|
(revised 05/02/2003)
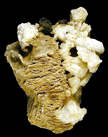
Nicholas J. Rose asks: The photo shows an unusual mineral association from a pocket in a Southern California Pegmatite. In the background is a rehealed base (now doubly terminated) of quartz crystals from which a highly corroded schorl extends out about 4 cm. Within it's hollowed out core are perched a number of orange red sphene crystals from 2mm to 5mm (see Photo 1). I have a number of questions concerning the changes in pocket chemistry that would lead to corrosion of some minerals within pockets such as schorl or beryl in one portion of the pegmatite while in other areas of the same pegmatite beryl or schorl will show little or no corrosion, so if you have anything I could read to help me in this area it would be greatly appreciated. Such zones of corrosion appear to be highly mappable and specific to individual minerals such as schorl, beryl, topaz, or herderite within the same dike. I am trying to see if an understanding of corrosion effects in the pegmatites I'm exploring can lead to better decisions on directions of future exploration.
There are three responses to date. The question and answers are also available as pdf file. (7 pages, 531 K) From Prof. Robert F. Martin, McGill University: "Hollow tourmaline need not have an origin by etching or by corrosion. Crystals that grow at high supersaturations may well grow as hollow prisms. I would suggest to Mr. Rose to consult The Canadian Mineralogist, volume 16, page 288, Figure 11. The synthetic crystals of chlorapatite look like hexagonal drinking straws. Hollow crystals result from growth, not dissolution or etching." From Dr. John S. White, Kustos "Even though I have written a short piece for Rocks & Minerals ("Disbelieving Dissolution" Nov/Dec 2002) in an attempt to refute the claim that tourmaline scepters are the result of etching, I hardly consider myself an expert on the subject. Just the same, the image that accompanied the Nicholas Rose query doesn't give one much to work with. In that particular instance I would be reluctant to assume that the hollow schorl was produced by dissolution of schorl at its center. Rather I would lean toward interrupted growth or the schorl having grown around a crystal of something else that has been etched away. I am reminded of an article that appeared some thirty or more years ago in Am. Min. (I believe) about quartz infilling hollow tourmaline crystals. I was troubled that the author offered no proof of interpretation and did not appear to consider the possibility that the quartz crystallized first and tourmaline then grew around it." From David London Drs. Martin and White have pointed out three possibilities for the hollow tourmaline crystal shown by Mr. Rose:
The tendency for crystals to grow rapidly on corners and edges, which follows from Pauling's rules for crystal growth, produces skeletal or hopper crystals in which faces have failed to advance at the same rate as corners and edges. As Prof. Martin notes, this growth habit normally results when crystallization commences from a highly supersaturated solution, and it also reflects a condition in which the growing crystal initially is far from chemical equilibrium with its nutrient solution (aqueous or magmatic). Morgan and London (1988) observed that when the driving force to precipitate tourmaline from an aqueous solution increased, the habit evolved from fully formed euhedral crystals to columnar or fibrous growth pillars in the [0001] direction of the crystal. This skeletal growth habit generates fibrous or bundled/compound single crystals of tourmaline, which are common in pegmatites, and it is also the source of the cat's-eye effect in tourmaline. The habit is different, however, from the hollow crystal shown by Mr. Rose. The second case begins as an extension of the first, except that the core of the hollow tourmaline crystal is filled with one or more minerals rather than an aqueous fluid phase. Hollow crystals of black tourmaline and beryl (filled with quartz, albite, etc) are common, as are skeletal crystals of phosphates intergrown with albite. All are analogous to graphic granite; the skeletal intergrowth of microcline and quartz (see Fenn, 1986); and all originate from the same process, in which crystals begin to grow from a medium (pegmatite-forming silicate melt in this case) that has become highly supersaturated in mineral components. Dr. White suggests that subsequent and selective dissolution might have removed the core-filling material but not the surrounding tourmaline in Mr. Rose's. Selective dissolution of certain minerals is widespread in pegmatites and may result in pseudomorphism (Fig. 1) or complete removal (Fig. 2), while surrounding minerals remain unscathed. The third case mentioned by Dr. White also entails selective dissolution, but in this case tourmaline also constitutes the core of the crystal. Compositional zoning parallel to the [0001] growth direction is common enough in tourmaline, but the question is: how different must the core composition be to dissolve completely while the rim remains essentially intact? Is this feasible? I concur completely with Prof. Martin and Dr. White that the processes they describe (skeletal growth, selective dissolution) are important phenomena in pegmatites. In the particular case of Mr. Rose's tourmaline, however, I have some samples; also from pegmatites in southern California; that appear to support an origin for these hollow crystals by selective dissolution of interior zones of tourmaline. Figure 3 shows a suite of tourmaline crystals (5 of 16 such pieces) from an extension of the Topaz Ledge at the Little Three Mine, Ramona (courtesy of L.B. Spaulding, Jr.). All samples are from the same pocket, and none have been analyzed to date. The tourmaline samples possess sharp euhedral terminations (Fig. 3a) and shiny, striated prism surfaces, but broken fragments show a physically distinct "black" core and "black" jacketing rim (Fig. 3b). Other crystal fragments manifest various degrees of dissolution of the core tourmaline only (Figs. 3c-d), up to complete dissolution leaving hollow crystal sections (Fig. 3e). There is nothing in these samples to suggest that the cores were filled with anything but tourmaline. Where dissolved, the tourmaline has recrystallized euhedral terminations projecting into the central voids from the caps. Figure 4 is a pink tourmaline crystal with a dark greenish blue jacket and cap from the Elizabeth R. mine, Pala (courtesy of Roland Reed). The pink core of the tourmaline is extensively replaced by lepidolite, which is evident as reflective cleavage flakes parallel to the [0001] growth direction, but the dark jacket shows no alteration. A backscattered electron image of a similar crystal (Fig. 5) shows the core tourmaline, partial replacement by lepidolite, and a void where the micaceous material plucked out between the partially replaced core and the unaltered cap (and jacket). The compositions of the pink core and the blue rim are quite similar; both are fluor-elbaite. One phase was highly susceptible to replacement by lepidolite (and its removal created voids in the core of the crystal), whereas the rim and cap compositions were not. There is a fundamental theory and quite a bit of published literature on the origins of skeletal crystal growth. I would suggest that interested readers consult the article by Fenn (1986) and then proceed to more theoretical works. The foundation for understanding the selective dissolution and precipitation of minerals in granite-pegmatite systems was presented by Korzhinskii (1959) and applied to granites and pegmatites by Burt (1981). It was used to explain the pseudomorphic alteration of Li-minerals in pegmatites of the White Picacho district, Arizona (London and Burt, 1982a,b), and I have discussed it again in a recent manuscript (submitted to Lithos). When a chemical system (e.g., a pegmatite-forming melt ± vapor) is forced to crystallize far from the conditions of its equilibrium, the system tends to oscillate (Nicolis and Prigogine, 1977). That is, it swings from the side of one mineral assemblage to another, and back, as it reacts to approach a state of equilibrium between crystals and melt and/or vapor. In recent papers (e.g., London, 1996), I have suggested that pegmatites represent systems that arise from this process; hence, periods in which a mineral or mineral assemblage is precipitated, then dissolved, then reprecipitated, and so forth, should be prevalent in pegmatites, and they are. Other oscillations that arise from this process include the formation of layered aplite, or "line rock" (e.g., London, 1999). The sharp mineral zonation of pegmatites can promote another episode of alternating dissolution and deposition of the same mineral phase, when at the hydrothermal (e.g., pocket) stage, aqueous fluid can make its way along fractures from one part of the pegmatite to another. Each time a parcel of fluid migrates to a new mineral assemblage, which might be a zone or just a single giant crystal (e.g., of microcline), it will react with that host to approach a new equilibrium composition. As it moves on to another assemblage or mineral environment, it is once again out of equilibrium, and continues to react by dissolving parts of one assemblage and precipitating another. None of this tells Mr. Rose, precisely, why the tourmaline at the core of his crystal might have dissolved while the rim remained intact, and that is just one of three possibilities proposed so far by Prof. Martin, Dr. White, and myself. If selective dissolution is the cause, then I suggest this may stem from the migration of hydrothermal fluid from pocket to pocket, reacting along the way. Channeled fluid flow (between pockets) and selective scouring and abrasion are evident in the pegmatites of the Little Three mine that I know of. If this hypothesis is correct, then it would suggest the presence of several pockets that became interconnected late in the history of pegmatite consolidation. I hope Mr. Rose can take that as a good omen for further prospecting. References:
Fenn, P.M. (1986) On the origin of graphic granite. American Mineralogist, 71, 325-330. Korzhinskii, D.S. (1959) Physicochemical basis for the analysis and paragenesis of minerals. Consultants Bureau, New York. London, D. (1996) Granitic Pegmatites. Transactions of the Royal Society of Edinburg, Earth Science Section 87, 305-319. London, D. (1999) Melt boundary layers and the growth of pegmatitic textures. (abstr.) Canadian Mineralogist 37, 826-827. London, D. and Burt, D.M. (1982a) Alteration of spodumene, montebrasite, and lithiophilite in pegmatites of the White Picacho district, Arizona. American Mineralogist, 67, 97-113. London, D. and Burt, D.M. (1982b) Chemical models for lithium aluminosilicate stabilities in pegmatites and granites. American Mineralogist, 67, 494-509. Morgan, G.B., VI and London, D. (1989) Experimental reactions of amphibolite with boron-bearing aqueous fluids at 200 MPa: implications for tourmaline stability and partial melting in mafic rocks. Contributions to Mineralogy and Petrology, 102, 1-17. Nicolis, N.G. and Prigogine, I. (1977) Self-organization in nonequilibrium systems: from dissipative structures to order through fluctuations. Wiley & Sons, New York.
Figure 1. pseudomorph of quartz, micas and clays after spodumene, 24 cm in length, Bennett Quarry, Buckfield, Maine.
Figure 2. quartz plus molds of albite surrounding voids once filled with spodumene, 13 cm in length, Elizabeth R mine, Pala, California. Traces of dark pink clay (referred to as Li-montmorillonite) remain in the voids.
Figure 3. Black tourmaline caps and prisms, Little Three mine, Ramona, California (courtesy of L.B. Spaulding, Jr.). Crystal diameters are ~ 1.5 cm.
Figure 4. zoned blue over pink tourmaline, partially replaced by lepidolite, 7 cm in length, Elizabeth R mine, Pala, California (courtesy of Roland Reed). Traces of dark pink clay (referred to as Li-montmorillonite) remain in the voids.
Figure 5. Backscattered electron image of pink and blue tourmaline, Elizabeth R mine, Pala, California (courtesy of Roland Reed). The pink tourmaline of the core is medium-gray, lepidolite is bright white, and the cavity formed by removal of tourmaline and mica is evident toward the left of the photo. Field of view is 1580 x 1580 micrometers. Jeffrey E. Patterson: (this reply is also available as a pdf file (1.7M!!). Drs. Martin, White, and London present interesting comments regarding some aspects of hollow tourmaline growth in pegmatites. However, they do not address the other primary questions posed by Mr. Rose, to wit, what can he read to further his knowledge base and can his observations be useful for further exploration. The literature is replete with papers dealing with criteria for exploration, including the use of mineralogical trends. Basic models, with assumptions, can be found in Cameron et al., (1949), Jahns and Burnham, (1969), and Sinkankas, (1988). An easily accessible bibliographical source on tourmalines and their properties is provided by Darrell Henry, at Louisiana State University, (http://www.geol.lsu.edu/henry/Research/tourmaline/bibliography/TurBibliography.htm). From my current research, I have observed that recent investigation of tourmaline stability and alteration to other minerals is a little studied phenomenon. The changing acidity/alkalinity of the late hydrothermal fluids circulating in pegmatites offers a unique opportunity to examine some of the basic characteristics of pegmatite formation. The work of Thomas, et al., (2003), provides several new insights to this problem. The example presented by Mr. Rose is exceptional in its characteristics, specifically with the apparent pristine nature of the remaining schorl, the terminated quartz on the exterior, and the terminated sphene on the interior. Dr. London's Figures 3, from the Little 3 Mine in Ramona, and 4 and 5, from the Elizabeth R Mine in Pala, serve to provide an additional base for the comment below. The tourmalines in Figures 1 and 2 are worldwide examples of similar phenomena, wherein the early formed tourmaline appears to be unstable with respect to the late formed rind and the interior portion has been hydrothermally leached, altered, or replaced by other minerals. For the past 9 years I have had the pleasure of being able to conduct basic pegmatite research at the Little 3 Mine. During a ground verification phase to a GPR study, (Patterson, 1996), I observed the phenomena of schorl alteration to a sodium-iron rich, lithian bearing muscovite, (Stern, et al., 1986; Foord, et al., 1989; and Figures 3 to 10). The mine owner, Louis B. Spaulding, Jr., told me that Dr. Richard H. Jahns, a constant visitor to the mine between 1954 and 1982, always said that they were muscovite pseudomorphs after tourmaline. Careful observation has proven this to be true. Apparently, late stage potassium rich hydrothermal fluids circulated upward, from either the lower dyke or the aplite / line-rock unit, and altered the primary schorl to muscovite, (Figure 3). During the early stages of alteration, the muscovite A-axis followed the schorl C-axis, forming an apparent parallel intergrowth, (Figures 5, 6, 9, and 10). During the late stages of alteration, the tourmaline was almost completely removed, leaving the muscovite to crystallize in jack-straw fashion within the tourmaline shaped cavity left in the host albite, (Figures 4 to 8). Figures 9 and 10 are photomicrographs of thin sections taken from a section of an altered tourmaline removed from the specimen shown in Figure 4. Apparently, the perthitic K-Feldspar in the footwall was also altered at this time, as it shows extensive leaching of albite along the rims of several crystals in the specimen shown in Figure 4. The released elements from this alteration of tourmaline, (Na, Fe, and B2O3), and associated Li, K, and H2O, appear to have been either incorporated into the muscovite or transported along the open pocket-line to form the new mineral boromuscovite, as exemplified by the material in the New Spaulding Pocket, (Stern, et. al., 1986; Foord, et. al, 1989; and Foord, et. al., 1991). This alteration sequence is traceable for several meters updip and along strike in the dyke. Following trends like these elsewhere, on the property, has led to the discovery of other productive pockets, (L.B. Spaulding, Jr., personal communications, 1995-2002). Alteration of tourmaline to muscovite or lepidolite is well known, as represented by the specimens in Figures 11 to 14 and in London's Figures 4 and 5. Perusal of the images in many of the issues of Mineralogical Record also depicts altered tourmalines, beryls, and other pegmatite minerals. I will leave it to the geochemists to present the appropriate chemical explanation. In answer to Mr. Rose's question on why some parts of a pegmatite are altered while others are not, I present an alteration sequence of a pegmatite in southern California that deals with the leaching of quartz from Graphic Granite intergrowths in the Garnet Ledge at the Little 3 Mine, (Figures 15 to 19). Here, in a 10 m dip-section of the dyke, all of the quartz was removed, leaving slightly altered perthitic K-feldspar and unaltered schorl, garnet, and apatite. The quartz and related hydrothermal solution appears to have been deposited updip as axinite and large, to 30 cm, quartz crystals enclosing schorl needles, garnet, and axinite. Some of this fluid escaped the confines of the pegmatite and reacted with the host granodiorite, (Patterson, 1997), to form an exocontact of very fine grained green muscovite and schorl-dravite, (Figure 18). In some pockets, cavernous beryl crystals were found, (Figure 19). Elsewhere in this same dyke, and in portions of the Topaz Ledge, similar sections containing quartz leached graphic granite can be observed. Other features of the alteration sequences of the Garnet Ledge are beyond the scope of this comment. If one considers the permeability of a crystallized pegmatite and the reactivity of the representative minerals, a common theme is observed. Where minerals are altered, they are open to exchange with late stage fluids. These fluids can either be trapped during progressive crystallization or be introduced during later episodes of emplacement. The amount of alteration appears to be a function of the stability of that phase when in direct contact with the hydrothermal solution at that moment. If a mineral is buffered by an impermeable layer or encased in a non-reactive phase, then no alteration will occur. However, if the permeability is high enough for a significant amount of fluid to infiltrate a particular zone, then the possibility exists to encounter a zone of altered material. In the case of the miarolitic pegmatites of San Diego County, if a zone of alteration is encountered, then the optimum gem or specimen grade material is probably minimal, but most probably up-dip a zone of productive material maybe found. An early example of gemmy morganite beryl altering to bavenite was presented by Schaller and Fairchild, (1932). An apparently similar scenario has been the case with the recent finds of pink and green tourmaline at the Cryo-Genie Mine, near Warner Springs, (http://home.earthlink.net/~goke/Cryo-Genie.htm). I suggest to Mr. Rose that he complete a thorough 3-D geologic map of his pegmatite to look for trends in mineralization and alteration. Some basic geochemistry might also be appropriate to delineate the levels of fractionation. I wish him luck with his endeavors and thank him for this opportunity to express my observations. References:
Foord, E.E., Martin, R.F., Fitzpatrick, J.J., Taggart, J.E., and Crock, J.G., 1991, Boromuscovite, a NEW member of the Mica Group, from the Little Three Mine Pegmatite, Ramona District, San Diego County, California. American Mineralogist, 76, 1998-2002. Foord, E.E., Spaulding, L.B., Jr., Mason, A.R., and Martin, R.F., 1989, Mineralogy and Paragenesis of the Little Three Mine Pegmatites, Ramona District, San Diego County California. The Mineralogical Record, 20, 101-127. Jahns, R.H. and Burnham, C.W., 1969, Experimental studies of pegmatite genesis: I. a model for the derivation and crystallization of granitic pegmatites, Economic Geology, 64, 843-864. Morgan, G.B.VI, and London, D., 1999, Crystallization of the Little Three layered pegmatite - aplite dike, Ramona District, California, Contributions to Mineralogy and Petrology, 136, 310-330. Patterson, J.E., 1996, Modeling of Layered Aplitic Pegmatite Dikes Using Ground Penetrating Radar, Little Three Mine, Ramona District, San Diego County, California. The Spirit of Inquiry, University of Arizona Undergraduate Research Program, 17. Patterson, J.E., 1997, The Little 3 Mine "Plug"; Evidence for a Zoned Plutonic Intrusion and a Plausible Parent Source of the Ramona Pegmatite District, Ramona, San Diego County, California, University of Arizona, unpublished B.Sc. Honors thesis, 152 pp. Schaller, W.T., and Fairchild, J.G., 1932, Bavenite, a Beryllium Mineral, Pseudomorphous after Beryl, from California, American Mineralogist, 17, 9, 409-422. Sinkankas, J., 1988, Field Collecting Gemstones and Minerals, Geoscience Press, Tucson, Arizona, 397 pp. Stern, L.A., Brown, G.E., Jr., Bird, D.K., Jahns, R.H., Foord, E.E., Shigley, J.E., and Spaulding, L.B., Jr., 1986, Mineralogy and Geochemical Evolution of the Little Three Pegmatite-aplite layered intrusive, Ramona, CA, American Mineralogist, 71, 3 / 4, 406-427. Thomas, R., Forster, H-J., and Heinrich, W., 2003, The behaviour of boron in a peraluminous granite-pegmatite system and associated hydrothermal solutions: a melt and fluid-inclusion study, Contributions to Mineralogy and Petrology, 144, 457-472. You can submit questions and comments in electronic formats only to Dr. David London at |