Cliff in Yellowstone National Park, an occurrence made famous by Iddings' description in the Seventh Annual Report of the United States Geological Survey. The typical appearance of tridymite is shown in Plate III, Fig. 12 and Plate V, Fig. 18. CRISTOBALITE This mineral was found associated with the tridymite from Cerro San Cristobal near Pachuca, Mexico, and was named by vom Rath in 1887. It was first established as a distinct mineral by Mallard in 1890, but was not generally recognized until 1913, when Fenner proved that its indices of refraction are different from those of tridymite. It is distinguished with difficulty from tridymite, and has often been mistaken for the latter mineral. Plate II, Fig. 5 and Plate III, Fig. 10, show its typical appearance in thin sections. Cristobalite is an isometric form of silica. It is found in minute octahedrons, in spherulites alone, and in spherulites intergrown with orthoclase. In mode of occurrence it is much like tridymite. In 1918 it was described by the writer 5 from two localities in California, and since that time it has been found in more than a dozen localities in the western part of the United States. It is a widely distributed mineral, but usually occurs in small quantities. In this country it is known from more localities than is tridymite.A typical occurrence of cristobalite in minute secondary spherulites in the lithophysae of a rhyolitic obsidian from Little Lake, Inyo County, California, has been described by the writer. 6Cristobalite and tridymite are of more than purely scientific interest. The artificial minerals are the principal constituents of silica bricks, 7 which are made by burning ground-up quartzites containing from 97 to 98 per cent of silica. Fig. 6, Plate II represents a thin section of silica brick in ordinary light. The curved fracture of cristobalite is characteristic. The specimen of silica brick was kindly furnished me by my colleague, Professor W. J. Crook.LECHATELIÉRITE The latest of the silica minerals to be described is lechateliérite, named by Lacroix 8 in 1915 in honor of Henry Le Chatelier, the famous French chemist, whose work9 on silica and silicates is well known. Lechateliérite is naturally occurring silica glass. It has not been generally recognized as a distinct mineral.In its properties lechateliérite is much like opal, but in origin it is absolutely different. As Miers 10 has well said: "The essential characters of a mineral, moreover, are quite independent of its source or previous history . . . . ." This statement is important for crystalline minerals, but is probably not valid for amorphous substances such as opal and lechateliérite. At any rate, it is convenient to treat these two as distinct minerals. About a century ago Breithaupt distinguished two classes of amorphous substances, the hyaline or glasses and the porodine or those of colloidal origin. It is unfortunate that the term hyalite has been used as a variety of opal.Lechateliérite is unique in that it is the only naturally occurring glass that is definite enough to be considered a mineral. Lechateliérite is the chief constituent of fulgurites which are formed by the fusion of quartzose sand by lightning. It also occurs in some volcanic inclusions. There is a unique occurrence at Meteor Crater, in Arizona, which I shall describe later. Plate I, Fig. 4 is a photograph of a thin section of the lechateliérite from Meteor Crater. Silica glass, which is often incorrectly called "quartz glass", is now made on a commercial scale in a very pure transparent form. Silica glass possesses some unusual physical properties such as transparency to ultraviolet light and a remarkably small coefficient of thermal expansion. A rod of the glass 1 meter in length will expand only about 0.6 mm. for a rise of temperature of 1000°C. Some remarkably clear silica glass has recently been produced by the General Electric Company at Lynn, Massachusetts. 11THE SILICA MINERALS IN GENERAL The various silica minerals and some of their properties are shown in the tabulation given on p. 78. All the known forms of silica, and they are legion in number, 12 are believed to be synonyms or varieties of the ones mentioned. Asmanite, found in meteorites, is a synonym of tridymite. Lussatite, considered by some to be distinct form, is probably a mixture of opal and fibrous chalcedony.It seems probable that all the forms of silica capable of existence at atmospheric pressures are now known. But it is not safe to say that all the possible distinct forms of silica are known, for polymorphism is a general phenomenon of nature and there is no theoretical limit to the number of polymorphous modifications of any chemical substance. The stability relations of most of the silica minerals is shown in a diagram which appeared in a paper by Fenner 13 of the Geophysical Laboratory, published in 1913.The artificial production of the various forms of silica under specified conditions in the laboratory has been a great help in the proper understanding of their natural history. Let us next consider the typical modes of occurrence of the various silica minerals with a view of determining, as far as possible, the conditions under which they were formed in nature. OCCURRENCE OF OPAL Opal is a late secondary mineral found in seams and cavities of various rocks, especially of volcanic igneous rocks. It is found in sedimentary beds which are in part at least formed from diatoms, radiolaria, and sponge spicules. Opal is unique in that it is the only silica mineral of direct organic origin. It must be formed over a considerable range of temperature, for it is found as geyserite in hot springs, and as a coating formed in broken rock surfaces made since the opening of quarries. For example, I have found it on broken Pennsylvanian limestones at Kansas City, Mo. It seems plausible to regard opal as a mineral which has formed rapidly. Our information concerning the formation of minerals of colloidal origin is rather meager. OCCURRENCE OF CHALCEDONY Chalcedony is a typical metacolloid, a convenient term introduced by Wherry 14 to denote microcrystalline minerals of colloidal origin. Its temperature range is probably considerably less than that of low quartz, for it seems to be limited to deposits formed near the surface. It is probably formed more rapidly than low quartz and this may account for its characteristic properties. The exact conditions of its formation, however, are unknown. More study is needed.OCCURRENCE OF LOW QUARTZ Low quartz is a typical hydrothermal mineral which is probably always formed below 575°C. Its temperature range is considerable. Its occurrence as secondary enlargements of sand grains in sandstones as well as numerous occurrences in sedimentary rocks proves that it may be formed at temperatures at little above the ordinary. Vein quartz usually has a prismatic habit.
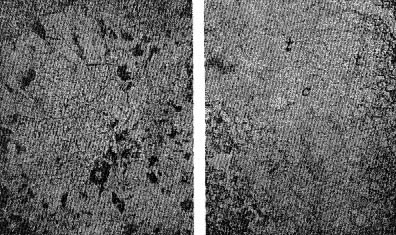
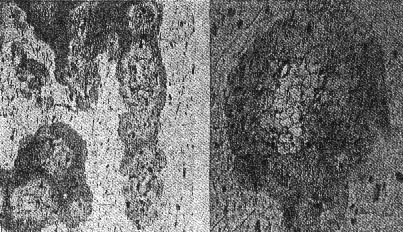
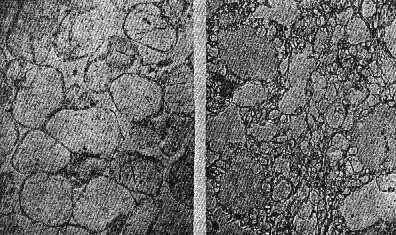
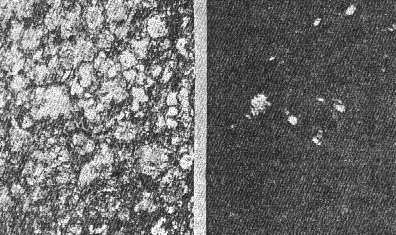
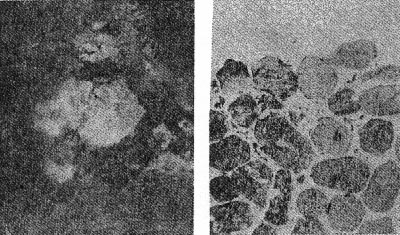
OCCURRENCE OF HIGH QUARTZ High quartz is the original mineral of persilicic igneous rocks present because there is an excess of silica left over after the affinities of silica for the various oxides are satisfied. At atmospheric pressure under laboratory conditions it is formed above 575°C. but in nature it has probably been formed at lower temperatures because of the influence of mineralizers. 575°C. as a point on the geological thermometer15 cannot be accepted too literally. High quartz usually shows a bipyramidal habit due to the minor development of the prism faces. (See Plate I, Fig. 1). OCCURRENCE OF TRIDYMITE Tridymite is the stable form of silica between 870°C. and 1470°C. under laboratory conditions. In occurrence it is practically confined to volcanic igneous rocks. As a rule it is formed after cristobalite. According to Fenner16 "tridymite is sometimes found as an essential constituent of acid effusives, associated in such manner with other minerals as to imply its separation from the melt as a primary constituent." I doubt very much whether tridymite is ever formed as a magmatic mineral. The "acid effusives" referred to by Fenner are the tridymite latites from the Creede district, Colorado, recently described by Emmons and Larsen.17 Concerning these rocks it is said that the "tridymite forms in large part during the later stages of the crystallization of the groundmass." For my part, I doubt whether tridymite is ever even a late magmatic mineral. The tridymite latite is a dark red-brown banded rock with irregular streaks and lenses of a paler tint which are coarsely crystalline and contain abundant euhedral crystals of tridymite. A similar occurrence of tridymite from Catalina Island, off the coast of southern California, has been studied by the writer. (See Plate II, Fig. 7). The tridymite-bearing streaks I would interpret as having been formed by pneumatolytic metamorphism. The term pneumatolysis was originally used by Bunsen for volcanic processes, but has since been employed largely in connection with alterations brought about by emanations from intrusive rocks. In my opinion tridymite is not a magmatic mineral, but a metamorphic one. The occurrence of euhedral crystals in cavities may be explained by local migration of the silica. To substantiate my conclusion that tridymite is a product of metamorphism I wish to present briefly the results of my study of a tridymite-feldspar rock which occurs near the Salton Sea in Imperial County, California. This rock makes up the whole of a small hill which before the recession of Salton Sea was known as Cormorant Island. A prospector, on sinking a shaft on this island or hill, was overcome by gas, and sent the rock encountered to Stanford University for identification. It proved to contain tridymite as an essential constituent. The rock superficially resembles a calcareous tufa. Field work proved that the white to light gray tridymite-feldspar rock was formed from a typical rhyolitic obsidian. The obsidian is found as bands and lenses in the tridymite-bearing rock. These bands and lenses are clearly residual. The origin of this unusual rock type has been confirmed by the microscopic examination of a considerable number of thin sections. Gradations may be recognized from rhyolitic obsidian through spherulitic obsidians (Plate III, Fig. 9) and dense devitrified obsidians (cristobalite-barbierite rocks) (Plate III, Fig. 11) to porous somewhat banded tridymite-barbierite (the feldspar is a monoclinic one with soda dominant over potassa, so that Schaller's term "barbierite" is used) rocks (Plate III, Fig. 12). Chemical analyses confirm the field and microscopic work. The final stage is a distinctive rock type which must be regarded as metamorphic if we adopt the definition of metamorphism suggested by Daly18 and used by Holmes19 and others. The metamorphic agent was doubtless hot gases. The presence of hot springs around the hill and mud volcanoes in the immediate vicinity lends support to this theory of origin. OCCURRENCE OF CRISTOBALITE Under laboratory conditions cristobalite is stable between 1470° and 1625°C. In nature it is practically always found in volcanic igneous rocks and is a frequent associate of tridymite. It is far more common in volcanic glasses such as rhyolitic obsidians, than is tridymite. It occurs frequently in cavities and has been formed either at a very late magmatic period or at a very early period of alteration, due to hot gases, in all probability. My study of cristobalite-bearing rocks convinces me that it is a characteristic mineral of spherulites (pseudospherulites of Rosenbusch) as an interstitial mineral between the rods or fibers of feldspar. Plate II, Fig. 8 is a photograph of a portion of a cristobalite-feldspar spherulite from Casa del Diablo, Mono County, California, collected by Mr. Robert Loofbourow. This interstitial mineral has usually been identified as tridymite or quartz or sometimes as opal. In the younger rhyolites and obsidians this interstitial material is cristobalite. If the interstitial mineral of persilicic spherulitic rocks is cristobalite, it means that it is not such a rare mineral as has been supposed. In the older devetrified rhyolites, quartz is found between the fibers of feldspar of the spherulites. This does not necessarily mean that quartz here is an original mineral. In fact it seems probable that in these cases quartz is an alteration product of cristobalite. There is as yet no direct proof of this, but it seems a reasonable explanation. OCCURRENCE OF LECHATELIÉRITE Above 1625°C., silica glass under laboratory conditions is the stable form of silica. It is certain that such a temperature has not often been reached on or near the earth's surface and it is no wonder that lechateliérite is such a rare mineral. I now wish to describe a unique occurrence of silica glass. The locality is Meteor Crater, or Coon Butte, a crater-like depression about three-fourths of a mile in diameter and nearly 600 feet in depth, in Coconino County, Arizona, near the main line of the Santa Fe railway. Around the rim of this immense depression there are beds of a white saccharoidal sandstone, a red calcareous sandstone, and an arenaceous magnesian limestone which are more or less disturbed from their original horizontal position. The white or pale gray saccharoidal sandstone (Coconino sandstone) has been altered to three different kinds of material as follows: (1) rock flour, (2) a rather dense, somewhat banded quartz rock of a unique type, the quartz grains of which show in thin sections prominent rhombohedral cleavage, and (3) a vesicular glass of nearly pure silica which is the lechateliérite. Microscopic examination proves conclusively that the silica glass is the result of fusion of the saccharoidal sandstone. This was first proved years ago by G. P. Merrill,20 and of this fact there is not the slightest doubt. A thin section section of the loosely-consolidated sandstone is shown in Plate IV, Fig. 13. Plate IV, Fig. 14 shows the highly vesicular silica glass. The three oval areas represent what were originally sand grains now converted entirely to lechateliérite. Plate IV, Fig: 15 represents a thin section of a banded, slightly porous silica glass in which the granular structure of the original sandstone is preserved. In this rock there are few residual specks of quartz, as may be seen when the thin section is examined between crossed nicols. (Plate IV, Fig. 16) The mineralogist or petrographer is interested in determining the origin of the lechateliérite, and the geologist is primarily concerned with the origin of the crater-like depression which Arrhenius declared is "the most interesting spot on the earth's surface." The fusion of the sandstone and the formation of the depression were without doubt brought about by the same geological agent. In the immediate region of this depression many hundreds of specimens of meteoric iron (these meteorites are known by the name "Canon Diablo") have been found, and this gave rise to the term Meteor Crater, used for the depression. A possible explanation of the formation of Meteor Crater is that it was due to the impact of an immense meteorite. Barringer,21 Fairchild, Branner, Tilghman, Elihu Thomson, Magie, and D. L. Webster have expressed themselves as being in favor of the meteoritic theory of origin. Gilbert, Guild, T. C. Chamberlin, Darton, and recently Thurmond, on the other hand, are in favor of its origin by a steam explosion of some kind and attribute the distribution of the Canon Diablo meteorites to a coincidence. Besides the lechateliérite and the quartz grains of the sandstone and limestone, the only silica mineral found in any of the rocks of this locality is a minute amount of cristobalite embedded in some of the the lechateliérite. Tridymite, chalcedony, opal, and newly formed quartz are conspicuous by their absence. The nearest volcanic rocks, which are basalts, are about ten miles away. Outside of the vesicular silica glass which occurs in layers at least 6 inches thick, there is nothing at Meteor Crater to suggest vulcanism in any form, and there is nothing except the depression to suggest a steam explosion. One would expect a steam explosion to leave some evidence of its work in some kind of rock alteration, but there is none visible. It is almost inconceivable that a temperature in the neighborhood of 1600°C. (about the melting-point of quartz) could have been reached during any part of a steam explosion even if the steam were superheated. Taking all the facts into consideration, it seems most reasonable to attribute the formation of Meteor Crater to the impact of a huge meteorite and the fusion of the sandstone to form lechateliérite to the beat generated by the impact. If this be true, we have at Meteor Crater, Arizona, a unique spot on the earth's surface, but one that perhaps could be duplicated if we had a complete geological history of the earth. TRANSFORMATIONS OF THE SILICA MINERALS When the silica minerals are studied in the laboratory, there are found to be two types of inversion: (1) Inversions of the low and high forms of quartz, tridymite, and cristobalite respectively, which take place rapidly. (2) Inversions of quartz, tridymite, and cristobalite from one into another. - These inversions are sluggish, and so are of geological importance. The inversion temperatures of the various polymorphous forms of silica have been determined under laboratory conditions by the Geophysical Laboratory.22 These laboratory experiments are of course extremely important, but the mineralogist and petrographer are primarily interested in the natural occurrences and transformations. I next wish to present the results of my study of some of the natural transformations of the silica minerals. OPAL TO CHALCEDONY Opal, being amorphous, is unstable. There is evidence that it changes to microcrystalline chalcedony. Many massive specimens of silica consist of mechanical mixtures of opal and chalcedony. The form of silica known as lussatite is in all probability such a mixture. In the Monterey formation of the Coast Ranges of south central California, bands of massive opal are often in whole or in part converted into chalcedony.23 The specimen shown in thin section by Plate V, Fig. 17 represents an intermediate stage in the transformation. TRIDYMITE TO QUARTZ Paramorphs of low quartz after tridymite were first described by Mallard from the Euganeen Hills, Italy, under the name "pseudo-tridymite". These are thin tabular six-sided crystals 3 or 4 mm. in size. They have the form of tridymite, but consist of quartz. Such pseudomorphs have been found at a number of localities. Within the last few years I have collected good examples of quartz paramorphs after tridymite in three different occurrences in Santa Clara County, California. One of these occurrences is an altered vitrophyre which occurs as a "puy" known as Lone Hill, near San Jose. A photograph of a thin section examined in ordinary light and between crossed nicols gives convincing proof of the transformation mentioned. (Plate V, Fig. 18) Tridymite is unstable under laboratory conditions below 870°C. and so under some circumstances we find it transformed to low quartz by hydrothermal solutions. In confirmation of this it should be noted that the altered vitrophyre contains pyrite. TRIDYMITE TO CRISTOBALITE In 1918 I discovered in the cavities of a hornblende andesite at Jamestown, California, crystal aggregates which have the characteristic form of tridymite but consist of cristobalite, as was proved by the refractive index. This is the first record of cristobalite paramorphs after tridymite.24 Since then a similar occurrence has been described from near Eschwege, Hesse-Nassau, Prussia, by Ramdohr,25 and recently I have found another good example in an andesite from near Tucson, Arizona, kindly furnished me by Professor F. N. Guild. Both the Jamestown and Tucson occurrences contain acicular crystals of an unusual amphibole and thin tabular crystals of anauxite, a pseudo-hexagonal aluminum silicate first described from Bilin, Bohemia, by Breithaupt in 1838. Ross and Kerr26 have recently shown that anauxite is an important .constituent of some clays. Plate V, Fig. 19, is a photograph of one of the cavities of the Tucson andesite containing the paramorphs, amphibole, and anauxite. In all the occurrences examined by me in which both tridymite and cristobalite occur together, I have found tridymite to be the later mineral, though Larsen27 in the Creede, Colorado, rocks, reports cristobalite to be later than tridymite. One would naturally expect to find tridymite paramorphs after cristobalite rather than the reverse, as the temperature would normally decrease on the cooling of the andesite. In view of the work of the Geophysical Laboratory, I would explain the cristobalite paramorphs after tridymite as due to a hot blast of gas probably brought about by chemical reactions in the gaseous state. In confirmation of this hypothesis, I would mention various alteration products which do not seem to be due to either hydrothermal solutions or meteoric waters, for example, the hematitic dust in the groundmass of the Jamestown andesite. This explanation supports Le Chatelier's28 view that cristobalite is formed rapidly. CRISTOBALITE TO QUARTZ The occurrence of quartz-feldspar spherulites in devitrified rhyolites with the same texture as cristobalite-feldspar spherulites in recent rhyolites and obsidians is considered evidence that the transformation of cristobalite to quartz has taken place in nature, but no direct proof can be given at the present time. SUPPOSED TRANSFORMATION OF QUARTZ TO OPAL The last photograph shown (Plate V, Fig. 20) represents a thin section of opalized wood from the Fernando (Pliocene) near Santa Maria, California, presented to me by my colleague, Professor James Perrin Smith. The area shown is one produced by insect borers of some sort. These seem to be opal paramorphs after quartz, and they were so interpreted. The change of quartz to opal would be a new and interesting type of transformation, and we might invent several hypotheses to account for this conversion. But even better than multiple hypotheses is the determination of new facts. On discussing this specimen with one of my biology colleagues, Professor Harold Heath, he told me and proved to me that the pellets (excreta) of recent termites are hexagonal in cross-section. The supposed paramorphs are nothing but opalized termite pellets, and these constitute a new type of fossil. In conclusion, I would suggest that the various silica minerals constitute one of the most important and interesting of all our mineral groups. While our knowledge in some respects is fairly complete, we have much to learn, especially with regard to the low temperature forms. PLATE I. THIN SECTIONS OF ROCKS WITH VARIOUS SILICA MINERALS.
PLATE II. THIN SECTIONS OF ROCKS WITH VARIOUS SILICA MINERALS
PLATE III. THIN SECTIONS OF ROCKS FROM CORMORANT ISLAND, SALTON SEA, IMPERIAL COUNTY, CALIFORNIA
PLATE IV. THIN SECTIONS OF ROCKS FROM METEOR CRATER (COON BUTTE), COCONINO COUNTY, ARIZONA.
PLATE V. THIN SECTIONS AND HAND SPECIMEN SHOWING TRANSFORMATIONS OF VARIOUS SILICA MINERALS.
NOTES * Presidential address presented at the eighth annual meeting of The Mineralogical Society of America, in joint session with The Geological Society of America, Cleveland, Ohio, December 29, 1927. 1 Am. Jour. Sci., [4], Vol. 27, pp. 421-447 (1909). 2 Mügge, Neues Jahrb. f. Min. Geol., u. Pal., Festband, pp. 181-196 (1907). 3 Jour. Frank. Inst., vol. 194, pp. 741-764 (1922). 3a Jour. Geol. vol. 25, p. 518 (1917). 4 Kerr, Econ. Geol., vol. 19, pp. 1-34 (1924). 5 Am. Jour. Sci., [4]], vol. 45, pp. 222-226 (1918). 6 Jour. Geol., vol. 30, pp. 211-216 (1922). (See Fig. 1 on p. 213). 7 McDowell, Bull. 119, Am. Inst. Min. Eng., pp. 1999-2055 (1916). 8 Bull. Soc. Fran, de Min., vol. 38, pp. 182-186 (1915). 9 La Silice et les Silicates, Paris (1914). 10 Mineralogy, p. v., London, (1905). 11 Berry, Smithsonian Report for 1924, pp. 213-218. 12 Hart, this journal, vol. 12, pp. 383-395 (1927). 13 Amer Jour. Sci., [4], vol. 36, pp. 331-384 (1913). 14 Jour. Wash. Acad. Sci., vol. 4, p. 112 (1914). 15 Wright and Larsen, loc. cit. 16 Loc. cit., p. 344. 17 Bull. 718, U. S. Geol. Surv., p. 48 (1923). 18 Bull. Geol. Soc. Am., vol. 28, p. 375 (1917). 19 The Nomenclature of Petrology, p. 155, London (1920). 20 Smithsonian Misc. Collections, vol. 50, pp. 461-498 (1907). 21 Meteor Crater in Northern Central Arizona. Paper read by D. M. Barringer at the 1909 meeting of the National Academy of Science. Privately printed. 22 Fenner, loc. cit. 23 Tolman, Econ. Geol., vol. 22, p. 470, plates III and IV, (1927). 24 Amer. Jour. Sci., [4], vol. 45, pp. 224-225 (1918). 25 Centralblatt f. Min. Geol., u. Pal., 1920, pp. 33-36. 26 Personal communication. 27 Emmons and Larsen, loc. cit. 28 Bull. Soc. Fran. de Min., vol. 40, pp. 44-57 (1917).
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||