| Home | AmMin | GMR | RiMG | Collectors Corner | Directory | Short Courses | |
|
|
|||||||

|
|
Volume 51, pages 1227-1233, 1966
AN UNUSUAL HOURGLASS STRUCTURE IN AUGITE J. PRESTON, Department of Geology, Queen's University, Belfast, Northern Ireland. Pleochroic hourglass structures are well known in titanaugites from the alkali basic igneous rocks and are easily recognized in thin section. A more obscure hourglass structure, defined by composition and exsolution features, characterizes a common augite from a dolerite pegmatite. The pegmatite occurs as segregations in an olivine tholeiitic feeder dike-a member of the Tertiary dike swarm of Northwest Ireland. The multiple dike which crosses County Fermanagh is being described in detail (Preston, in prepn.) and the pegmatite samples were collected from its later intrusions, exposed in an old quarry in the Townland of Drogan, near Doraville, northwest of Enniskillen. In hand specimen, the augites display a fine silvery Schiller and a well developed parting on both (001) and (100) surfaces. In thin section, these features may be related to very thin exsolved lamellae. A soft serpentinous pseudomorph of low Ca content (circa 9%) contains herring-bone twins of fresh augite lamellae which demonstrate parallel growth between the original pigeonite and the coexisting augite. Exsolution has produced augite lamellae about ½ µ thick and spaced at 10-15µ intervals through the pigeonite. Exsolved lamellae in the augite are much thinner and barely visible under an oil-immersion objective. With a thickness of they are spaced more closely at ¼-½ µ intervals. Lamellae parallel to (001) are better developed in the augite and are usually confined to definite domains within a single crystal. In thin section and in transmitted light, the augite free of exsolution is clear and transparent whereas with exsolution the grain is translucent and clouded by a fine parting which masks the otherwise prominent cleavage. These two domains of a single crystal are commonly irregularly developed, but many sections oblique to c show a well defined hourglass structure. In such sections the crystal sectors which embrace the (100-001) edge show the basal parting and those which include the (001-010) edge are free from exsolution on (001) but show incipient unmixing parallel to (100) (Fig. 1, a-c).
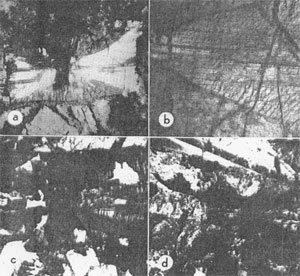
FIG. la. Section oblique to c, almost perpendicular to (001) and elongate along the b axis. A schiller reflection from the (100) exsolution lamellae illuminates the (010) sectors. Basal lamellae in the (100) sectors invade the (010) domains along narrow zones. Altered pigeonite makes a discontinuous rim near the (100) edge. X20. b. Enlarged portion of the grain in la. Fine striations due to basal lamellae define the (100) sectors and end abruptly against the clear augite, a few lamellae persist beyond the hourglass boundary. Stepped lines mark the outcropping (100) lamellae in the (010) sectors. X60. c. Herring-bone twin of altered pigeonite and augite. Basal lamellae in the augite define an elongated hourglass structure. (100) surfaces separate the twins and the parallel growths. X20. d. Near basal section of twinned pigeonite and augite in parallel growth. The pigeonite-augite boundary follows an hourglass structure. Earlier parallel growths share plane surfaces but the final augite shell covers a very irregular pigeonite surface in one twin individual. X20.
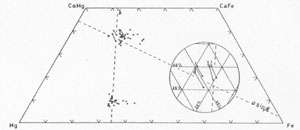
Pyroxene compositions were determined on the electron probe microanalyser in the Department of Mineralogy and Petrology, Cambridge. The Ca and Fe contents were measured and the results, corrected for matrix absorption and atomic number effect, are plotted in Fig. 2. Pairs of sample points, one from each domain, were chosen as close together as practicable (within 0.05 mm) from the core and rim of two individual grains. The results demonstrate the complementary nature of the different hourglass sectors. The translucent areas with the basal parting and exsolution are on an average 2.85 at. % richer in Ca and 2.41 and 0.43 at. % poorer in Fe and Mg respectively than the clear areas. The Mg content is practically constant and the lines linking sample pairs are almost parallel to the Ca-Fe side of the pyroxene trapezium (Fig. 2, inset). These results represent the original composition of each domain prior to exsolution. The lamellae and intervening host proved too small for individual analysis on the microanalyser. FIG. 2. Pyroxene compositions in terms of atomic % for coexisting pigeonite and augite in the dolerite pegmatites. Compositions of the two hourglass domains are joined by solid lines and replotted in the inset. Broken lines follow the isodimensional lines for b' and asinß as drawn by Brown (1960).
The (001) exsolution lamellae which characterize the Ca-rich domains are presumably of pigeonite whereas the incipient (100) lamellae of the Fe-rich sectors are assumed to be hypersthene. This implies a difference in temperature of exsolution between the two volumes. In both instances the Ca-poor pyroxene has altered to a soft chloritic substance. This enhances the parting, and thin films of chlorite and intervening plates of augite can be lifted from the fresh broken surfaces and examined in isolation. Many elongate augite prisms split their full length along the (100) parting. The chlorite pseudomorphs after hypersthene show a marked lineation parallel to (001) of the host mineral. Slight differences in color and birefringence distinguish one elongated chlorite flake from its neighbor, and, if this is due to varying Fe content, it may reflect an attempt at Ca-Fe ordering in the original pyroxene. Larger flakes of chlorite show no preferred orientation, and small calcite grains, if present, develop along fine cracks through the host. One chlorite pseudomorph after hypersthene had broad lamellar twins inclined at 45° to c of the enclosing augite. In alternate twin lamellae, the fast or slow vibration directions were parallel to c of the host.
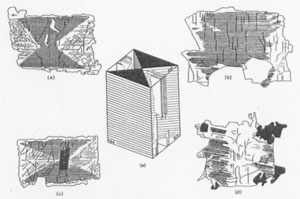
FIG. 3a. Basal section with well defined hourglass structure. Cleavage and (100) lamellae are visible in the clear augite. b. (100) section with basal lamellae in a broad central zone. c. Section oblique to c as in Fig. 1, a-b. d. (010) section With basal lamellae in marginal prismatic zones. e. Reconstruction of the hourglass structure and exsolution features. Basal lamellae are present throughout the (100) sectors whereas (100) lamellae are only partially developed in the (010) sectors. The thin plates of augite between chlorite lamellae were too thin for precise optical determinations. Their refractive index is similar to that of the exsolution-free augite whereas the 2V angle is 5-10° higher. This difference in 2V is borne out by measurements on normal thin sections. Consistent results and differences were obtained despite the masking effect of the chloritized lamellae. The transparent (010) sectors have a 2V of 47° close to that expected for the determined composition. No (100) sectors remain free from exsolution and a comparable value for 2V cannot be quoted. After exsolution the host augite changed its 2V to 58° and 52° for the (100) and (010) sectors respectively. This bears out Brown's (1960) assumption that the exsolved material would be similar in composition to the coexisting Ca-poor pyroxene which in other pegmatites averages Ca10Mgs60Fe30 at. %. As noted above, sections oblique to c may show a well defined hourglass structure (Fig. 3 a and c). Sections close to (010) display longitudinal domains of transparent and translucent augite (Fig. 3d) and the (001) exsolution is confined to the prism edge leaving a clear central zone. Similar sections slightly inclined to c develop an elongated hourglass structure (Fig. 1c). In many instances however the distribution of Ca- and Fe-rich areas is irregular, and the pattern of growth difficult to reconstruct. Sections close to (100) are also irregular but may show clear zones along the prism edges (Fig. 3b). The ideal pattern is reconstructed in Fig. 3e which demonstrates plainly that growth in the direction of a has shown a preference for Ca over Fe, and that the reverse is true for growth along b. In the direction of the c axis, growth was unaffected by selection of this kind. Growth in the direction of a appears to reach a stable composition of 40 at. % Ca; this could indicate a pattern of ordering in the M2 positions. Morimoto (1960) claims that the remaining M2 positions are filled by Fe2+ in which case the presence of Ca2+ and Fe2+ in M2 would be in the ratio of 4:1. No such ordering is apparent for the (010) sectors. The preference of a cation for a particular face may be explained by reference to Brown's (1960) work on the cell dimensions of the clinopyroxenes. In theory and observation the substitution of Ca2+, Mg2+ and Fe2+ has little effect on the c-dimension but a great effect on b and asinß. For Ca2+ and Fe2+ substitution in M2 the influence on asinß is more marked than on b. Increasing Ca content increases the (100), and decreases the (010) spacing. The larger Ca2+ cation may then prefer an ordered concentration on a growing face whose spacing it more readily determines. For the augite compositions given in Fig. 2, the Ca- content. would largely determine the asinß-cell dimension and have little influence on b, whereas the Fe-content would have no effect on asinß but control the b-cell dimension. The exsolution of the different crystal domains presents certain contradictory features, for the higher temperature unmixing of pigeonite is confined to the Ca-rich augite, and the incipient lower temperature hypersthene lamellae occur in the Fe-rich sectors. Unmixing may however reflect the greater ease of ionic migration along rather than across the silicon-oxygen chains, a factor noted by Poldervaart and Hess (1951) and a high initial degree of ordering in the Ca-rich domains. Parallel growth between augite and pigeonite is of common occurrence throughout the dolerites and pegmatites of this Tertiary dike. In the pegmatite, where both pyroxene phases occur in approximately equal amounts, this relationship is the rule and in all examples the shared plane is (100). Indeed the interatomic dimensions of augite and pigeonite are a better match on (100) than on (001), though, for the reason given above, exsolution would use the basal plane. In many instances the augite completely encloses a core of altered pigeonite and a later development of pigeonite occurs as an interrupted rim near the margin of the composite grain. The shared (100) surface is often a single plane, but the overgrowth on other lattice planes may also follow a definite pattern. (100) sections show a serrated boundary between the two phases defined by (011) clinodomes. Herring-bone twins of pigeonite have parallel growths of augite on both individuals, or vice versa (Fig. lc). Figure 1d illustrates one example where sharing of the (100) plane has been influenced by the hourglass growth. Pigeonite has obviously competed unsuccessfully for sites on the (100) plane against the Fe-rich sector of the augite. In other crystals the Ca-rich domains have been suppressed completely without the aid of pigeonite. The (100) face of the augite has the slower growth rate and crystallizing plagioclase in the vicinity may have reduced the Ca concentration below the critical level for augite growth. Poldervaart and Hess (1951) comment on the occurrence of pigeonite in highly volatile systems, especially in the pegmatitic segregations of basic igneous rocks. Pigeonite is certainly more abundant and better crystallized in these pegmatites than in the adjacent dolerite. Higher volatility would permit freer migration of the pyroxene elements through the residual melt, and the coarse-grain of the rock, the narrow crystallization interval of the augite, and the degree of exsolution all indicate very slow cooling. This last condition would be inevitable in patches of residual melt trapped in the crystal mesh of a 300 ft. wide feeder dike which had a long and active history. Conditions such as these would not be unusual during the consolidation of basic igneous rocks, and obscure hourglass structures may be of commoner occurrence among their constituent minerals. This study would not have been possible without the help of the Department of Mineralogy and Petrology at Cambridge. The author is particularly indebted to Dr. S. O. Agrell and Dr. J. V. P. Long for their assistance with the electron probe microanalyses. REFERENCES BROWN, G. M. (1960) The effect of ion substitution on the unit cell dimensions of the common clinopyroxenes. Am. Mineral. 45, 15-38. MORIMOTO, N., D. E. APPLEMAN AND H. T. EVANS JR. (1960) The crystal Structure of clinoenstatite and pigeonite. Zeit. Krist. 114, 120-147. POLDERVAART, A. AND H. H. HESS (1951) Pyroxenes in the crystallization of basaltic magma. Jour. Geol. 59, 472-489. PRESTON, J. In preparation.
|